Purpose
To investigate the association of a novel metric, percent tissue altered, with the occurrence of ectasia after laser in situ keratomileusis (LASIK) in eyes with normal corneal topography and to compare this metric with other recognized risk factors.
Design
Retrospective case-control study.
Methods
The study included 30 eyes from 16 patients with bilateral normal preoperative Placido-based corneal topography that developed ectasia after LASIK (ectasia group) and 174 eyes from 88 consecutive patients with uncomplicated LASIK and at least 3 years of postoperative follow-up. The following metrics were evaluated: age, preoperative central corneal thickness, residual stromal bed, Ectasia Risk Score System scores, and percent tissue altered, derived from [PTA = (FT + AD)/CCT], where FT = flap thickness, AD = ablation depth, and CCT = preoperative central corneal thickness.
Results
In the ectasia group, percent tissue altered ≥40 was the most prevalent factor (97%), followed by age <30 years (63%), residual stromal bed ≤300 μm (57%), and ectasia risk score ≥3 (43%) ( P < .001 for all). Percent tissue altered ≥40 had the highest odds ratio (223), followed by residual stromal bed ≤300 μm (74) and ectasia risk score ≥4 (8). Stepwise logistic regression revealed percent tissue altered ≥40 as the single most significant independent variable ( P < .0001).
Conclusions
Percent tissue altered at the time of LASIK was significantly associated with the development of ectasia in eyes with normal preoperative topography and was a more robust indicator of risk than all other variables in this patient population.
While most patients who have developed ectasia after laser in situ keratomileusis (LASIK) have, in retrospect, had identifiable risk factors, particularly irregular topographic patterns, that placed them at higher risk for this complication, ectasia cases in patients with normal preoperative topography still present a conundrum. Postoperative corneal ectasia most likely represents a reduction in biomechanical integrity below the threshold required to maintain corneal shape and curvature. This could theoretically occur when a cornea already destined to manifest ectasia has surgery, when a preoperatively weak but clinically stable cornea has surgery, or when a relatively normal cornea is weakened below a safe threshold.
As corneal tensile strength is not uniform throughout the central corneal stroma, with a progressive weakening in the deeper 60%, the relative extent of biomechanical alteration after refractive surgery, expressed as depth, definitely plays a role in postoperative weakening. Flap thickness factors directly into this alteration, as the anterior lamellar flap does not contribute significantly to postoperative corneal tensile strength.
There is an integrated relationship between preoperative corneal thickness, ablation depth, and flap thickness in determining the relative amount of biomechanical change that has occurred after a LASIK procedure. We have investigated a metric, the percent of anterior tissue depth altered, that describes this interaction during excimer laser refractive surgery, which for LASIK can be described as:
P T A = ( F T + A D ) / C C T
The purpose of this study was to investigate the association of the percent tissue altered with the occurrence of ectasia after LASIK in eyes with normal preoperative Placido disk–based corneal topography and to compare this metric to other recognized risk factors.
Methods
This retrospective comparative case-control study included eyes that developed ectasia after LASIK for myopia and myopic astigmatism with bilateral normal preoperative Placido disk–based corneal topography identified in the authors’ institutions (ectasia group), and a contemporaneous population composed of eyes with bilateral normal preoperative Placido disk–based corneal topography that underwent uncomplicated LASIK for myopia and myopic astigmatism at 1 author’s institution (M.R.S.) without developing ectasia with at least 3 years of postoperative follow-up (control group). The study was approved by the Federal University of Rio de Janeiro Institutional Review Board (IRB) and the Emory University IRB before the study began and was conducted in adherence with the tenets of the Declaration of Helsinki.
The following preoperative information was obtained for both ectasia and control cases: patient age; sex; preoperative central corneal thickness (CCT) based on ultrasound pachymetry; Placido disk–based color corneal topography; manifest refraction spherical equivalent (MRSE) in diopters (D); and best spectacle-corrected distance visual acuity (CDVA). Perioperative and postoperative information included date of surgery (year), LASIK flap thickness (measured), calculated central ablation depth, and calculated residual stromal bed thickness (CCT − measured flap thickness − calculated central ablation depth). LASIK flap measurements were taken intraoperatively with ultrasound pachymetry using the subtraction method or postoperatively with optical coherence tomography (OCT) or confocal microscopy. Specific cut-off values of recognized risk factors were identified for comparative purposes. These included individual metrics used in the Ectasia Risk Score System: patient age in years (<30), CCT (≤510 μm), residual stromal bed (≤300 μm), and preoperative myopia (≥8 D). We also calculated the original Ectasia Risk Score System summed values for each group.
The percentage of anterior tissue depth altered during LASIK was obtained from the equation: Percent Tissue Altered = (Flap Thickness + Ablation Depth)/preoperative Central Corneal Thickness. Percent tissue altered was calculated for all eyes in the study and represents the percentage of anterior tissue that is modified during LASIK refractive surgery.
For this study, normal preoperative topography was defined as regular and symmetric patterns (including round, oval, or symmetric bowtie patterns) or mildly asymmetric (steepening ≤0.5 D and without a skewed radial axis) based on Placido disk analysis. All patients included had bilateral normal Placido-based topographic patterns preoperatively.
Postoperative corneal ectasia was defined as progressive inferior steepening, increasing myopia, and astigmatism; loss of uncorrected visual acuity; and often loss of best-corrected acuity (CDVA). All ectasia patients included in the analysis had uneventful surgery with initially good outcomes prior to developing the aforementioned findings.
Inclusion criteria for both ectasia and control cases included having all relevant necessary preoperative and perioperative information for analysis. Eyes that developed ectasia with abnormal preoperative Placido disk–based color topography were excluded from this study. Topographically normal fellow eyes of eyes that had abnormal patterns were excluded. Patients with significant between-eye topographic asymmetry or family history of keratoconus were excluded.
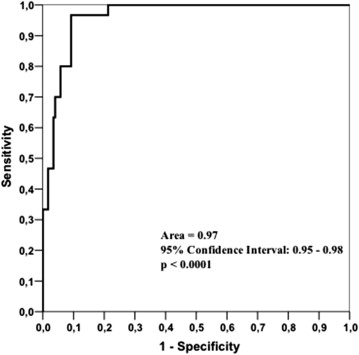
Statistical analyses were performed using JMP software (version 8.0; SAS Institute, Inc, Cary, North Carolina, USA). While percent tissue altered is a continuous variable, it was also evaluated as a discontinuous risk factor (cut-off) variable using 40 based on analysis of receiver operating characteristic (ROC) curve, which revealed a cut-off of 40.0 as the value with the maximized sum of sensitivity (97%) and specificity (89%), shown in Figure 1 and Table 1 . Odds ratio values and the 95% confidence interval were calculated. The odds ratio represents the odds that an outcome will occur given a particular factor (risk factor, in case odds ratio >1, or protective factor, in case odds ratio <1), compared to the odds of the outcome occurring in the absence of that factor. Logistic stepwise regression was performed to investigate the significance as predictors of the event (ectasia). Normality of data was evaluated with the Kolmogorov-Smirnov test. When parametric analysis was not possible the nonparametric Wilcoxon analysis was used to compare data between the 2 groups. The analysis of primary outcome measures was based on a non-normal distribution of the data. When parametric analysis was possible, the Student t test was used to compare the outcomes. Categorical variables were compared using either χ 2 or the Fisher test as appropriate. To correct for multiple comparisons performed in this study (Bonferroni method), only P values less than .007 were considered significant. Continuous data were expressed as mean values ± standard deviation and confidence interval. Categorical variables were expressed as frequency (n) and percent (%).
| Cut-off Percent Tissue Altered Value (%) | Sensitivity (%) | Specificity (%) |
|---|---|---|
| 48 | 27 | 100 |
| 47 | 33 | 100 |
| 46 | 33 | 98 |
| 45 | 53 | 97 |
| 44 | 63 | 96 |
| 43 | 77 | 94 |
| 42 | 87 | 91 |
| 41 | 90 | 91 |
| 40 a | 97 | 89 |
| 39 | 97 | 87 |
| 38 | 97 | 83 |
| 37 | 97 | 82 |
| 36 | 97 | 79 |
| 35 | 100 | 72 |
| 34 | 100 | 64 |
a The results of this table are derived from receiver operating characteristic (ROC) curve, and revealed a cut-off of 40% as the value with the maximized sum of sensitivity and specificity; PTA = Percent Tissue Altered (Flap Thickness + Ablation Depth)/Central Corneal Thickness.
Results
The study population included 30 eyes from 16 patients in the ectasia group and 174 eyes from 87 consecutive patients in the control group. Ectasia cases had LASIK between 2002 and 2010; control cases had LASIK between 2008 and 2010, with a minimum of 3 years of uneventful follow-up. Table 2 shows the demographic data of eyes that developed ectasia with normal preoperative topography. In the ectasia group, 14 patients (28 eyes) developed ectasia bilaterally, while 2 (Cases 5 and 6) developed unilateral ectasia after bilateral LASIK. The fellow eyes of Cases 5 and 6 that did not develop ectasia presented percent tissue altered values of 38 and 37, respectively.
| Case | Age (y) | Sex | MRSE | Preoperative CCT (μm) | Flap Thickness (μm) | Flap Creation Method | Tissue Ablated (μm) | RSB (μm) | ERSS | PTA |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 27 | F | −5.33 | 542 | 161 | MK | 80 | 300 | 1 | 45 |
| 2 | 27 | F | −5.00 | 540 | 164 | MK | 75 | 301 | 1 | 44 |
| 3 | 32 | F | −6.37 | 524 | 141 | FS | 95.5 | 288 | 1 | 45 |
| 4 | 32 | F | −6.37 | 522 | 143 | FS | 95.5 | 287 | 1 | 45 |
| 5 | 23 | M | −6.07 | 532 | 160 | FS | 91 | 281 | 3 | 47 |
| 6 | 39 | M | −7.80 | 568 | 177 | MK | 117 | 274 | 2 | 52 |
| 7 | 34 | M | −8.67 | 541 | 162 | MK | 130 | 249 | 4 | 54 |
| 8 | 34 | M | −8.67 | 540 | 130 | MK | 130 | 280 | 2 | 48 |
| 9 | 50 | F | −7.47 | 580 | 150 | MK | 112 | 318 | 0 | 45 |
| 10 | 50 | F | −6.80 | 580 | 150 | MK | 102 | 328 | 0 | 43 |
| 11 | 28 | F | −7.00 | 529 | 160 | MK | 105 | 264 | 3 | 50 |
| 12 | 28 | F | −6.47 | 526 | 159 | MK | 97 | 270 | 3 | 49 |
| 13 | 27 | M | −5.93 | 512 | 131 | FS | 89 | 292 | 2 | 43 |
| 14 | 27 | M | −4.80 | 529 | 140 | FS | 72 | 317 | 1 | 40 |
| 15 | 26 | M | −7.53 | 540 | 153 | MK | 113 | 274 | 3 | 49 |
| 16 | 26 | M | −7.00 | 540 | 122 | MK | 105 | 313 | 1 | 42 |
| 17 | 29 | F | −4.93 | 588 | 186 | MK | 74 | 328 | 1 | 44 |
| 18 | 29 | F | −5.93 | 594 | 180 | MK | 89 | 325 | 1 | 45 |
| 19 | 27 | M | −3.00 | 550 | 201 | MK | 45 | 304 | 1 | 45 |
| 20 | 27 | M | −3.07 | 546 | 189 | MK | 46 | 311 | 1 | 43 |
| 21 | 26 | M | −7.75 | 525 | 130 | MK | 84 | 311 | 1 | 41 |
| 22 | 26 | M | −9.00 | 530 | 130 | MK | 105 | 295 | 4 | 44 |
| 23 | 30 | M | −5.63 | 527 | 140 | MK | 80 | 307 | 1 | 42 |
| 24 | 30 | M | −6.75 | 509 | 150 | MK | 94 | 265 | 3 | 48 |
| 25 | 21 | F | −5.63 | 497 | 170 | MK | 88 | 239 | 9 | 52 |
| 26 | 21 | F | −4.63 | 480 | 155 | MK | 71 | 254 | 7 | 47 |
| 27 | 21 | M | −4.75 | 529 | 125 | MK | 101 | 303 | 3 | 43 |
| 28 | 21 | M | −5.25 | 522 | 120 | MK | 65 | 337 | 3 | 35 |
| 29 | 30 | M | −6.00 | 498 | 110 | MK | 98 | 290 | 4 | 42 |
| 30 | 30 | M | −5.50 | 500 | 110 | MK | 91 | 299 | 4 | 40 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


