Purpose
To evaluate the therapeutic effects of topical tacrolimus ointment on refractory inflammatory ocular surface diseases.
Design
Retrospective interventional consecutive case series.
Methods
In Severance Hospital, Seoul, South Korea, 0.02% tacrolimus ointment was topically applied 1 to 3 times per day, depending on disease severity, for up to 31 months in eyes of 12 consecutive patients with refractory inflammatory ocular surface diseases who had previously been treated with steroid therapy. Seven patients had chronic cicatrizing conjunctivitis (6 cases caused by Stevens-Johnson syndrome and 1 attributable to ocular cicatricial pemphigoid); 4 had scleritis (3 necrotizing scleritis, 1 recurrent nodular scleritis); and 1 patient had Mooren ulcer with corneal perforation. The therapeutic outcomes after tacrolimus treatment were evaluated according to the following criteria: change in clinical findings (eg, decrease of hyperemia, ocular pain, epithelial defect, and pseudomembrane), intraocular pressure (IOP), and need for steroid therapy.
Results
In all 3 groups, tacrolimus showed an immunosuppressive effect, especially on scleritis and Mooren ulcer. These effects included suppression of corneoscleral melting and reduction of hyperemia. In chronic cicatrizing conjunctivitis, simultaneous topical tacrolimus while tapering steroid therapy suppressed inflammatory relapse. The elevated IOP in steroid responders recovered to normal range after successful tapering of steroid. No adverse side effects were noted after 1.5 to 31 months of continuous tacrolimus treatment.
Conclusion
The use of topical tacrolimus ointment is effective in controlling refractory inflammatory ocular surface disease, and can reduce the need for steroid use while reducing inflammation recurrence.
Chronic inflammatory ocular surface disorders with immunologic causes, such as refractory allergic conjunctivitis, Stevens-Johnson syndrome (SJS), ocular cicatricial pemphigoid disease (OCP), scleritis, and Mooren ulcer, can cause considerable impairment of quality of life. Their treatment is mainly driven by systemic or topical glucocorticosteroids, which potentially have undesirable side effects including steroid-induced glaucoma, cataract formation, superinfection, and delayed corneal healing. These problems often limit long-term steroid use for treating inflammatory ocular surface diseases. Therefore, it is necessary to develop alternative nonsteroidal therapeutic modalities that have more potent anti-inflammatory actions with fewer side effects than steroids.
Tacrolimus (previously known as FK506) is a macrolide immunosuppressant and immunomodulatory agent isolated from Streptomyces tsukubaensis that has been approved by the US Food and Drug Administration for treating moderate to severe atopic dermatitis. Tacrolimus acts by suppressing T cell–mediated immune response, similar to cyclosporin, but is 10 to 100 times more potent and shows better cutaneous penetration because of its much lower molecular weight. Recently, it has been successfully used as a systemic immunomodulator to prevent allograft rejection in liver, kidney, and heart transplant patients. Topical tacrolimus ointment (Protopic 0.03%; Fujisawa Healthcare Inc, Munich, Germany) is used as a highly effective nonsteroidal treatment for managing atopic dermatitis in the United States and Japan. With dermatitis, tacrolimus shows higher efficacy with fewer adverse effects than do corticosteroid ointments. In the field of ophthalmology, a number of studies have reported the effect of tacrolimus ointment for treating immunologic refractory ocular diseases, but most of them have studied allergic conjunctivitis, such as atopic or vernal allergic conjunctivitis.
Therefore, we hypothesized that topical 0.02% tacrolimus ointment, originally approved for dermatologic purposes, would be effective in treating diverse inflammatory ocular surface diseases that are refractory to conventional treatment. We evaluated tacrolimus effectiveness in 12 patients suffering from various ocular inflammatory diseases, including chronic cicatrizing conjunctivitis, scleritis, and Mooren ulcer with corneal perforation, and we have demonstrated its clinical utility in treating these disorders.
Patients and Methods
This was a retrospective review of 12 consecutive patients with various inflammatory ocular surface diseases who were treated with topical 0.02% tacrolimus ointment at Severance Hospital, South Korea, between October 1, 2009 and June 30, 2012. Ointment was applied in the lower conjunctival sac 1 to 3 times per day, depending on the severity of each disease. Approval to conduct this retrospective study was obtained from the Institutional Review Board of the Yonsei University College of Medicine, Seoul, South Korea. Clinical research in this study followed the tenets of the Declaration of Helsinki.
The commercial tacrolimus ointment (Protopic 0.03%) that has been used to treat dermatologic disorders is extremely viscous and is not appropriate for direct application into the conjunctival sac. Because this formulation restricts the long-term use of dermatologic tacrolimus ointment for eye diseases, we created a preparation of 0.02% tacrolimus ointment by diluting the 0.03% tacrolimus ointment 2:1 by volume with a less-viscous lubricating ophthalmic ointment (Duratears; Alcon Laboratories Inc, Fort Worth, Texas, USA), which is composed of 30 mg anhydrous liquid lanolin per gram of mineral oil base. Topical Duratears forms a layer that slows tear evaporation and increases the moisture content by increasing water-holding capacity.
Our 0.02% tacrolimus ointment was used as an additional agent on inflamed eyes that had previously received steroid treatment to inhibit progression of acute or chronic inflammation, and to achieve clinical improvement of refractory ocular surface diseases. Refractory, in this context, means that the case showed aggressive, deteriorating inflammation progression despite receiving an intensive steroid-based immunosuppression therapy for more than 2 weeks, or inflammation relapse after corticosteroid tapering after remission.
These data included patient age, sex, diagnosis, surgical history, disease duration, previous medication, clinical photography, and duration of drug use. We also collected data on clinical features including objective and subjective findings before and after tacrolimus administration or the tapering or discontinuation of steroid eye drops, the duration of tacrolimus maintenance, and the follow-up period, and any adverse side effects of tacrolimus.
The clinical outcomes of tacrolimus treatment were assessed by the changes of representative parameters at the time of tapering the tacrolimus using the modified scoring system, which was described by Miyazaki and associates. The inflammation of chronic cicatrizing conjunctivitis was scored regarding hyperemia degree, corneal or conjunctival fluorescein staining, pseudomembrane, progression of corneal conjunctivalization or symblepharon, the presence of persistent epithelial defect, and progression of corneal neovascularization. The severity of symptoms and signs in each patient was scored using a scale of 0 to 4, where 0 indicates no inflammation and scores of 1, 2, 3, and 4 represent mild, moderate, severe, and very severe degrees of inflammation, respectively. Anterior segment inflammation such as scleritis and Mooren ulcer was scored regarding severity of ocular pain, hyperemic degree, the presence of scleral or corneal melting, and anterior chamber inflammation (AC reaction) using a scale of 0 to 4, where 0, 1, and 2 indicate no, mild, and moderate hyperemia, respectively; 3 indicates severe but localized hyperemia without AC reaction; and a score of 4 describes extensive, painful hyperemia with AC reaction and/or scleral or corneal melting.
The specific steroid treatments necessary for initial treatment also were scored on a scale of 0 to 4. A score of 0 indicates no steroids used; 1 indicates topical fluorometholone 0.1% (Ocumetholone; Samil Pharmaceutical Co, Ltd, Seoul, Korea); 2 indicates topical prednisone acetate 0.12% (Optilon; Chongkundang Pharmaceutical Co, Seoul, Korea); 3 indicates topical prednisone acetate 1% (Predforte; Allergan, Inc, Irvine, California, USA) or rimexolone 1% (Vexol; Alcon Laboratories Inc); and 4 indicates systemic steroids with/without topical prednisone acetate 1% use, or intensive, frequent use of topical prednisone more than 6 times per day.
The statistical analyses of therapeutic effects before and after tacrolimus treatment were performed using the SPSS statistical software package (version 20.0; SPSS Inc, Chicago, Illinois, USA) using Wilcoxon signed rank test. P values less than .05 were considered to be statistically significant.
Results
The study group consisted of 18 eyes of 12 patients; 3 patients (25.0%) were male and 9 (75.0%) were female. Patients had a median age of 54.0 years (range 10-70) at presentation. The median ocular disease duration was 5.5 months (range 0.5-180 months). The study group was divided into 3 kinds of immunologic inflammatory ocular surface diseases: (1) chronic cicatrizing conjunctivitis caused by SJS and OCP; (2) necrotizing scleritis and nodular scleritis; and (3) Mooren ulcer. The use of topical 0.02% tacrolimus ointment for these patients was initiated for the reasons shown in Table 1 . Of these 12 patients, 4 eyes of 3 patients had topical steroid–induced intraocular pressure (IOP) elevation (ie, steroid responders). None of the patients had a history of herpetic viral keratitis.
| Reason for Treatment | Number of Patients |
|---|---|
| Chronic cicatrizing conjunctivitis (6 SJS, 1 OCP) | 7 |
| Chronic intractable disease course: persistent epithelial defect, progression of conjunctivalization despite prolonged steroid use | 5 |
| Rapid and aggressive progression of active severe inflammation | 2 |
| Increased IOP from topical steroids | 2 |
| Scleritis (3 necrotizing scleritis, 1 nodular scleritis) | 4 |
| Rapid and aggressive progression of active severe inflammation | 3 |
| Disease relapse | 1 |
| Increased IOP from topical steroids | 1 |
| Intractable Mooren ulcer | 1 |
| Attempted suppression of corneal melting despite prolonged steroid use | 1 |
Characteristics of the Study Population
Table 2 summarizes the characteristics of the 12 patients who were treated with 0.02% topical tacrolimus ointment. All patients had a history of moderate to severe disease requiring frequent (≥6 times/day) or continuous use (more than 3 months) of topical steroid therapy to control active or recurrent chronic inflammation. In severe cases of necrotizing scleritis and Mooren ulcer, oral or intravenous administration of steroid was used, and Patients 10 and 12 had taken immunosuppressive agents such as cyclophosphamide and methotrexate in addition to steroid therapy. Topical tacrolimus was applied as an adjunct immunosuppressive therapy in addition to previous steroid treatments.
| Patient | Age (y) | Sex | Diagnosis Ocular Surgical History | Disease Duration (mo) | Steroid Tx Duration (mo) | IOP (mm Hg) (OD/OS) | Frequency of Tacrolimus |
|---|---|---|---|---|---|---|---|
| Chronic cicatrizing conjunctivitis | |||||||
| 1 | 67 | F | SJS S/p limbal transplantation | 180 | 7/- | 26/- | 1 |
| 2 | 14 | M | SJS | 72 | 6 | 14/12 | 1 |
| 3 | 64 | F | SJS | 16 | 15 | 8/9 | 1 |
| 4 | 32 | F | SJS | 6 | 6 | 25/26 | 1 |
| 5 | 70 | F | SJS | 2 | 2 | 15/15 | 1 |
| 6 | 48 | F | SJS | 5 | 5 | 16/14 | 1 |
| 7 | 10 | F | OCP S/p PKP (OS) S/p limbal transplantation | 120 | 72 | 20/21 | 1 |
| Scleritis | |||||||
| 8 | 54 | F | Necrotizing scleritis S/p pterygium removal | 1 | 0.5 | 17 | 2 |
| 9 | 54 | F | Necrotizing scleritis S/p pterygium removal | 0.5 | 0.5 | 19 | 2 |
| 10 | 70 | F | Necrotizing scleritis S/p pterygium removal Rheumatoid arthritis | 1 | 0.5 | 20 | 2 |
| 11 | 51 | M | Nodular scleritis | 12 | 3 | 23 | 1 |
| Mooren ulcer | |||||||
| 12 | 65 | M | Mooren ulcer with corneal perforation S/p AMT | 6 | 6 | 8 | 3 |
Tacrolimus Efficacy
Table 3 summarizes all of the therapeutic assessment parameters, including changes in inflammatory score, steroid score, and IOP. After tacrolimus had begun, all patients except Patient 10 reported an improvement in symptoms and signs within 1 to 4 weeks. Then systemic or topical steroids were tapered or discontinued gradually, with continued use of tacrolimus. The median follow-up period was 15.0 months (range 1.5-31.0 months) after initiation of tacrolimus. The remission period refers to how long each inflammatory score was maintained at 0 after inflammation suppression during follow-up, regardless of tacrolimus use.
| Patient | Diagnosis | IOP (mm Hg) | Inflammation Score | Steroid Score | Time to Improvement (wk) | Duration of Tacrolimus Maintenance (mo) | Follow-up (mo) | Remission (mo) | Tacrolimus Effect(s) | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Before | After | Before | After | Before | After | |||||||
| Chronic cicatrizing conjunctivitis | ||||||||||||
| 1 | SJS | 26/- | 10/- | 3/- | 0/- | 3/- | 0/- | 2 | 31 | 31 | 25 | Steroid tapering (after 4 mo) IOP stabilization No inflammatory flare-up Elongation of follow-up period |
| 2 | SJS | 14/12 | 13/12 | 2/2 | 0/0 | 2/2 | 1/1 | 2 | 27 | 27 | 3 | Stabilization of inflammation Steroid tapering (after 8 mo) |
| 3 | SJS | 8/9 | 11/14 | 3/3 | 1/1 | 2/2 | 0/0 | 4 | 12 | 12 | 2 | Prevention of inflammation flare-up |
| 4 | SJS | 25/26 | 18/20 | 4/3 | 3/3 | 3/3 | 2/2 | 4 | 1.5 | 1.5 | – | IOP stabilization |
| 5 | SJS | 15/15 | 17/15 | 4/4 | 3/3 | 3/3 | 3/3 | 4 | 15 | 18 | – | No definite response |
| 6 | SJS | 16/14 | 12/9 | 3/3 | 2/2 | 1/1 | 1/1 | 4 | 6 | 14 | – | Slight reduction of inflammation |
| 7 | OCP | 20/21 | 15/18 | 3/3 | 2/2 | 4/4 | 3/3 | 4 | 4 | 10 | – | Slight reduction of inflammation |
| Scleritis | ||||||||||||
| 8 | Necrotizing scleritis | 17 | 10 | 4 | 0 | 4 | 0 | 1 | 12 | 16 | 13 | Prevention of relapse Cessation of steroid (after 3 mo) Suppression of inflammation |
| 9 | Necrotizing scleritis | 19 | 13 | 4 | 0 | 4 | 0 | 3 | 7 | 19 | 16 | Prevention of relapse Cessation of steroid (after 3 mo) |
| 10 | Necrotizing scleritis | 20 | 15 | 4 | 4 | 4 | 4 | – | 4 | 4 | – | No response |
| 11 | Nodular scleritis | 23 | 14 | 3 | 0 | 4 | 1 | 3 | 10 | 12 | 2 | IOP stabilization Suppression of inflammation |
| Mooren ulcer | ||||||||||||
| 12 | Mooren ulcer | 8 | 7 | 4 | 0 | 4 | 0 | 3 | 6 | 17 | 11 | Suppression of inflammation Prevention of relapse |
Tacrolimus showed a good anti-inflammatory effect in all 3 study subgroups ( Figures 1 and 2 ). Especially, there was great change in both inflammatory score and steroid score after tacrolimus treatment in the necrotizing scleritis and Mooren ulcer groups, despite lack of statistical significance attributable to the small number of cases. All patients with topical steroid–induced IOP elevation showed recovery into the normal IOP range after steroid tapering.
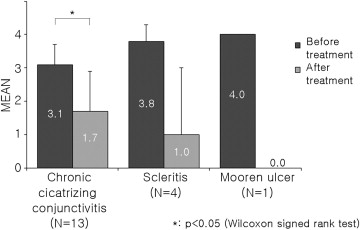
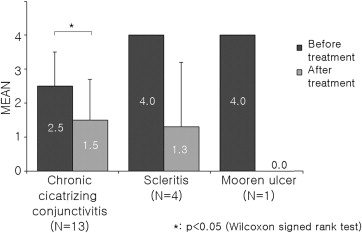
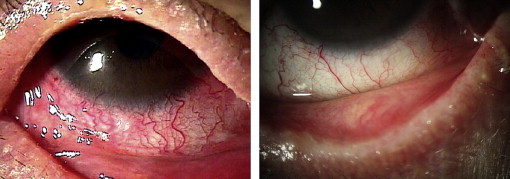
In 7 patients with chronic cicatrizing conjunctivitis, tacrolimus showed statistically significant therapeutic effects; but it seemed not as efficacious in the other 2 subgroups. However, in Patients 1, 2, and 3, additional topical tacrolimus treatment decreased corneal vascularization and recurrent corneal epithelial defect, and facilitated steroid tapering so that steroid-elevated IOP was normalized ( Figure 3 ). In Patients 4, 5, and 6, who were in an acute active inflammatory stage, tacrolimus therapy was attempted to suppress the aggravation and progression of active inflammation despite the frequent use of steroids in these patients. Tacrolimus likely helped to slightly reduce the hyperemia, corneal vascularization, epithelial defect, and symblepharon progression in these patients, although the effect on these parameters was not as marked ( Table 3 ).