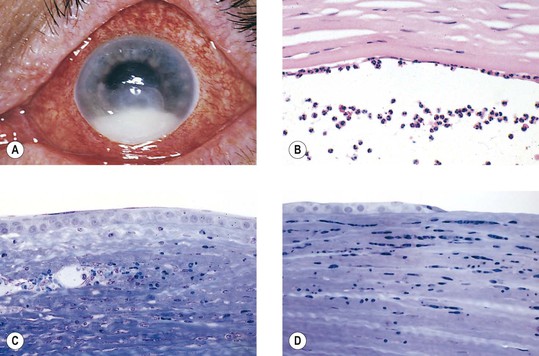
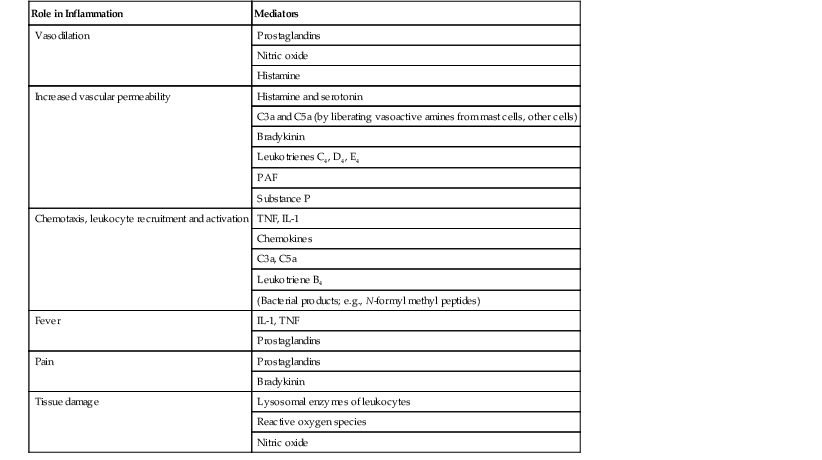
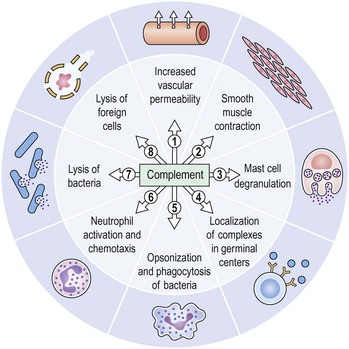
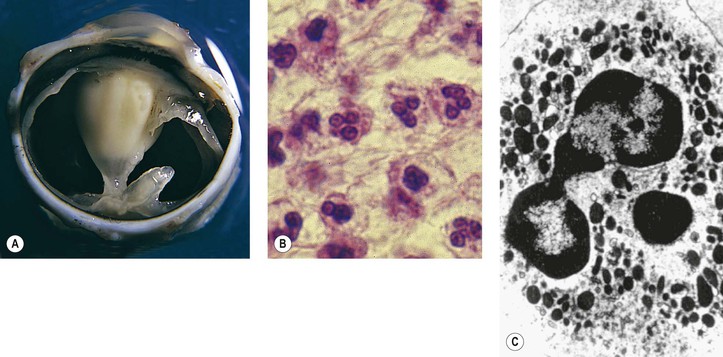
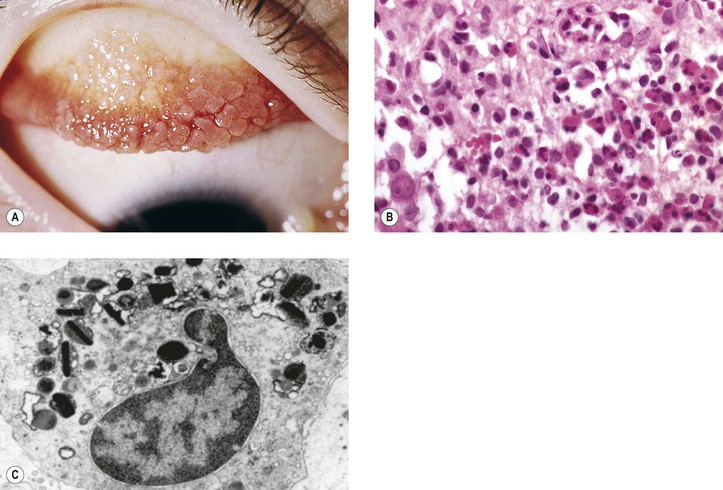
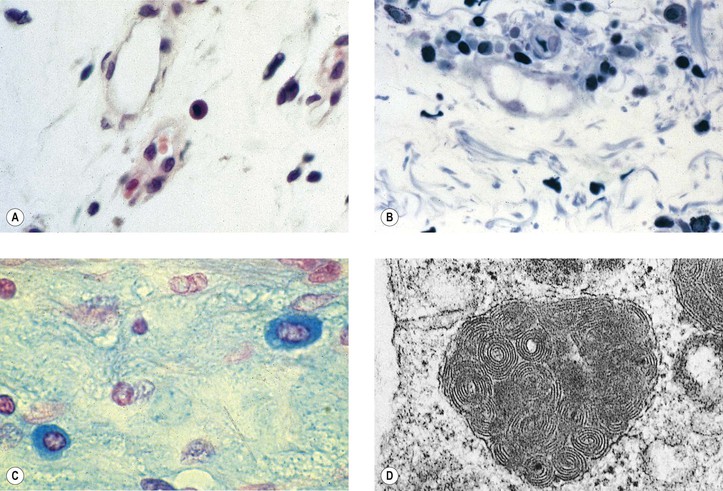
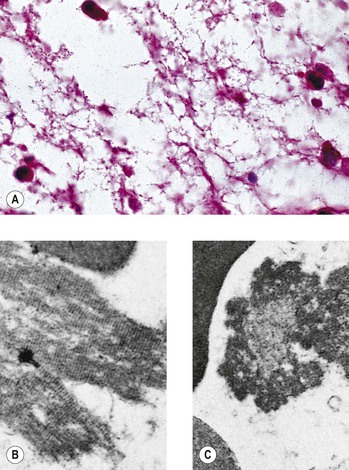
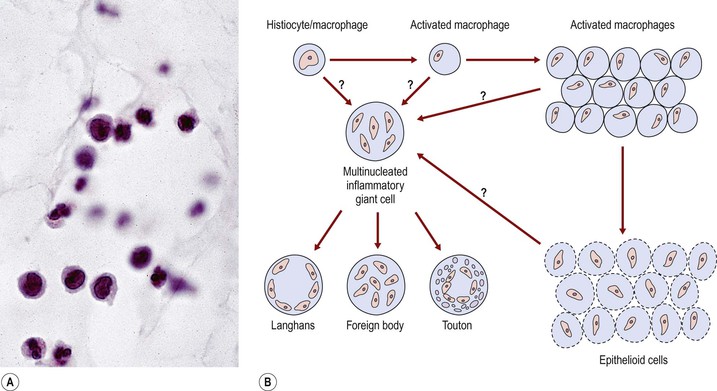
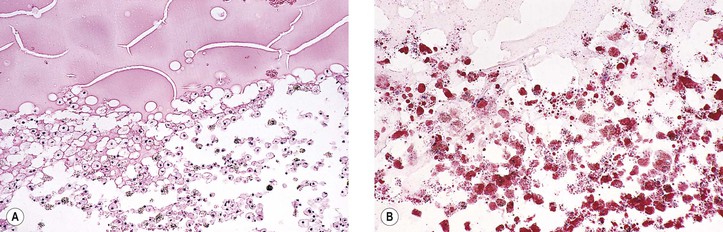
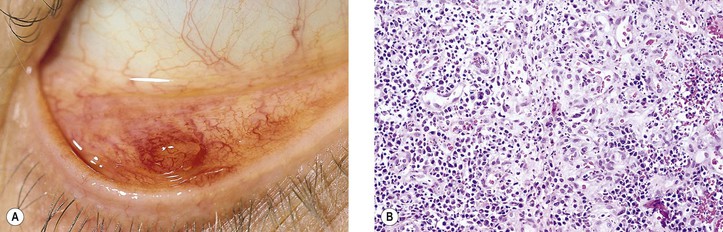
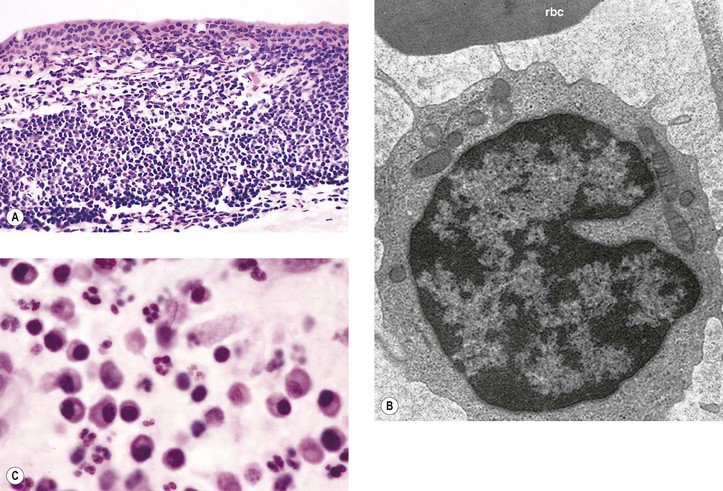
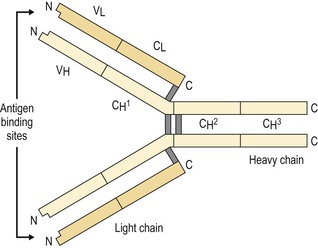
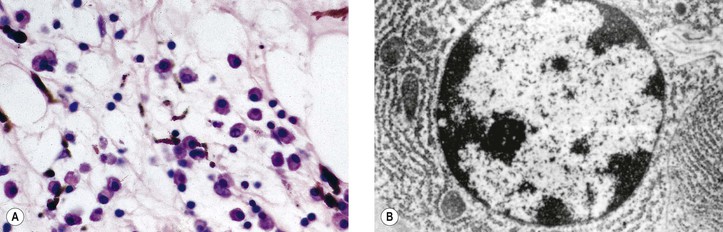
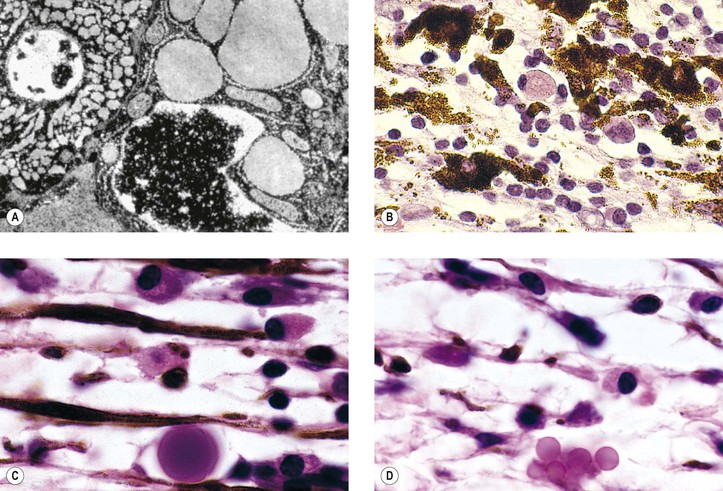
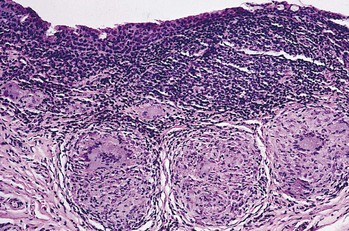
I. Acute (immediate or shock) phase (Fig. 1.1) A. Five cardinal signs: (1) redness (rubor) and (2) heat (calor)—both caused by increased rate and volume of blood flow; (3) mass (tumor)—caused by exudation of fluid (edema) and cells; (4) pain (dolor) and (5) loss of function (functio laesa)—both caused by outpouring of fluid and irritating chemicals. Table 1.2 lists the roles of various mediators in the inflammatory reaction. B. The acute phase is related to histamine release from mast cells and factors released from plasma (kinin, complement, and clotting systems). 4. The complement system consists of 35–40 proteins present in blood plasma or on cell surfaces. More than 45 genes encode proteins of complement components or related receptors, etc. Complement achieves its effect through a cascade of the separate components working in special sequences (Fig. 1.2). a. Major roles in immune defense against microorganisms and in clearing damaged host components. b. Complement proteins opsonize or lyse cells so they may injure healthy tissue, particularly when there is a defect in complement regulation. 2) C3 has a major role in complement activation and generation of immune responses. c. System, components and their genetic deficiency. 5. Prostaglandins (prostanoids), which have both inflammatory and anti-inflammatory effects, are 20-carbon, cyclical, unsaturated fatty acids with a 5-carbon ring and two aliphatic side chains. a. They are produced by mast cells, macrophages, endothelial cells, and others. d. Cyclooxygenase helps catalyze the biosynthesis of prostaglandins from arachidonic acid. e. Prostaglandins, cytokines, and leukotrienes function to dilate lymphatics at a site of injury. f. Prostaglandins play an important role in nociception and pain. 6. Major histocompatibility complex (MHC), called the human leukocyte antigen (HLA) complex in humans, is critical to the immune response. a. HLAs are present on all nucleated cells of the body and platelets. b. The three genetic loci belonging to HLA class I are designated by the letters HLA-A, HLA-B, and HLA-C. Class II MHC molecules are encoded at the locus HLA-D with three subregions HLA-DP, HLA-DQ , and HLA-DR. d. The HLA system is the main human leukocyte isoantigen system and the major human histocompatibility system. 2) HLA-B 5 is positive in a high percentage of patients who have Behçet’s disease. 7. Nonspecific soluble mediators of the immune system include cytokines, such as interleukins, which are mediators that act between leukocytes, interferons (IFNs), colony-stimulating factors (CSFs), tumor necrosis factor (TNF), transforming growth factor-β, and lymphokines (produced by lymphocytes). D. During the early period after injury, the leukocytes (predominantly the PMNs) stick to the vessel walls, at first momentarily, but then for a more prolonged time; this is an active process called margination (see Fig. 1.1C). F. PMNs (neutrophils; Fig. 1.3) are the main inflammatory cells in the acute phase of inflammation. 1. PMNs are born in the bone marrow and are considered “the first line of cellular defense.” 3. PMNs are the most numerous of the circulating leukocytes, making up 50–70% of the total. 5. The PMNs remove noxious material and bacteria by phagocytosis and lysosomal digestion. G. Eosinophils and mast cells (basophils) may be involved in the acute phase of inflammation. 1. Eosinophils (Fig. 1.4) originate in bone marrow, constitute 1 or 2% of circulating leukocytes, increase in number in parasitic infestations and allergic reactions, and decrease in number after steroid administration or stress. They elaborate toxic lysosomal components (e.g., eosinophil peroxidase) and generate reactive oxygen metabolites. 2. Mast cells (basophils; Fig. 1.5) elaborate heparin, serotonin, and histamine, and they are imperative for the initiation of the acute inflammatory reaction. H. The acute phase is an exudative2 phase (i.e., an outpouring of cells and fluid from the circulation) in which the nature of the exudate often determines and characterizes an acute inflammatory reaction. 2. Fibrinous exudate (Fig. 1.6) has high fibrin content (e.g., as seen clinically in a “plastic” aqueous). 3. Purulent exudate (see Figs 1.1 and 1.3) is composed primarily of PMNs and necrotic products (e.g., as seen in a hypopyon). 4. Sanguineous exudate is composed primarily of erythrocytes (e.g., as in a hyphema). II. Subacute (intermediate or reactive countershock and adaptive) phase3 B. PMNs at the site of injury release lysosomal enzymes into the area. 1. The enzymes directly increase capillary permeability and cause tissue destruction. C. Mononuclear (MN) cells (Fig. 1.7) include lymphocytes and circulating monocytes. 4. These cells are the “second line of cellular defense,” arrive after the PMN, and depend on release of chemotactic factors by the PMN for their arrival. a. Once present, MN cells can live for weeks, and in some cases even months. b. MN cells cause much less tissue damage than do PMNs, and they are more efficient phagocytes. 5. Monocytes have an enormous phagocytic capacity and are usually named for the phagocytosed material [e.g., blood-filled macrophages (erythrophagocytosis) and lipid-laden macrophages (Fig. 1.8)]. 6. Monocytes replace neutrophils as the predominate cell 24–48 hours after the onset of inflammation. F. In larger injuries, granulation tissue is produced. 1. Granulation tissue (Fig. 1.9) is composed of leukocytes, proliferating blood vessels, and fibroblasts. 3. Fibroblasts (see Fig. 1.9), which arise from fibrocytes and possibly from other cells (monocytes), proliferate, lay down collagen (Table 1.3), and elaborate ground substance. III. Chronic phase B. Chronic nongranulomatous inflammation is a proliferative inflammation characterized by a cellular infiltrate of lymphocytes and plasma cells (and sometimes PMNs or eosinophils). 1. The lymphocyte (Fig. 1.10) constitutes 15–30% of circulating leukocytes and represents the competent immunocyte. b. Two principal types of lymphocytes are recognized: (1) The bone marrow-dependent (or bursal equivalent) B lymphocyte is active in humoral immunity, is the source of immunoglobulin production (Fig. 1.11), and is identified by the presence of immunoglobulin on its surface; (2) the thymus-dependent T lymphocyte participates in cellular immunity, produces a variety of lymphokines, and is identified by various surface antigens. 2. The plasma cell (Fig. 1.13) is produced by the bone marrow-derived B lymphocyte, elaborates immunoglobulins (antibodies), and occurs in certain modified forms in tissue sections. a. Plasmacytoid cell (Fig. 1.14A and B): This has a single eccentric nucleus and slightly eosinophilic granular cytoplasm (instead of the normal basophilic cytoplasm of the plasma cell). b. Russell body (see Fig. 1.14C and D): This is an inclusion in a plasma cell whose cytoplasm is filled and enlarged with eosinophilic grapelike clusters (morular form), with single eosinophilic globular structures, or with eosinophilic crystalline structures; usually the nucleus appears as an eccentric rim or has disappeared. C. Chronic granulomatous inflammation is a proliferative inflammation characterized by a cellular infiltrate of lymphocytes and plasma cells (and sometimes PMNs or eosinophils). 1. Epithelioid cells (Fig. 1.15) are bone marrow-derived cells in the monocyte–histiocyte–macrophage family (Fig. 1.16). 2. Inflammatory giant cells, probably formed by fusion of macrophages rather than by amitotic division, predominate in three forms: a. Langhans’ giant cell (Fig. 1.17; see Fig. 1.15): This is typically found in tuberculosis, but it is also seen in many other granulomatous processes. When sectioned through its center, it shows a perfectly homogeneous, eosinophilic, central cytoplasm with a peripheral rim of nuclei. b. Foreign-body giant cell (Fig. 1.18): This has its nuclei randomly distributed in its eosinophilic cytoplasm and contains foreign material. c. Touton giant cell (Fig. 1.19), frequently associated with lipid disorders such as juvenile xanthogranuloma, appears much like a Langhans’ giant cell with the addition of a rim of foamy (fat-positive) cytoplasm peripheral to the rim of nuclei. 3. Three patterns of inflammatory reaction may be found in granulomatous inflammations: a. Diffuse type (Fig. 1.20A): This typically occurs in sympathetic uveitis, disseminated histoplasmosis and other fungal infections, lepromatous leprosy, juvenile xanthogranuloma, Vogt–Koyanagi–Harada syndrome, cytomegalic inclusion disease, and toxoplasmosis. The epithelioid cells (sometimes with macrophages or inflammatory giant cells or both) are distributed randomly against a background of lymphocytes and plasma cells. b. Discrete type (sarcoidal or tuberculocidal; see Fig. 1.20B): This typically occurs in sarcoidosis, tuberculoid leprosy, and miliary tuberculosis. An accumulation of epithelioid cells (sometimes with inflammatory giant cells) forms nodules (tubercles) surrounded by a narrow rim of lymphocytes (and perhaps plasma cells). c. Zonal type (see Fig 1.20C): This occurs in caseation tuberculosis, some fungal infections, rheumatoid scleritis, chalazion, phacoanaphylactic (phacoantigenic) endophthalmitis, toxocara endophthalmitis, and cysticercosis. 2) Granulation tissue often envelops the entire inflammatory reaction. TABLE 1.1 The Actions of the Principal Mediators of Inflammation IL-1, interleukin-1; MAC, membrane attack complex; TNF, tumor necrosis factor. (Reproduced from Table 2.4, Kumar R, Abbas A, DeLancey A et al.: Robbins and Cotran Pathologic Basis of Disease, 8th edn. Philadelphia, Saunders. © 2010 by Saunders, an imprint of Elsevier Inc.) TABLE 1.3 Heterogeneity of Collagens in the Cornea* * At least 10 genetically distinct collagens have been described in the corneas of different animal species, ages, and pathologies. Types I, II, III, and V collagens are present as fibrils in tissues. Types IV, VI, VII, and VIII form filamentous structures. Types IX and XII are fibril-associated collagens. The sizes of the structures are not completely known. Type II collagen is found only in embryonic chick collagen associated with the primary stroma. Type III collagen is found in Descemet’s membrane and in scar tissue. Types I and V form the heterotypic fibrils of lamellar stroma. Type VII has been identified with the anchoring fibrils, and type VIII is present only in Descemet’s membrane. Type IX collagen, associated with type II fibrils in the primary stroma, and type XII collagen, associated with type I/V fibrils, are part of a family of fibril-associated collagens with interrupted triple helices. Both type IX and type XII are covalently associated with a chondroitin sulfate chain. (Reproduced from Cintron C: The molecular structure of the corneal stroma in health and disease. In Podos SM, Yanoff M, eds: Textbook of Ophthalmology, vol. 8. London, Mosby. © Elsevier 1994.)
Basic Principles of Pathology
Inflammation
Definition1
Phases of Inflammation (Table 1.1)
Mediator
Principal Sources
Actions
Cell-Derived
Histamine
Mast cells, basophils, platelets
Vasodilation, increased vascular permeability, endothelial activation
Serotonin
Platelets
Vasodilation, increased vascular permeability
Prostaglandins
Mast cells, leukocytes
Vasodilation, pain, fever
Leukotrienes
Mast cells, leukocytes
Increased vascular permeability, chemotaxis, leukocyte adhesion and activation
Platelet-activating factor
Leukocytes, mast cells
Vasodilation, increased vascular permeability, leukocyte adhesion, chemotaxis, degranulation, oxidative burst
Reactive oxygen species
Leukocytes
Killing of microbes, tissue damage
Nitric oxide
Endothelium, macrophages
Vascular smooth muscle relaxation, killing of microbes
Cytokines (TNF, IL-1)
Macrophages, endothelial cells, mast cells
Local endothelial activation (expression of adhesion molecules), fever/pain/anorexia/hypotension, decreased vascular resistance (shock)
Chemokines
Leukocytes, activated macrophages
Chemotaxis, leukocyte activation
Plasma Protein-Derived
Complement products (C5a, C3a, C4a)
Plasma (produced in liver)
Leukocyte chemotaxis and activation, vasodilation (mast cell stimulation)
Kinins
Plasma (produced in liver)
Increased vascular permeability, smooth muscle contraction, vasodilation, pain
Proteases activated during coagulation
Plasma (produced in liver)
Endothelial activation, leukocyte recruitment
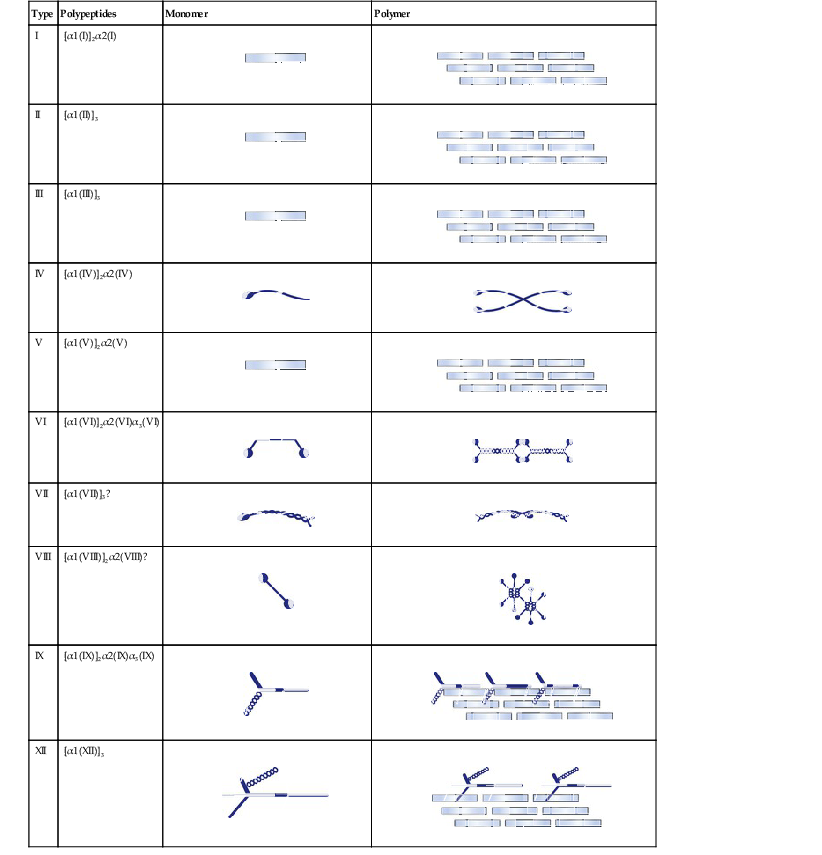
Type
Polypeptides
Monomer
Polymer
I
[α1(I)]2α2(I)


II
[α1(II)]3


III
[α1(III)]3


IV
[α1(IV)]2α2(IV)


V
[α1(V)]2α2(V)


VI
[α1(VI)]2α2(VI)α3(VI)


VII
[α1(VII)]3?


VIII
[α1(VIII)]2α2(VIII)?
IX
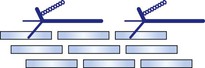
[α1(IX)]2α2(IX)α3(IX)
XII
[α1(XII)]3

![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Basic Principles of Pathology
1