Purpose
To evaluate the short-term efficacy, predictability, stability, and safety of the foldable Artiflex Toric phakic intraocular lens (pIOL; Ophtec) for the correction of myopia with astigmatism.
Design
Prospective, nonrandomized multicenter study.
Methods
One hundred fifteen eyes of 73 patients were implanted with an Artiflex Toric pIOL. Mean implanted spherical and cylindrical powers were −7.10 ± 2.70 diopters (D) and −2.14 ± 0.80 D, respectively. Total follow-up was 6 months. Outcome parameters included uncorrected visual acuity (UCVA), best spectacle-corrected visual acuity (BSCVA), subjective manifest refraction (used for vector analysis), intraocular pressure, and endothelial cell density (ECD).
Results
At 6 months, 99.0% of eyes had a UCVA of ≥20/40, and 81.8% of eyes were ±0.5 D of the intended refraction. In 74.5% postoperative UCVA was equal to or better than preoperative BSCVA; 2 eyes lost 2 or more lines of BSCVA postoperatively because of the development of synechiae. In 75.5% of eyes the remaining cylinder was ±0.5 D. There was a significant decrease in ECD after 3 months (4.8% ± 11.9%), with no additional decline between 3 and 6 months. There were few complications, except for the incidence of pigment and nonpigment precipitates (14.8% and 12.2%, respectively at 6 months). Mean misalignment was 0.6 ± 1.5 degrees (range 0–8).
Conclusions
The Artiflex Toric pIOL effectively and safely corrects myopia and astigmatism in the short term, with stable and predictable visual results after 6 months, when strict inclusion criteria are applied. Deposition of pigment and nonpigment precipitates on the pIOL warrants further investigation.
Several treatment options are available to correct myopia with astigmatism. Laser refractive surgery, such as photorefractive keratectomy (PRK) and laser in situ keratomileusis (LASIK), can only be used in mild to moderate myopia. Especially in thin corneas, these refractive procedures cannot adequately and safely correct high ametropia, because of the risk of post-LASIK ectasia. Although these procedures are adjustable, they can never be reversed. Another available treatment option is refractive lens exchange with implantation of a toric intraocular lens (IOL). However, young refractive patients lose their ability to accommodate, which makes them dependent on reading glasses. Phakic intraocular lenses (pIOLs) can correct high myopia and astigmatism, and patients keep their ability to accommodate. Also, the quality of vision in patients with high myopia has been reported to be better after pIOL implantation as compared to LASIK.
The first iris-fixated pIOL was introduced in 1986 (mentioned iris-fixated pIOLs throughout this manuscript are made by Ophtec BV, Groningen, The Netherlands). The first Artisan pIOL had a biconcave design, which was changed into a convexo-concave model in 1991 to reduce potential complications. In the last decade, this lens has proven to be stable, predictable, and safe, when strict inclusion criteria are applied to maintain a safe distance from the pIOL to the endothelium and natural lens. In 2001 the Toric Artisan pIOL was marketed in Europe after a multicenter study showed that it effectively reduced or eliminated high ametropia and astigmatism in only 1 procedure.
The developments in cataract surgery with IOL placements through smaller incisions led to the development of a foldable iris-fixated pIOL type in 2005: the Artiflex Myopia pIOL, which can be inserted through a 3.2-mm incision. This Artiflex pIOL may offer an advantage over the Artisan pIOL because of its smaller incision size, with a decrease in surgically induced astigmatism and a faster visual recovery. Combining the toric technology with the foldable Artiflex pIOL resulted in the development of the Artiflex Toric. This new toric pIOL corrects myopia and astigmatism and has a flexible, cylindrical 6-mm optic, which is different from the 5-mm rigid, spherical optic of the Toric Artisan pIOL. It is a 3-piece lens that consists of a flexible optical part made of ultraviolet-absorbing silicone and 2 rigid haptics made of polymethyl methacrylate (PMMA) ( Figure 1 ). It is available in spherical powers of −1.0 to −13.5 diopters (D) (in increments of 0.50 D) and cylindrical powers of −1.0 to −5.0 D (in increments of 0.50 D). The sum of spherical and cylindrical power should not exceed −14.5 D.
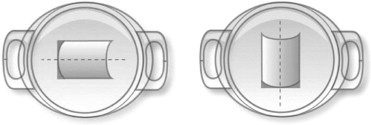
Most surgeons prefer to place the non-toric Artiflex pIOL in the horizontal position. To facilitate the surgeons’ implantation of the Artiflex Toric pIOL in the position to which they are accustomed, the cylindrical correction can be manufactured with the cylinder axis at 0 degrees or 90 degrees ( Figure 2 ). When the cylinder axis of the patient is between 0 and 45 degrees or between 135 and 180 degrees, the manufacturer recommends a pIOL with the cylinder axis at 0 degrees. The Toric pIOL should be implanted in the axis of the refractive cylinder of the patient. However, when the patient presents with a cylinder between 45 and 135 degrees, the manufacturer recommends a pIOL with the cylinder axis at 90 degrees. In this case, the implantation axis in which the pIOL should be positioned will be the axis of the refractive cylinder of the patient plus 90 degrees. To ensure a proper placement of the Artiflex Toric pIOL and avoid implantation errors, all surgeons receive an illustration of the correct toric pIOL position.
The purpose of this study was to evaluate the short-term safety and effectiveness of this new Artiflex Toric pIOL for the correction of myopia and astigmatism.
Patients and Methods
Study Design and Study Population
In this prospective, nonrandomized, open-label multicenter study, 115 eyes of 73 patients were analyzed. Toric Artiflex pIOLs were implanted in the period between September 2007 and March 2009 by 7 investigators. The inclusion criteria for the conducted study were: age between 18 and 60 years; stable refraction for a minimum of 12 months prior to the surgery and regular astigmatism between 0.75 and 4.50 D; a minimum best-corrected spherical equivalent of 0.5; a difference between cycloplegic and manifest refraction of <0.75 D; anterior chamber depth (ACD) ≥3.2 mm (measured from center of corneal epithelium to crystalline lens); no corneal, pupil, or iris abnormalities; pupil ≤7.0 mm in scotopic light conditions; endothelial cell density (ECD) count according to age, with a minimum of >2000 cells/mm 2 for the highest age group; no prior intraocular or corneal surgery; and no history of glaucoma, monocular vision, amblyopia, or diabetes mellitus. Immune-compromised patients and pregnant women were excluded from the study.
Preoperative and Postoperative Examinations
All included subjects were evaluated preoperatively and 1 day, 1 week, 1 month, and 3 and 6 months postoperatively. At the 6-month follow-up visit 6 eyes were lost to follow-up (5.2%). Examinations during the preoperative visit included uncorrected visual acuity (UCVA), best spectacle-corrected visual acuity (BSCVA), subjective and objective manifest refraction, cycloplegic subjective refraction, axial length, ACD, intraocular pressure (IOP) by applanation tonometry, slit-lamp and fundus examination, pupillometry, keratometry (Pentacam HR [software version 2.73]; Oculus, Wetzlar, Germany), Orbscan II (software version 3.10.31; Bausch & Lomb, Munich, Germany), and Humphrey Atlas topographer (software version A12.2; Carl Zeiss, Jena, Germany), and ECD measurements by specular microscopy (different types of Topcon [Tokyo, Japan] and Konan Medical Inc [Hyogo, Japan] devices). Before the examinations, contact lenses were not worn for at least 2 weeks if using rigid contact lenses or at least 3 days if using soft contact lenses.
All postoperative visits included UCVA, BSCVA, subjective and objective manifest refraction, IOP, slit-lamp biomicroscopy to evaluate lens position and axis of enclavation, and keratometry. Furthermore, ECD measurements were performed at 3 and 6 months postoperatively, using a noncontact specular microscope. Three consecutive images of the central corneal endothelium were obtained and the mean of these 3 measurements was used for statistical analysis. All examinations were performed by the clinical investigator or by a qualified optometrist supervised by the investigator.
Lens Power Calculation
The power calculation of the Artiflex Toric pIOL was performed by Ophtec BV (Groningen, The Netherlands) using the Van der Heijde formula. This formula uses the corneal curvature, adjusted ACD (ACD − 0.9), and the patient’s subjective refraction to calculate the spherical and cylindrical power of the pIOL. Calculations were performed for 2 cylinder axes perpendicular to each other.
Surgical Procedure
All surgeons had at least 1 year of experience with implantation of Artiflex Myopia pIOLs and Artisan Toric pIOLs. There were some minor differences in surgical approach among the 7 surgeons involved in the study. Preoperatively, 1 surgeon marked the enclavation spots on the iris with an argon laser in 20.9% of eyes (24/115). Furthermore, in 51.3% of the eyes (59/115) limbal reference marks were placed using a special surgical marker with the patient sitting in an upright position to correct for cyclotorsion. Intraoperatively, the actual implantation axis was marked using a toric axis marker. Alternatively, 2 surgeons used digital photography in 27.8% of eyes (32/115), using the iris structures (eg, crypts, pigment, vessels) as a natural marker for lens enclavation. Miotic eye drops (pilocarpine 1% or 2%) were administered to prepare the iris for lens fixation and reduce the risk of lens touch during surgery.
For implantation of the Artiflex Toric pIOL a 3.2-mm (range 3.0–3.4 mm) primary incision was made at the 12 o’clock position in 85.2% (98/115) of eyes. Furthermore, 2 1.2-mm paracenteses were made at 2 and 10 o’clock for access of instruments to fixate the lens in the mid-peripheral iris stroma. For some surgeons (9.6%; 11/115) the site of the approach depended on the predetermined location of implantation and cylindrical axis. The surgeons used several incision types: clear corneal incision in 13.0% (15/115) of eyes, limbal incision in 86.1% (99/115) of eyes, and corneoscleral incision in 0.9% (1/115) of eyes.
After intracameral injection of a miotic agent and the insertion of a cohesive viscoelastic substance, the Artiflex Toric lens was inserted with a specially designed spatula (Ophtec BV, Groningen, The Netherlands). The lens was fixated using a disposable enclavation needle (Ophtec BV, Groningen, The Netherlands), aligning the haptics on the cylindrical axis (or perpendicular to the axis, depending on the lens used) according to the alignment marks on the cornea. In 73.9% (85/115) of the eyes an iridectomy was performed at 12 o’clock during surgery to avoid pupillary block glaucoma. Two surgeons (11.3%; 13/115 of eyes) always made an iridotomy during the procedure and another surgeon (13.0%; 15/115 of eyes) performed the iridotomy preoperatively using a yttrium-aluminum-garnet (YAG) laser. At the end of the procedure, the viscoelastic substance was exchanged for balanced salt solution (Alcon, Fort Worth, Texas, USA). Safety sutures were placed depending on the preference of the surgeon. One surgeon injected cefuroxime (Zinacef; GlaxoSmithKline, Brentford, UK) into the anterior chamber at the end of the procedure. Postoperatively, a steroid/antibiotic combination was used for at least 3 weeks.
Vector Analysis of Refractive Astigmatism
Preoperative and postoperative astigmatism were evaluated using the vector analysis according to Alpins. We calculated refractive astigmatism to the corneal plane by adjusting for a back vertex distance of 12 mm. Individual magnitude (D) and axis values were transformed into rectangular x and y coordinates and used to calculate the following vectors: target-induced astigmatism (TIA), which represents the change (in magnitude and axis) that the toric pIOL was intended to induce; surgically induced astigmatism (SIA), which is the astigmatic change the surgery actually induced; difference vector (DV), which represents the vector that enables the achieved astigmatic outcome to achieve the intended astigmatic outcome (this is an absolute measure of success and is preferably zero); magnitude of error (ME), which is defined as the arithmetic difference between the magnitudes of the SIA and TIA (positive for overcorrection and negative for undercorrection); angle of error (AE), which is the angle between the SIA vector and TIA vector (positive if the achieved correction is counterclockwise to the intended axis and negative if the achieved correction is clockwise to the intended axis); flattening effect (FE), which is the amount of astigmatism reduction achieved at the intended meridian of treatment (TIA meridian); flattening index, which is calculated by dividing the FE by the TIA and is preferably 1.0; correction index, which is calculated by the ratio of the magnitude of SIA to the magnitude of TIA (preferably 1.0; >1.0 with overcorrection; <1.0 with undercorrection); and index of success, which is calculated by dividing DV by TIA (relative measure of success and is preferably zero). Furthermore, overall mean magnitude of residual astigmatism and the residual astigmatism at the meridian of treatment were calculated.
Statistical Analysis
All data were collected on standardized case-record forms and then entered in a Microsoft Access 2003 database; data were exported to Microsoft Excel (Microsoft Office Excel 2003) and SPSS (SPSS for Windows, version 15.0; SPSS Inc, Chicago, Illinois, USA) for statistical analysis. Continuous variables were described as mean ± standard deviation (SD). Snellen visual acuities were converted to logarithm of the minimal angle of resolution (logMAR) values for statistical analysis. Pairwise comparisons were used to evaluate the results over time. To compare differences between groups, independent sample t tests or 1-way ANOVA analyses were applied. Preoperative and postoperative astigmatism were evaluated using vector analysis according to Alpins. Postoperative changes in astigmatism vectors were analyzed using a repeated-measures analysis of variance (ANOVA). A P value < .05 was considered statistically significant.
Results
Mean age of the included patients was 37.7 ± 8.0 years (range 20 to 60 years). Sixty-seven of the lenses (58.3%; 67/115) were implanted in female eyes. Left (47.8%; 55/115) and right eyes were equally implanted. Mean preoperative spherical equivalent (SE) was −7.53 ± 2.70 D, with a mean absolute cylinder of −2.18 ± 0.79 D (range −0.75 to −4.50 D). Mean ACD was 3.65 ± 0.25 mm (range 3.20 to 4.28 mm), and the mean axial length was 26.20 ± 1.24 mm (range 23.53 to 29.32 mm). Mean scotopic pupil size was 5.70 ± 0.97 mm. Figure 3 shows the lens power distribution of the implanted Artiflex Toric pIOLs.
Short-term Safety
The mean preoperative BSCVA was 0.03 ± 0.09 logMAR. After 1, 3, and 6 months, the mean postoperative BSCVA was −0.01 ± 010, −0.01 ± 0.08, and −0.02 ± 0.09 logMAR, respectively. Ninety-nine percent (99.0%; 104/105), 100.0% (100/100), and 100.0% (102/102) of eyes had a BSCVA of 20/40 or better at 1, 3, and 6 months postoperatively, respectively. Furthermore, the safety indices (ratio of mean postoperative BSCVA to mean preoperative BSCVA) at these 3 follow-up visits were 1.12, 1.13, and 1.13, respectively. A total of 95.1% (97/102), 83.3% (85/102), and 40.2% (41/102), of eyes had a BSCVA of 20/25, 20/20, and 20/16 or better, respectively, 6 months after pIOL implantation. In 90.5% (95/105), 94.0% (94/100), and 92.2% (94/102) of eyes, there was no change or an improvement in Snellen lines at 1, 3, and 6 months postoperatively compared to preoperative BSCVA, respectively ( Figure 4 ). At 6 months postoperatively, 2.0% (2/102) lost 2 lines of Snellen BSCVA.
Short-term Efficacy
At 1, 3, and 6 months postoperatively the UCVA was 0.05 ± 0.14, 0.03 ± 0.12, and 0.02 ± 0.10 logMAR, respectively. The efficacy index (mean postoperative UCVA/mean preoperative BSCVA) was 0.99, 1.02, and 1.04 at 1, 3, and 6 months after Artiflex Toric pIOL implantation, respectively. UCVA was 20/40 or better in 97.1% (102/105), 99.0% (98/99), and 99.0% (101/102) of the cases at 1, 3, and 6 months postoperatively, respectively. A total of 90.2% (92/102), 65.7% (67/102), and 29.4% (30/102) of eyes had a UCVA of 20/25, 20/20, and 20/16 or better respectively, 6 months after pIOL implantation. The postoperative UCVA was equal to or better than preoperative BSCVA in 66.7% (70/105), 76.7% (76/99), and 74.5% (76/102) of eyes at 1, 3, and 6 months after pIOL implantation, respectively. Postoperative UCVA for all follow-up visits is visualized in Figure 5 .
Predictability
The intended vs achieved correction at 6 months of follow-up is presented in Figure 6 . After 6 months, 81.4% (83/102) of eyes were within ±0.5 D and 99.0% (101/102) of eyes within ±1.0 D from the intended refraction. All eyes were within ±1.1 D of the intended correction 6 months after pIOL implantation.
The absolute refractive cylinder was reduced from 2.20 ± 0.84 D (range 0.75 to 4.50 D) preoperatively to 0.38 ± 0.41 D (range 0.00 to 2.00 D) at 6 months postoperatively. In 75.5% (77/102) of eyes, the remaining cylinder was 0.50 D or less and in 96.0% (98/102) it was 1.00 D or less.
Stability
The change in SE from 1 day to 6 months postoperatively was not statistically significant, with a mean SE of −0.05 ± 0.45 D after 1 day and −0.06 ± 0.31 D after 6 months ( P > .05). Stability of the absolute cylinder values are demonstrated in Figure 7 , with no significant changes between 1 day and 6 months after implantation ( P > .05). At 1, 3, and 6 months postoperatively, 86.7% (90/105), 88.8% (88/99), and 93.0% (95/102) of eyes had a SE within ±0.5 D from emmetropia and 98.1% (103/105), 94.9% (94/99), and 100.0% (102/102) were within ±1.0 D from emmetropia, respectively. There were no significant changes in BSCVA between the postoperative follow-up visits, indicating a stable visual acuity after Artiflex Toric implantation ( P > .05). UCVA improved from 1 day postoperatively to 3 and 6 months after pIOL implantation ( P < .05).
Mean preoperative corneal astigmatism of 2.15 ± 0.70 D was significantly reduced to 1.78 ± 0.61 D at 6 months postoperatively ( P < .05). There was no significant difference in surgically induced astigmatism between the clear corneal and the limbal incisions ( P > .05). The effect of the corneoscleral incision could not be compared to the other incision types, since it was only used in 1 eye.
The Table shows the results of the Alpins vector analysis based on preoperative and postoperative refractive astigmatism. Figure 8 demonstrates preoperative refractive astigmatism and achieved refractive astigmatism at 6 months postoperatively. The difference vector at 6 months was significantly lower when compared to 1 week postoperatively ( P = .048). All other calculated vectors were not significantly different in the repeated-measures ANOVA between all follow-up visits (all with P > .05).



