Purpose
To describe the clinical characteristics and outcome of eyes with extrafoveal polypoidal choroidal vasculopathy (PCV) treated with argon laser.
Design
Prospective cohort, noninterventional study.
Methods
A prospective study of Asian patients with extrafoveal PCV, confirmed on indocyanine green angiography and treated with argon laser with and without anti-vascular endothelial growth factor (VEGF) therapy. Patients were followed-up over 12 months with visual, angiographic, and structural outcomes recorded.
Results
Of the 93 eyes with PCV at baseline, 33 eyes (35.5%) in 31 patients had extrafoveal involvement and were treated with argon laser. Foveal involvement with fluid or blood at baseline was apparent in 23 eyes (69.7%), despite the extrafoveal location of 1 or more polyps. Of these 33 eyes, 12 (36.4%) also received anti-VEGF injections (median, 2.5 injections) over the 12-month period. Two eyes received photodynamic therapy rescue during subsequent follow-up and were excluded for visual outcome analysis. In the remaining 31 eyes, mean visual acuity improved from 0.57 logarithm of the minimal angle of resolution (logMAR) units (range, 0.00 to 2.0 logMAR; standard deviation, 0.51 logMAR) at baseline to 0.39 logMAR (range, 0.00 to 2.0 logMAR; standard deviation, 0.43 logMAR) at month 12 ( P = .01), with a mean gain in visual acuity of 9.0 letters at month 12. Stable or improved vision (defined as losing 5 letters or fewer) was achieved in 28 eyes (90.3%). Use of anti-VEGF was associated with significantly thicker central subfield at baseline (347.6 vs 258.1 μm; P = .02) and resulted in similar vision and OCT results at month 3 and 12 compared with eyes that did not receive anti-VEGF.
Conclusions
Argon laser treatment with selected use of anti-VEGF therapy achieves stable or improved visual outcome in most eyes with extrafoveal PCV, including eyes with fluid or blood affecting the fovea at presentation.
Age-related macular degeneration (AMD) is the leading cause of blindness in people older than 50 years in many parts of the world. In the past few years, anti-vascular endothelial growth factor (VEGF) agents, including ranibizumab and bevacizumab, have become the mainstay of treatment for neovascular AMD.
Polypoidal choroidal vasculopathy (PCV), a subtype or variant of neovascular AMD, is more common in Asians. Although it is widely believed that PCV may respond differently to treatment strategies compared with typical neovascular AMD, there is currently a lack of similar high level evidence regarding the optimal treatment in PCV. Indeed, the role of VEGF in PCV remains debated, and clinical evidence of the efficacy of anti-VEGF therapy is limited mainly to noncomparative, interventional case series. Anti-VEGF monotherapy has been reported to reduce macular edema and to improve vision, but the underlying pathologic issues are not resolved (polyp lesions are not closed), and a randomized trial showed that anti-VEGF monotherapy was less effective than photodynamic therapy (PDT) in polyp closure.
In PCV, although fluid and blood may involve the fovea, a substantial proportion of polyps is extrafoveal. For extrafoveal PCV, focal argon laser therapy of polyps has been a therapeutic option. However, there is uncertainty about the effectiveness of argon laser therapy, and several small retrospective cases studies have shown variable results. Importantly, whether the adjunctive anti-VEGF therapy improves the results of argon laser therapy is also unclear, with no previous studies reporting argon laser treatment with and without adjunctive anti-VEGF therapy. In addition, successful treatment with argon laser, as opposed to anti-VEGF therapy, often is based on careful analysis of indocyanine green angiography (ICGA), such as location and extent of polyps and associated branching vascular network. There are few studies that have examined ICGA features and their relationship to visual outcomes.
Therefore, we conducted a prospective study to determine whether argon laser therapy, with and without selective use of adjunctive anti-VEGF therapy, is effective for the management of extrafoveal PCV, concentrating on clinical and angiographic features of these cases and 12-month treatment outcomes. Our hypothesis was that argon laser therapy with the use of adjunctive anti-VEGF therapy in selected cases can achieve stable or improved visual outcome in most eyes with extrafoveal PCV.
Methods
Study Design and Population
This was a cohort study approved by the SingHealth Centralised Institutional Review Board. Patients provided informed consent to participate in this research. Specifically, the Asian AMD Phenotyping Study is a prospective, observational clinical study of treatment-naïve Asian patients with exudative maculopathy secondary to choroidal neovascularization from AMD or PCV in Singapore. Detailed methodology has been described elsewhere. The study aims to recruit consecutive patients with new-onset AMD or PCV who will provide a comprehensive set of data to phenotype the clinical condition.
Participants received treatment according to standard of care, and treatment was not altered by entering into the study. Patients were followed up prospectively at month 1, month 3, and month 12. All patients underwent a complete standardized ophthalmic examination at baseline and follow-up. This included best-corrected visual acuity (measured on Snellen charts and converted to logarithm of the minimal angle of resolution [logMAR] units for analysis), dilated fundus examination, fluorescein angiography, ICGA, fundus autofluorescence, color fundus photography, and optical coherence tomography (OCT) according to a standardized protocol at baseline.
Fundus photography was performed using a digital mydriatic retinal camera (TRC-50X/IMAGEnet 2000; Topcon, Tokyo, Japan). Autofluorescence images were acquired using the same fundus camera with the Spaide filter, which has a 580-nm exciter wavelength and a 695-nm barrier wavelength (Topcon, Tokyo, Japan). OCT was performed with the Cirrus OCT (Carl Zeiss Meditec, Dublin, California, USA) using the 512 × 128-volume cube setting.
Best-corrected visual acuity (tested with Snellen chart and converted to logMAR units) and repeat fundus photography and OCT were performed at each follow-up visit, and repeat fluorescein angiography and ICGA were performed as deemed appropriate by the treating retinal specialist.
Angiographic Grading
PCV was diagnosed by ICGA. Primary grading of ICGA and treatment decisions were made by the treating clinician. In addition, secondary ICGA grading to confirm the presence of polyps and to record specific angiographic features was performed in a masked manner by a single retinal specialist (C.M.G.C.). The diagnosis of definitive PCV lesions was based on the Japanese Study Group guidelines. Additional angiographic features regarding the location and arrangement of the polyp(s) and presence or absence and characteristics of any branching vascular network were graded according to a standardized protocol.
Argon Laser Treatment
Focal argon laser treatment was performed by retinal specialists. Generally, treatment was performed with an argon green laser 532 μm with a focal contact lens using the following parameters: 100- to 200-μm spot size and 0.15- to 0.3-ms’ duration to achieve a greyish-white burn to the active polypoidal lesions identified on ICGA. Laser could be repeated at more than 1 session if closure was believed to be incomplete at follow-up. Laser was not performed on the associated branching network.
Anti-Vascular Endothelial Growth Factor Therapy
The need for adjunctive anti-VEGF therapy was determined by the treating clinician. Although there were no strict criteria, the common indication for adding anti-VEGF therapy to argon laser was the presence of significant subretinal or sub–retinal pigment epithelium hemorrhage or fluid, or the persistence of fluid after laser therapy.
Statistical Analyses
The primary end point was change in visual acuity at month 12 from baseline. Secondary outcome measures were central subfield thickness change at month 3 and month 12 compared with baseline and the proportion of eyes with improved, stable, and worsened vision at month 12. Comparison of proportions between treatment groups was performed by the Fisher exact test or Wilcoxon Mann–Whitney U test as appropriate.
Results
Patient Characteristics
During the study period (March 2010 through March 2012), 158 patients were recruited to the Asian AMD Phenotyping study. Of these, PCV was diagnosed in 93 eyes. Within this group, argon laser was used as the primary treatment (either monotherapy or combined with anti-VEGF therapy) in 34 eyes (32 patients) in which polyps were located extrafoveally on ICGA; 1 patient defaulted after treatment (1 visit) and was excluded, leaving 33 eyes (31 patients) for subsequent analysis. Thus, the proportion of laser treatment for PCV cases was 35.5%. For the remaining patients, 33 eyes (35.5%) received PDT (8 in combination with anti-VEGF), 9 eyes (9.7%) received anti-VEGF monotherapy, and 18 eyes (19.4%) did not receive treatment (either declined by the patient or deemed unlikely to benefit significantly).
The demographic and ocular characteristics of participants are included in Table 1 . The median age at presentation was 69.78 years (range, 52.79 to 85.70 years; standard deviation [SD], 7.79 years). Twenty patients (60.6%) were male. The mean presenting visual acuity was 0.56 logMAR (range, 0.02 to 2.0 logMAR; SD, 0.50 logMAR). The mean baseline central subfield thickness was 308.4 μm (range, 185 to 758 μm; SD, 106.4 μm).
| Anti-VEGF Adjunctive Therapy | P Value a | ||
|---|---|---|---|
| No (n = 21) | Yes (n = 12) | ||
| Age (y) | 70.63 (7.43) | 68.28 (8.50) | .30 |
| Male gender, n(%) | 11 (52.4%) | 9 (75%) | .28 |
| Body mass index (kg/m 2 ) | 23.69 (3.05) | 23.00 (2.33) | .62 |
| Systolic blood pressure (mm Hg) | 150.29 (19.53) | 146.62 (17.46) | .64 |
| Diastolic blood pressure (mm Hg) | 77.57 (10.07) | 77.74 (9.42) | .88 |
| Hypertension, n(%) | 18 (85.7%) | 8 (66.7%) | .38 |
| Diabetes, n(%) | 11 (57.9%) | 1 (12.5%) | .04 |
| Current smoking, n(%) | 3 (15.8%) | 1 (12.5%) | >.99 |
| Baseline visual acuity (logMAR) | 0.57 (0.56) | 0.55 (0.40) | .74 |
| Baseline central subfield thickness (μm) | 258.1 (43.5) | 347.6 (145.3) | .02 |
| Presence of network (%) | 11 (52.4%) | 7 (58.3%) | >.99 |
| Fovea involvement (%) | 13 (61.9%) | 10 (83.3%) | .2659 |
a Wilcoxon Mann–Whitney U test or Fisher exact test as appropriate.
Polypoidal Choroidal Vasculopathy Characteristics
For this analysis, all polypoidal lesion(s) were extrafoveal by inclusion criteria. The polypoidal lesion(s) were 500 to 1000 μm from the center of the fovea in 7 eyes (21.2%) and more than 1000 μm from the center of the fovea in 26 eyes (78.8%; 2 were outside the arcades). Most eyes (23 eyes; 69.7%) had blood (7 eyes), subretinal fluid (15 eyes; 45.5%), pigment epithelial detachment (10 eyes; 30.3%), or a combination thereof involving the foveal center. The fovea was not involved in 10 eyes (30.3%).
The configuration of polypoidal lesions was classified as follows: solitary, 14 eyes (42.4%); string, 9 eyes (27.3%); solitary, 5 eyes (15.15%); and cluster, 5 eyes (15.15%). An associated network visible on ICGA was seen in 19 eyes (57.6%), 16 of which were extending under the fovea.
Additional Treatment
All eyes received thermal laser to the polypoidal lesion(s). In addition, 12 eyes (36.4%) received anti-VEGF injections (10 eyes with bevacizumab, 2 eyes with ranibizumab; Figures 1 and 2 ). In these 12 eyes, the total number of injections ranged from 1 to 7 (median, 2.5). In 2 of these 12 eyes, PDT subsequently was added as rescue treatment during over the 12-month follow-up.
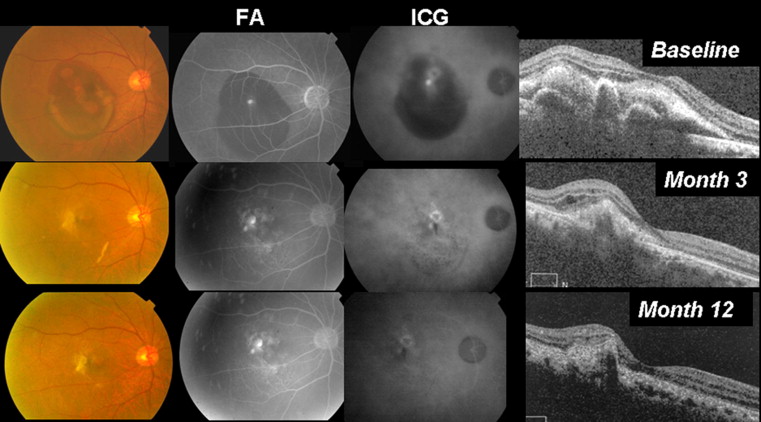
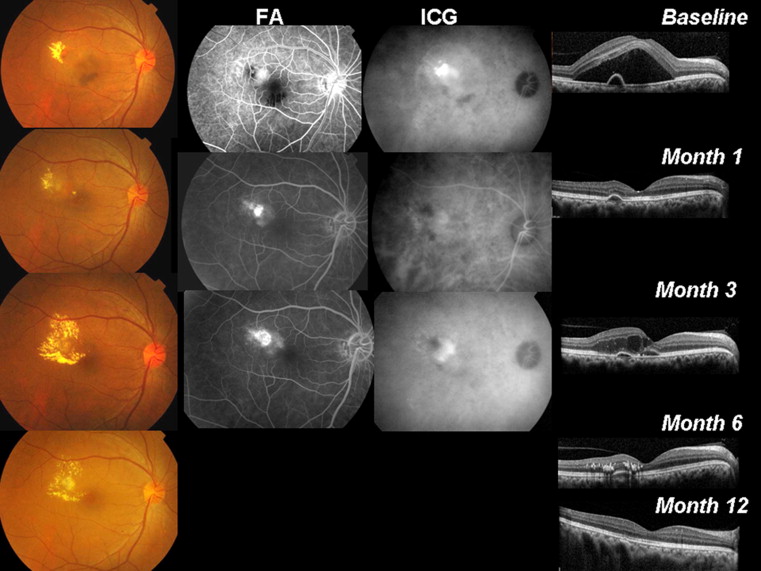
Visual Outcome
We excluded the 2 eyes that received PDT in visual outcome analysis. The mean visual acuity improved from 0.57 logMAR (range, 0.00 to 2.00 logMAR; SD, 0.51 logMAR) to 0.49 logMAR (range, 0.00 to 2.0 logMAR; SD, 0.50 logMAR) at month 3 ( P = .21) and to 0.39 logMAR (range, 0.00 to 2.0 logMAR; SD, 0.43 logMAR) at month 12 ( P = .01; Figure 3 ). The mean gain in visual acuity was 4.0 letters at month 3 and 9.0 letters at month 12 (6.5 letters if 2 eyes that underwent cataract removal during the follow-up period were excluded). Fifteen eyes (48.4%) had improved by more than 5 letters and 13 eyes (41.9%) had stable vision (change in vision, fewer than 5 letters). Three eyes (9.6%) had visual loss of more than 5 letters, of which 1 eye (3.2%) had visual loss of more than 15 letters.
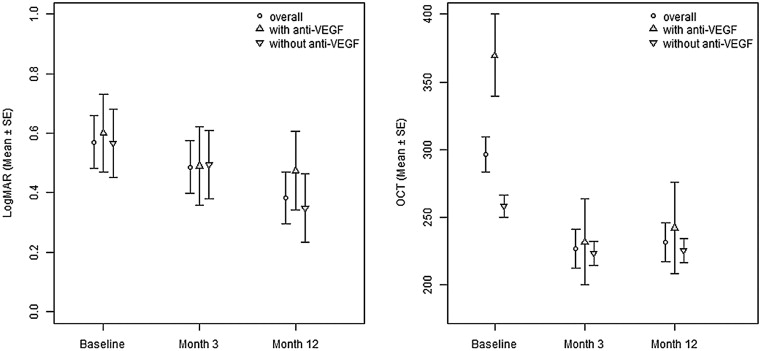
The mean central subfield thickness was reduced to 222.8 μm (range, 142 to 326 μm; SD, 31.7 μm; P = .001) at month 3 and was maintained at 229.8 μm (range, 167 to 296 μm; SD, 29.3 μm; P = .002) at month 12.
Presence of fluid or blood at the fovea at presentation was associated strongly with the presence of a branching vascular network on ICGA (94.4% vs 40.0%; P = .001). Among predictive factors analyzed for visual outcome, eyes with worse baseline vision were more likely to remain stable or to improve (0.69 vs 0.2 logMAR; P = .01; Table 2 ). Anti-VEGF treatment also was more likely to be used in eyes with any visual loss at month 12 (66.7% vs 21.7%; P = .056).
| VA Worsened (n = 6) | VA Stable/Improved (n = 25) | P Value a | |
|---|---|---|---|
| Baseline VA | 0.2 (0.36) | 0.69 (0.52) | .009 |
| Fovea involved | |||
| No | 4 (66.7%) | 6 (26.1%) | .143 |
| Yes | 2 (33.3%) | 17 (73.9%) | |
| Subfoveal blood | |||
| No | 6 (100%) | 18 (78.3%) | .553 |
| Yes | 0 (0%) | 5 (21.7%) | |
| PCV location (μm) | |||
| 500 to 1000 | 0 (0%) | 5 (21.7%) | .553 |
| > 1000 | 6 (100%) | 18 (78.3%) | |
| Network | |||
| No | 4 (66.7%) | 9 (39.1%) | .364 |
| Yes | 2 (33.3%) | 14 (60.9%) | |
| Anti-VEGF | |||
| No | 2 (33.3%) | 18 (78.3%) | .056 |
| Yes | 4 (66.7%) | 5 (21.7%) | |
The characteristics of eyes treated with argon laser alone were compared with those treated with laser combined with anti-VEGF ( Table 3 ). There was no significant difference in vision between the 2 groups at any time point. In eyes with foveal involvement, anti-VEGF therapy use was associated significantly with greater central subfield thickness at baseline. However, by month 3, central subfield thickness was comparable in both groups and remained so at month 12.
| Anti-VEGF Adjunctive Therapy | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| All (n = 31) | Fovea Involved (n = 21) | Fovea Not Involved (n = 10) | |||||||
| No (n = 21) | Yes (n = 10) | P Value a | No (n = 13) | Yes (n = 8) | P Value a | No (n = 8) | Yes (n = 2) | P Value a | |
| VA (logMAR) | |||||||||
| Baseline | 0.57 (0.56) | 0.58 (0.42) | .67 | 0.63 (0.51) | 0.73 (0.33) | .42 | 0.46 (0.65) | 0 (0) | .10 |
| Month 3 | 0.50 (0.57) | 0.47 (0.33) | .55 | 0.54 (0.55) | 0.48 (0.35) | .86 | 0.43 (0.64) | 0.44 (0.37) | .43 |
| Change from baseline to month 3 | 0.07 (0.02) | 0.11 (0.41) | .54 | 0.09 (0.23) | 0.25 (0.29) | .18 | 0.04 (0.13) | −0.44 (0.37) | .03 |
| Month 12 | 0.35 (0.44) | 0.46 (0.41) | .30 | 0.34 (0.25) | 0.54 (0.43) | .324 | 0.36 (0.66) | 0.14 (0.06) | .78 |
| Change from baseline to month 12 | 0.22 (0.40) | 0.13 (0.34) | .47 | 0.29 (0.46) | 0.19 (0.35) | .74 | 0.10 (0.24) | −0.14 (0.06) | .11 |
| Central subfield thickness on OCT (μm) | |||||||||
| Baseline | 258.1 (43.5) | 369.7 (149.6) | < .01 | 273.5 (42.3) | 395.3 (157.6) | .01 | 224.8 (24.4) | 267.5 (36.1) | .18 |
| Month 3 | 223.9 (26.9) | 231.6 (39.7) | .93 | 218.6 (22.5) | 230.9 (42.4) | .82 | 234.6 (34.5) | 237 (30.9) | .77 |
| Month 12 | 225.9 (29.2) | 241.9 (28.5) | .30 | 222.6 (30.9) | 245.7 (31.7) | .23 | 232.4 (27.3) | 230.5 (17.7) | .85 |
| Recurrence within first 12 months | |||||||||
| % | 2 (9.5%) | 3 (30.0%) | .30 | 1 (7.7%) | 3 (37.5%) | .25 | 1 (12.5%) | 0 (0%) | >.99 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


