Purpose
To evaluate the effect of intravitreal anti–vascular endothelial growth factor (VEGF) injections on submacular hemorrhage secondary to polypoidal choroidal vasculopathy.
Design
Retrospective, interventional case series.
Methods
Twenty-seven eyes from 27 polypoidal choroidal vasculopathy patients with submacular hemorrhage involving the fovea were included in the analyses. All patients were treated by anti-VEGF injection with an initial 3 loading injections by month, followed by an as-needed reinjection. Visual acuity, central macular thickness, submacular hemorrhage size, and the occurrence of vitreous hemorrhage were examined during a 12-month follow-up period.
Results
The mean number of injections administered over the course of 12 months was 3.59 ± 1.04. The size of submacular hemorrhages averaged 18.2 ± 13.8 mm 2 . The mean logarithm of the minimal angle of resolution (logMAR) visual acuity at baseline was 1.02 ± 0.51 (Snellen equivalent, 20/204) and improved significantly to 0.76 ± 0.48 (Snellen equivalent, 20/115) at 12 months ( P = .02). Mean central macular thickness decreased from 311.7 ± 124.5 μm at baseline to 246.8 ± 102.8 μm at 12 months ( P = .01). At 12 months, visual acuity improved by 0.3 logMAR or more in 10 eyes (37%), stabilized (change between 0 and 0.3 logMAR) in 11 eyes (40.7%), and decreased by 0.3 logMAR or more in 6 eyes (22.2%). Three eyes (11.1%) were subjected to vitrectomy to clear a vitreous hemorrhage that occurred after anti-VEGF therapy.
Conclusions
Intravitreal anti-VEGF injection monotherapy may be a valuable therapeutic option in treating eyes with submacular hemorrhage associated with polypoidal choroidal vasculopathy.
Submacular hemorrhage secondary to choroidal neovascularization (CNV) in age-related macular degeneration (AMD) may cause sudden vision loss with a poor visual prognosis. Hemorrhages can cause retinal damage directly through various mechanisms.
Polypoidal choroidal vasculopathy (PCV) is classified as a subgroup of exudative AMD. It is characterized by a branching polypoidal-shaped vascular network with choroidal lesions that result in subretinal leakage, subretinal hemorrhage, and pigment epithelial detachment (PED). Subretinal hemorrhages and hemorrhagic PEDs occur more often in patients with PCV than in patients with typical neovascular AMD. Moreover, submacular hemorrhage also is a more common complication of PCV than of typical neovascular AMD.
A frequently used procedure to treat submacular hemorrhage is an intravitreal injection of recombinant tissue plasminogen activator (rTPA) and sulfur hexafluoride (SF 6 ) for pneumatic displacement of the hemorrhage. Vitrectomy combined with pneumatic displacement and rTPA also can be considered for massive subretinal hemorrhage. Although treatment with pneumatic displacement, rTPA, or both is effective in displacing the submacular hemorrhage, it has several limitations. Sub–retinal pigment epithelium (RPE) hemorrhage is resistant to pneumatic displacement. rTPA and SF 6 injections have limited efficacy in treating submacular hemorrhages that are older than 2 weeks and hemorrhages with diameter of more than 5 mm. In case of AMD or PCV, long-term visual outcome usually is limited by progression of the underlying CNV. Moreover, unlike typical neovascular AMD, PCV is the major risk factor for vitreous hemorrhage after rTPA and pneumatic displacement of submacular hemorrhage.
Intravitreal injections of anti–vascular endothelial growth factor (VEGF) agents recently have been considered as a treatment option for submacular hemorrhage. Several retrospective studies have reported success with intravitreal anti-VEGF agents, in combination with SF 6 pneumatic displacement, for submacular hemorrhage in patients with neovascular AMD. Encouraging results for intravitreal anti-VEGF monotherapy also have been reported for patients with submacular hemorrhage related to neovascular AMD.
Although intravitreal anti-VEGF agents are used commonly to treatment PCV, there have been no reports of anti-VEGF therapy for submacular hemorrhage in patients with PCV. The current study retrospectively evaluated the efficacy of intravitreal anti-VEGF injection monotherapy in patients with submacular hemorrhage secondary to PCV.
Methods
This study was approved by the Institutional Review Board of Kim’s Eye Hospital, Konyang University College of Medicine. Clinical research in this study followed the tenets of the Declaration of Helsinki. The medical records for 40 eyes from 39 consecutive patients who were treated for submacular hemorrhage associated with PCV were reviewed retrospectively. All patients were examined and treated between November 2008 and November 2011 at the Retina Center of Kim’s Eye Hospital at Konyang University College of Medicine.
Inclusion and Exclusion Criteria
Patients were included if they met all of the following criteria.
- 1.
Age older than 50 years.
- 2.
Acute deterioration of vision because of a hemorrhage of more than 1 disc diameter directly beneath the fovea. We did not define a maximum hemorrhage size or duration as inclusion or exclusion criteria.
- 3.
Confirmation of PCV with fluorescein angiography and indocyanine green angiography (ICGA), performed using a confocal laser scanning system (HRA-2; Heidelberg Engineering, Dossenheim, Germany). We included only patients whose ICGA results showed a branching vascular network with polypoidal-shaped choroidal vascular lesions. In case of thick submacular hemorrhage interfering with identification of underlying pathologic features, the diagnosis was based on the results of the follow-up examinations, including ICGA.
- 4.
Patients who were treatment naïve.
- 5.
Patients who were treated with an anti-VEGF agent (either bevacizumab or ranibizumab).
- 6.
A minimum follow-up period of 12 months.
Patients were treated with intravitreal anti-VEGF injections only if no other treatment option was suitable. Some patients declined therapy with rTPA or SF 6 gas injection, some were unable to maintain the face-down position for medical reasons, some hemorrhages were located primarily superior to the foveola, and some submacular hemorrhages were older than 2 weeks or were very large (>5 mm in diameter).
Exclusion criteria included the following: combination treatment with pneumatic displacement, intravitreal rTPA, or both; prior treatment with PDT; treatment combined with PDT; pathologic myopia; idiopathic CNV; other secondary CNV; other ocular diseases that could affect visual acuity; trauma during the study or in the fellow eye; aphakia; and previous vitreoretinal surgery.
Documentation and Follow-Up
All patients were followed up monthly for 12 months after the initial intravitreal anti-VEGF injection. Follow-up examinations, such as best-corrected visual acuity (BCVA) examination, fundus photography, and spectral-domain optical coherence tomography (Spectral OCT/SLO; OTI Ophthalmic Technologies, Inc, Miami, Florida, USA) were performed 3, 6, 9, and 12 months after the initial visit. For all analysis, Snellen BCVA was converted to logarithm of the minimal angle of resolution (logMAR) values, and the logMAR visual acuity for counting fingers was defined as +2.0, as established by Holladay.
Fluorescein angiography, ICGA, or both were performed at baseline, after 3 monthly loading injection treatments, and then repeated when needed, considering further treatment. ICGA was used to determine the greatest linear dimension (GLD) and the area of the subretinal hemorrhage by using the HRA-2 built-in software. The GLD included the entire PCV vascular lesion, including the polypoidal lesion, the network of branching vessels, and any type 2 CNV. Pigment epithelial detachments that had no underlying vascular components were not included in the GLD measurement.
Central macular thickness was taken as the average thickness of the 1-mm central area of the spectral-domain optical coherence tomography map. Only well-centered scans without overt algorithm failure messages were included in the analyses.
Anti–Vascular Endothelial Growth Factor Treatment
The off-label nature of the treatment and its potential risks and benefits were discussed in detail with each patient. Intravitreal anti-VEGF injections of either bevacizumab (1.25 mg/0.05 mL; Avastin; Genentech, Inc, South San Francisco, California, USA) or ranibizumab (0.5 mg/0.05 mL; Lucentis; Genentech, Inc) were administered to all patients according to the same treatment and retreatment protocols. After performing 3 monthly loading injections, retreatment for each patient was performed following a retreat as needed protocol, if any of the following were observed: visual deterioration of more than 2 lines (more than 0.2 logMAR visual acuity), evidence of persistent fluid or hemorrhage involving the macula on OCT at least 1 month after the previous injection, newly developed hemorrhage or recurrence of fluid involving the macula, and evidence of an active PCV lesion, as found on fluorescein angiography, ICGA, or OCT. In cases where vitreous hemorrhage occurred, anti-VEGF therapy was stopped and a pars plana vitrectomy was performed.
Statistical Analysis
SPSS software (version 13.0, SPSS, Inc, Chicago, Illinois, USA) was used for all statistical analyses. Frequencies were compared between treatment groups using χ 2 tests. Comparative statistics were determined using unpaired 2-sided t tests. For the nonparametric data, the Mann–Whitney U and Kruskal–Wallis tests were used.
Results
Of the 39 patient records examined, 27 eyes (27 patients) were included in the analyses and 13 eyes (12 patients) were excluded. Ten eyes were treated by combination treatment with pneumatic displacement and intravitreal rTPA. Three eyes were treated by a combination of intravitreal anti-VEGF injection, rTPA and PDT. Table 1 summarizes patient characteristics and treatment results. A total of 27 eyes from 27 consecutive Korean patients (9 women, 18 men) were included in the analyses. Their mean age was 65.4 ± 7.8 years. The mean duration of symptom was 9.8 ± 7.5 days. The mean logMAR visual acuity was 1.02 ± 0.51 (Snellen equivalent, 20/204) at baseline (median, 20/200; range, counting fingers to 20/30). The mean size of submacular hemorrhages was 18.2 ± 13.8 mm 2 (median, 16.21 mm 2 ; range, 3.02 to 60.92 mm 2 ). The average number of injections administered was 3.59 ± 1.04 over 12 months. The location of polypoidal lesions was subfoveal or juxtafoveal in 22 of 27 eyes (81.5%) and extrafoveal in 5 of 27 eyes (18.5%). The GLD of the entire PCV lesion averaged 2314 ± 949 μm (median, 2013 μm; range, 1020 to 4640 μm). Ranibizumab was administrated in 63% (58/92) of patients and bevacizumab was given in the remaining 37% (34/92).
| Patient No. | Age (y) | Sex | Duration of Symptoms (d) | Submacular Hemorrhage Size (mm 2 ) | PCV Location | GLD on ICGA (μm) | Hemorrhage Location of Fovea | Baseline VA (Snellen) | VA at 12 Months | Baseline CMT (μm) | CMT at 12 Months (μm) | No. of Injections | Complication and Treatment |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 63 | M | 10 | 7.87 | Subfoveal | 1994 | Subretinal | 20/30 | 20/50 | 273 | 212 | 3 | None |
| 2 | 71 | M | 2 | 7.11 | Subfoveal | 2240 | Sub-RPE | 10/200 | 5/200 | 451 | 302 | 3 | None |
| 3 | 69 | M | 28 | 35.56 | Subfoveal | 1621 | Subretinal | 20/30 | 20/30 | 240 | 221 | 5 | None |
| 4 | 61 | M | 3 | 44.45 | Juxtafoveal | 1950 | Subretinal | 20/100 | 20/70 | 258 | 189 | 6 | None |
| 5 | 60 | F | 2 | 2.54 | Juxtafoveal | 1221 | Subretinal | 20/40 | 20/30 | 278 | 195 | 3 | None |
| 6 | 55 | F | 42 | 10.52 | Juxtafoveal | 1950 | Sub-RPE | 20/100 | 20/25 | 336 | 189 | 4 | None |
| 7 | 77 | M | 3 | 3.11 | Extrafoveal | 1254 | Subretinal, sub-RPE | CF | 20/50 | 418 | 224 | 3 | None |
| 8 | 60 | M | 15 | 11.43 | Extrafoveal | 1452 | Subretinal | 10/200 | 20/100 | 467 | 195 | 4 | None |
| 9 | 55 | F | 1 | 49.53 | Subfoveal | 3442 | Subretinal | CF | 20/200 | 346 | 304 | 5 | None |
| 10 | 76 | F | 30 | 16.26 | Subfoveal | 2993 | Sub-RPE | 20/100 | 20/100 | 199 | 184 | 3 | None |
| 11 | 79 | M | 5 | 13.21 | Juxtafoveal | 1780 | Subretinal | 10/200 | 10/200 | 302 | 327 | 4 | None |
| 12 | 71 | M | 1 | 50.81 | Juxtafoveal | 2441 | Subretinal, sub-RPE | CF | CF | 514 | 320 | 3 | None |
| 13 | 59 | F | 7 | 55.21 | Juxtafoveal | 4127 | Subretinal | 10/200 | CF | 449 | 342 | 2 | VH & vitrectomy |
| 14 | 57 | M | 6 | 3.30 | Subfoveal | 1020 | Subretinal | 20/40 | 20/40 | 232 | 222 | 4 | None |
| 15 | 51 | F | 3 | 3.81 | Juxtafoveal | 1867 | Subretinal | 20/50 | 20/25 | 254 | 188 | 4 | None |
| 16 | 74 | M | 5 | 60.92 | Juxtafoveal | 2246 | Subretinal, sub-RPE | CF | 20/200 | 255 | 249 | 1 | VH & vitrectomy |
| 17 | 67 | F | 7 | 46.96 | Juxtafoveal | 4640 | Subretinal | 20/40 | 5/200 | 221 | 318 | 2 | VH & vitrectomy |
| 18 | 68 | M | 2 | 4.01 | Extrafoveal | 3712 | Subretinal | 20/200 | 5/200 | 181 | 225 | 3 | None |
| 19 | 76 | M | 10 | 11.43 | Subfoveal | 2812 | Sub-RPE | CF | 20/200 | 224 | 233 | 4 | None |
| 20 | 70 | F | 15 | 44.45 | Juxtafoveal | 1990 | Subretinal | 5/200 | 5/200 | 278 | 303 | 3 | None |
| 21 | 58 | F | 3 | 3.02 | Juxtafoveal | 2750 | Subretinal | 20/70 | 20/40 | 210 | 194 | 4 | None |
| 22 | 63 | M | 1 | 19.56 | Extrafoveal | 2013 | Subretinal | 20/50 | 20/200 | 242 | 289 | 3 | None |
| 23 | 73 | M | 35 | 29.21 | Juxtafoveal | 2480 | Subretinal | 20/100 | 20/40 | 293 | 165 | 6 | None |
| 24 | 56 | M | 3 | 3.56 | Subfoveal | 1584 | Subretinal, sub-RPE | 10/200 | 20/30 | 302 | 189 | 3 | None |
| 25 | 66 | M | 15 | 41.91 | Extrafoveal | 2436 | Subretinal, sub-RPE | CF | 5/200 | 398 | 308 | 4 | None |
| 26 | 65 | M | 10 | 18.83 | Juxtafoveal | 1434 | Subretinal | 20/200 | 20/30 | 340 | 198 | 3 | None |
| 27 | 66 | M | 3 | 5.59 | Subfoveal | 2215 | Subretinal | 20/200 | 5/200 | 355 | 314 | 5 | None |
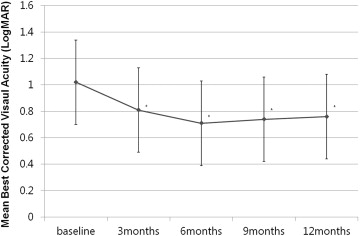
After treatment, the mean logMAR visual acuity improved significantly from 1.02 ± 0.51 (Snellen equivalent, 20/204) to 0.81 ± 0.66 (Snellen equivalent, 20/129; P = .04) at 3 months, to 0.71 ± 0.44 (Snellen equivalent, 20/102; P = .01) at 6 months, to 0.74 ± 0.36 (Snellen equivalent, 20/109; P = .01) at 9 months, and to 0.76 ± 0.48 (Snellen equivalent, 20/115; P = .02) at 12 months. Figure 1 shows the changes in BCVA (logMAR) observed over the course of follow-up. Ten (37%) of 27 eyes showed improvements in logMAR visual acuity of 0.3 or more ( Figure 2 ), 6 patients (22.2%) had a decrease in logMAR visual acuity of 0.3 or more, and 11 patients (40.8%) had a change in the logMAR visual acuity of less than 0.3 unit ( Table 2 ). There was no association between the change in BCVA and the duration of symptoms, the size of the hemorrhage, or the baseline visual acuity ( Table 3 ). Of the 6 eyes that had a decrease of more than 3 lines (in logMAR visual acuity that exceeded 0.3), a disciform scar developed in 2 eyes, a diffuse RPE atrophy occurred in 2 eyes, and a large subfoveal PED occurred in 2 eyes. These findings were thought to cause the severe decrease in visual acuity. Baseline central macular thickness was 311.7 ± 124.5 μm, which decreased significantly over time to 255.5 ± 142.1 μm at 3 months ( P = .01), to 218.5 ± 142.1 μm at 6 months ( P = .002), to 240.5 ± 152.3 μm at 9 months ( P = .01), and to 246.8 ± 102.8 μm at 12 months ( P = .01).