Purpose
To investigate the morphologic features and clinical efficacy of barely visible Pascal (Optimedica Corporation) photocoagulation burns in diabetic macular edema (DME) using Fourier-domain optical coherence tomography (FD OCT) and fundus autofluorescence (AF).
Design
Interventional case series.
Methods
Retrospective evaluation of 10 eyes with newly diagnosed DME that underwent barely visible Pascal photocoagulation using an array of 10-μm, 10-millisecond photocoagulation burns. FD OCT and camera-based AF was performed at baseline and at 1 hour, 2 weeks, 4 weeks, and 12 weeks after laser. Changes in retinal thickening after laser treatment were measured using retinal thickness maps within the treated sector and the central foveal subfield.
Results
At 1 hour after treatment, burns were visualized partially with clinical biomicroscopy. AF demonstrated spots lacking autofluorescence that confirmed effective laser uptake within the Pascal arrays. Sequential changes in hyperreflectivity on FD OCT correlated with morphologic alterations seen on AF. Burns became increasingly hyperautofluorescent between 2 and 4 weeks. There were significant reductions in the retinal thickness within treated sectors on FD OCT at 2 weeks (26 ± 32 μm; P = .012) and 3 months after laser (20 ± 21 μm; P = .02) compared with baseline. Clinical biomicroscopic reduction of DME was the most common finding in 80%.
Conclusions
Barely visible 10-millisecond Pascal laser seems to produce an effect at the level of the inner and outer photoreceptor segments and apical retinal pigment epithelium, with minimal axial and lateral spread of burns. FD OCT confirmed spatial localization of AF signal changes that correlated with laser burn–tissue interactions over 3 months. The technique of lower-fluence barely visible 10-millisecond laser may reduce retinal edema within affected sectors and effectively treat DME with minimization of scar formation.
Diabetic macular edema (DME) remains the most common cause of visual loss in diabetic patients. The Early Treatment Diabetic Retinopathy Study (ETDRS) reported both the efficacy of visible end-point laser burns placed in a grid pattern up to the edge of the fovea and the focal photocoagulation of microaneurysms in DME. The ETDRS demonstrated that immediate photocoagulation with gray-white burns reduced moderate visual loss by 50% (from 24% to 12%) 3 years after initiation of treatment. However, after 12 months, 40% of treated patients had persistent DME with associated visual loss. It is well recognized that conventional (long duration, 100 milliseconds) laser scar expansion in the macula may be associated with enlarging atrophy of the retinal pigment epithelium (RPE) and paracentral scotomas over time.
Controversy remains as to whether a reduction in laser fluence may continue to treat macular edema effectively secondary to diabetic retinopathy. Barely visible (light gray, threshold) laser burns using a conventional long-pulse argon-green laser have been investigated in comparison with ETDRS gray-white photocoagulation burns in case series. Bandello and associates reported effective treatment of DME in the long term with threshold laser power. However, in a larger randomized study, the effect of mild macular grid conventional laser was not shown to be significantly more effective than modified ETDRS treatment methods. The modified ETDRS technique has recommended barely visible (light gray, threshold) burn intensity for conventional 50-millisecond macular laser photocoagulation. The Writing Committee for the Diabetic Retinopathy Clinical Research Network stated, “In an attempt to reduce these adverse effects, many retinal specialists now treat patients using burns that are lighter and less intense than what was originally specified in the ETDRS.”
Recent methods such as selective retina therapy report therapeutic effects using ultrashort pulse durations. Micropulse and subthreshold photocoagulation have been reported to produce equally effective or improved visual outcomes than standard modified ETDRS laser treatment. However, the absence of visible laser uptake may prompt inappropriate higher-power laser titration or unnecessary retreatment.
The Pascal (pattern scanning laser; Optimedica Corporation, Santa Barbara, California, USA) photocoagulator is a semiautomated laser delivery system that reduces procedural time by delivering multiple laser burns within a single application. Importantly, medium pulse durations (10 milliseconds, 20 milliseconds) may result in less destruction to the outer retina, compared with conventional laser burns, presumably because of reduced axial and lateral thermal spread.
Despite recent knowledge of the Pascal therapeutic parameters, benchmark laser parameters for treating DME have yet to be demonstrated in randomized clinical trials. Jain and associates reported threshold visible burns using 10-millisecond pulses in animal models. In clinical practice, it is important to image the in vivo effects of any lower fluence laser burns on human retina. This will ensure that laser photocoagulation is safe and that treatment may be better understood with monitoring over time.
Our current practice involves the imaging of barely visible 10-millisecond photocoagulation burns with Fourier-domain optical coherence tomography (FD OCT) and fundus autofluorescence (AF) at different points in short-term postlaser treatment. This article reports on the location of 10-millisecond burns within outer retina and the effects of laser on the RPE–neuroretinal tissue over time with FD OCT. In DME, the morphologic effects of barely visible laser burns were examined retrospectively in relation to clinical effect and macular thickness with FD OCT and AF.
Methods
Subjects
We retrospectively retrieved case notes for all patients who had undergone unilateral or bilateral barely visible 10-millisecond Pascal macular laser treatment for sight-threatening DME and further analyzed these cases. All patients had given informed consent for treatment, and all investigations were performed as part of routine care at Manchester Royal Eye Hospital, Manchester, United Kingdom. Exclusion criteria for this sample of subjects then included laser dosimetry that produced barely visible intensity burns (modified ETDRS light-gray grade) in all eyes, with no previous or repeated laser, medical, or surgical treatment performed. Exclusion criteria also included complete clinical and investigational data capture at the following follow-up visits: 1 hour after laser and at 2, 4, and 12 weeks after laser. Based on all stated exclusion criteria, we retrospectively identified and retrieved data for 10 eyes of 8 diabetic subjects who had undergone a single Pascal unilateral or bilateral macular laser treatment for DME, and these eyes then were studied.
Digital fundus photography, FD OCT, and AF images were analyzed retrospectively. FD OCT and AF images were examined at 1 hour, 2 weeks, and 4 weeks to assess the laser uptake, the laser burn coverage at the macula, and the presence of intraretinal edema.
Two experienced retina specialists (L.B.Y., P.E.S.) examined the amount of clinical thickening visible on biomicroscopy using either a Volk Digital High Mag lens (Volk Optical, Inc., Mentor, Ohio, USA) or Goldmann 3 mirror lens (Ocular Instruments, Bellevue, Washington, USA). Assessment of clinical effect was evaluated by the absence (positive clinical effect) or presence (negative clinical effect) of retinal thickening and edema on clinical biomicroscopy at 3 months after laser treatment. Additional clinical evaluation of macular edema involved comparison of prelaser and postlaser fundus color and red-free images to evaluate any changes in the number and extent of macular hard exudates. This was a retrospective study of standard laser management, and the Greater Manchester Central Research Ethics Committee waived ethical approval for this work. This study adhered to the tenets of the Declaration of Helsinki.
Pascal Photocoagulation System
This is a frequency-doubled neodymium:yttrium–aluminum–garnet solid-state laser with a wavelength of 532 nm. In full-grid treatment of DME, the A plus B pattern consisting of 4 concentric rings with 112 spots encircling the fovea was used. Each octant of the array contains 14 spots, and focal macular treatment comprised either single or multiple octants. To configure a horseshoe-shaped grid pattern, the supranasal and inferonasal octants are excluded to produce a total array of 84 spots.
Barely Visible Pascal Technique
Each patient had undergone laser power titration within a macular zone below the superior arcade, as described in the following steps. A Mainster Focal contact lens was used, with a 100-μm and 10-millisecond laser spot. Starting at 100 mW, power was increased in 25-mW steps until a visible (gray-white, modified ETDRS threshold) burn was produced. The power setting then was reduced by 25 mW (equivalent to fluence level of 3 J/cm 2 ) that produced a barely visible burn. Patients received one of full grid, horseshoe-shaped grid, or focal macula arrays ( Table ). Microaneurysms routinely are not photocoagulated directly. Spot spacing within all arrays was equivalent to one burn width. Immediately after laser application, all burns were barely visible clinically within each array.
| Eye | DME OCT Subtype | Pascal Laser Array | Energy Power (mW) | Energy Fluence (J/cm 2 ) | Pulse Duration (ms) | Spot Size (μm) | Clinical Regression in DME |
|---|---|---|---|---|---|---|---|
| 1 | SP | FM-DO | 125 | 16 | 10 | 100 | Yes |
| 2 | SP/CYST | FG | 125 | 16 | 10 | 100 | Yes |
| 3 | SP | FM-SO | 125 | 16 | 10 | 100 | Yes |
| 4 | SP | FM-SO | 100 | 13 | 10 | 100 | Yes |
| 5 | SP/CYST | HSG | 100 | 13 | 10 | 100 | No |
| 6 | SP/CYST | FG | 150 | 19 | 10 | 100 | No |
| 7 | SP | FM-SO | 100 | 13 | 10 | 100 | Yes |
| 8 | SP | HSG | 150 | 19 | 10 | 100 | Yes |
| 9 | SP | FM-SO | 150 | 19 | 10 | 100 | Yes |
| 10 | SP | FG | 125 | 16 | 10 | 100 | Yes |
Ocular Imaging
Fundus Autofluorescence
We used a fundus flash-camera system (Topcon TRC-50DX, type IA; Topcon Instruments, Newbury, United Kingdom) to image AF in all cases. The AF exciter filter has a bandwidth of 30 nm and central wavelength of 580 nm with 60% transmission. The AF barrier filter has a bandwidth of 40 nm and a central wavelength of 695 nm. AF images show the spatial distribution of signal intensities for each pixel in gray values. Dark signals correspond to low pixel values and a lack of autofluorescence, and bright signals correlate with high pixel values and increased autofluorescence because of a window-type effect.
Fourier-Domain Optical Coherence Tomography
FD OCT (Topcon, 3D OCT-1000) allows in vivo high-definition visualization of the retina. Important reflective signals include the inner highly reflective layer, which corresponds to the interface between the vitreous and the internal limiting membrane, and the outer highly reflective layer within outer retina. The thin band of high reflectivity immediately internal to the outer highly reflective layer is believed to correspond to the junction between the inner segment and outer segment of the photoreceptors, and the outer highly reflective layer has been reported to represent the melanin in the RPE.
Retinal Thickness Measurements
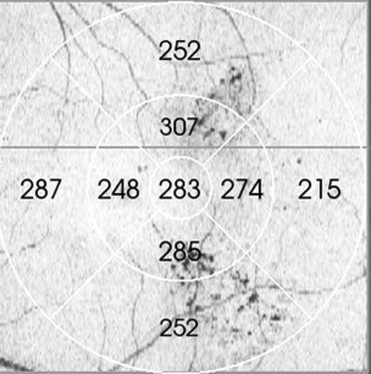
Changes in retinal thickening after barely visible laser were measured using retinal thickness maps at all time points. Each macular map is divided automatically by the FD OCT software into 9 ETDRS sectors, and the analysis program automatically calculates the average thickness for each sector. The central 500 μm subfield is designated the foveal sector, with 4 inner and 4 outer rings comprising the other 8 sectors ( Figure 1 ). The Pascal treatment arrays for all patients were extrapolated to the ETDRS map using shadowgram overlays and ETDRS grid within the 3-dimensional FD OCT analysis. For example, in Patient 1, Pascal focal macula treatment used 2 single octants to treat. The area of laser coverage was 6 ETDRS sectors, 3 inner and 3 outer sectors, because the foveal sector is not photocoagulated ( Figure 1 ). The central retinal thickness (CRT) measurements were evaluated at baseline, 2 weeks, 4 weeks, and 3 months after laser treatment.
We used the paired 2-tailed t test to explore changes in retinal thickness and Snellen visual acuity, and the null hypothesis was rejected for P values less than .05.
Results
A total of 10 eyes of 8 patients with DME treated with barely visible laser were retrieved retrospectively (6 males, 2 females). Five eyes had diffuse DME and 5 had focal DME. Macular edema was classified using time-domain OCT before laser intervention: sponge-like (7/10 eyes) and sponge-like with cystoid (3/10 eyes). There were no cases of serous retinal detachment ( Table ).
Postoperative Laser Parameters
After 1 hour, laser burns were partially visible within the treatment arrays using clinical biomicroscopy. AF imaging demonstrated spots lacking autofluorescence at 1 hour after laser treatment. Individual burns within the arrays were designated by spots lacking autofluorescence of uniform size and shape signal ( Figure 2 ). In all eyes, hyperautofluorescent laser spots were visible after 2 weeks within the treatment arrays. On color and red-free fundus photography, laser burns were not completely visible. Hyperautofluorescence remained present at 4 weeks in all eyes.




