This article discusses the currently available techniques used for the diagnosis of IgE-mediated upper respiratory allergy. These methods are necessary to confirm the presence and the intensity of allergy in an effort to select patients for immunotherapy and to dose immunotherapy properly. Specific techniques discussed include epicutaneous and intradermal skin tests and in vitro tests designed to measure antigen-specific IgE antibody.
It generally is accepted that the pathophysiology and symptoms of allergic rhinitis are based on an IgE-mediated type I Gell and Coombs reaction. During the management of patients suspected of having allergic rhinitis, there are various points at which it is appropriate to confirm this mechanism. Doing so opens the door for treatment options not otherwise available to the patient and bolsters diagnostic confidence for the patient and the physician. In many circumstances this confirmation will set the stage for more effective, timely, and efficient pharmacologic, environmental, and immunotherapeutic management. Confirming the mechanism and the offending triggers responsible for symptoms and measuring antigen sensitivity objectively sets the stage for a more tailored approach to symptom control and improved outcomes.
Box 1 summarizes the currently acceptable tests that can confirm IgE-mediated respiratory allergen sensitivity. This article focuses on skin and in vitro testing techniques.
Skin tests
Intracutaneous (intradermal)
Intradermal single-dilution test
Intradermal dilutional test
Epicutaneous tests
Prick/puncture tests
Multiple-antigen
Single-antigen
End-organ provocation tests
Nasal provocation
Bronchial provocation
Conjunctival provocation
In vitro serologic tests to measure aeroallergen-specific IgE
Radioisotope labeling
Enzyme-linked labeling
Fluorescence labeling
Skin testing
Using the skin as a test organ to identify aeroallergen sensitivity was first reported in 1873 . Skin testing remains the most widely practiced method for several good reasons. Mast cells are the key effector cells in the type I reaction and reside in the subepithelial layer of nearly all epithelial organs, including the respiratory tract and the skin. Obviously, the skin is visible, accessible, and measurable. The abundance of skin affords the clinician the option of tailoring strategies by being able to challenge the patient with multiple antigen strengths, thereby improving test sensitivity and leading to fewer false-negative results. A skin test challenges the patient with the same antigen extracts that will be used in immunotherapy. In effect, the patient’s response to specific antigens is being assessed by bioassay . Thus, skin testing should assist in a safe and seamless transition to immunotherapy for the patient who is contemplating immunotherapy.
Intradermal testing
Intracutaneous (intradermal) testing by injection of appropriately diluted liquid allergen extract into the dermal layer of the skin is one of the most reliable and proven skin-testing techniques. Akin to a tuberculin test, a 4-mm intracutaneous wheal is created by injecting a small amount of antigen (usually 0.01–0.02 mL) using a one-piece syringe and a 27-gauge needle. This technique allows contact of antigen with subepithelial mast cells. If IgE-mediated sensitivity is present, and the patient has been previously sensitized to the test allergen, the allergen-specific IgE proteins coating the mast cells will become cross-linked with the antigen, causing mast cell degranulation and eventual growth/expansion of the wheal. The wheal is remeasured within 10 to 20 minutes; if the wheal has grown to 7 mm or greater, the test is positive. An accompanying erythematous reaction surrounding the positive wheal, termed the “flare reaction,” offers further confirmation.
Unfortunately there are factors besides allergy that can affect skin whealing responses, making interpretation sometimes challenging. Medications that inhibit the whealing response include antihistamines, tricyclic antidepressants, and systemic beta agonists. Also, pediatric and geriatric patients can be less reactive. Factors that enhance the whealing response include dermatopathologies (dermatographism, eczema, urticaria), and beta antagonists (beta blockers). Because almost all antigen extracts contain glycerin, sensitivity to glycerin also can enhance the whealing response. Enhancement also can occur if the patient recently ingested a food (eg, cantaloupe) that contains surface proteins that are similar enough to the antigenic determinants (epitopes) of an allergen (eg, ragweed) used in a skin test. This phenomenon, referred to as “food cross-reactivity,” effectively augments the immune response. Finally, intradermal wheal tests that are placed too close together (<2 cm) that cause positive wheal and flare reactions may initiate axonal reflexes that can further enhance other proximate whealing responses . Under many circumstances these factors must be minimized, if not eliminated, before performing skin tests, or at least must be accounted for in interpreting the results of skin tests.
Therefore, before proceeding with skin testing, the clinician needs to determine that the skin responds normally to mast cell provocation by exhibiting the usual effects of mast cell degranulation and mediator release that lead to normal wheal growth. Likewise, it also is necessary to make sure that the skin does not respond abnormally by exhibiting unsuspected degranulation and thus abnormal or unusual wheal growth to stimuli that ordinarily should not cause degranulation . These goals should be easy to accomplish by taking a good history and by performing positive- and negative-control tests. If these control tests do not yield appropriate results, skin testing should be suspended, because the results of further tests probably will not be reliable.
The positive-control intradermal test will determine the capability of the patient’s skin to mount a histamine-mediated wheal and flare reaction. This test is administered by creating a 4-mm wheal using histamine at a strength of approximately 0.004 mg/mL ( Box 2 ) and should yield a 7-mm or larger wheal after 10 minutes (a positive response). If this test fails to produce a positive response, false-negative responses should be expected if allergenic extracts are also applied; thus, testing should be suspended for that day, and a search for wheal-inhibition factors should be performed. In vitro tests may be indicated in this situation .
Method 1
Mix 2 mL of aqueous (not glycerinated) histamine phosphate (0.275 mg/mL) with 3 mL of phenolated saline. Then make a #2 dilution by titrating two times (1:5 dilutions).
Method 2
Use aqueous (not glycerinated) histamine phosphate (2.75 mg/5 mL). Then make a #3 dilution by titrating three times (1:5 dilutions).
The negative-control intradermal test, akin to a placebo control, will determine the capability of the patient’s skin to resist a false-positive wheal or flare reaction to what should be an innocuous substance. This test is accomplished by placing a 4-mm intradermal wheal using a diluent such as phenolated saline or human serum albumin. The 4-mm negative-control wheal should not enlarge beyond 5 mm after 10 minutes. If further enlargement does occur, such “positive” wheal growth would be assumed to occur also when antigens are applied, yielding false-positive test results. If a false-positive response occurs, further skin testing should be suspended, and a search for nonallergic wheal-enhancement factors should be sought. In vitro tests may be indicated in this situation.
Glycerin is the preservative used in almost all manufactured allergenic extracts. Because glycerin sensitivity can cause wheal enhancement and thus false-positive results, the glycerin-control test, which is another variation of a negative-control test, becomes necessary when glycerin-containing extracts are used.
Most extract concentrates used by the clinician contain 50% glycerin. Testing should not be performed with intradermal tests using extract concentrates, but there may be instances in which weaker dilutions prepared from these concentrates still contain high concentrations of glycerin. For instance, preparation of #1, #2, and #3 dilutions using the fivefold concept will yield concentrations of 10%, 2%, and 0.04% glycerin, respectively. Many allergists use a 10-fold dilution system, in which 1:100, 1:1000, and 1:10,000 dilutions yield glycerin concentrations of 5%, 1%, and 0.05%, respectively. To eliminate the possibility of false positives from glycerin sensitivity, these glycerin concentrations need to be tested in the absence of antigen if they are to be used in antigen testing.
For the fivefold system, the glycerin-control test is accomplished by creating fivefold dilutions #1, #2, and #3 from a vial containing 50% glycerin. One then creates 4-mm wheals from the strongest dilution anticipated during antigen testing and reads the test after 10 minutes. If the test is negative (little or no wheal growth), there is no need to test other glycerin dilutions. If the test is positive (≥7 mm), weaker dilutions should be tested up to a #3 dilution if the use of comparable dilutions is anticipated during antigen testing. If responses to any of the first three dilutions are positive (≥7 mm), wheals from antigen dilutions of comparable glycerin strength must be compared with these control wheal sizes and interpreted with caution. If the wheal growth from an antigen test is 2 mm larger than a glycerin-control test at the same dilution, the antigen test is considered positive and valid. Therefore, a glycerin-control test that yields positive wheal growth (≥7 mm) does not necessarily contraindicate further skin testing, but caution should be exercised.
If control tests have yielded the appropriate results, the clinician can proceed with either single-dilution or multiple-dilution intradermal testing. Various dilutions for use in testing should be prepared ahead of time in individual glass vials and should be refrigerated when not in use. These vials should be arranged in a logical sequence in a test tray/board. Dilutions in common use by otolaryngologists are based on a fivefold system. These dilutions are created by withdrawing exactly 1 mL of antigen from the stock bottle with a one-piece sterile needle syringe and injecting it into a 5-mL vial containing 4 mL of diluent. This vial is labeled the #1 dilution and now contains antigen that is five times less concentrated than the concentrate in the stock bottle. The #2 dilution, which will contain antigen that is five times less concentrated than the #1 dilution, is created by withdrawing exactly 1 mL from the #1 vial and injecting it into another 5-mL vial containing 4 mL of diluent. This process usually is repeated until dilutions #1 through #6 have been created. This sequence is repeated for each antigen anticipated for testing. In clinical practice it almost never is necessary to make or test with dilutions weaker than the #6 dilution.
Diluents are relatively inert liquids that are used to dilute the strengths of allergenic extracts to create various dilutions. The most popular diluent in use is phenolated normal saline, because it is relatively inexpensive, nonirritating, and sterile because of the 0.4% phenol that is added. Some clinicians use phenolated human serum albumin, which reduces adherence to the walls of a glass vial (“walling effect”), but it is more expensive and does have the theoretical risk of promoting human viral transmission.
Because of protein degradation, the potency of allergenic extracts can decrease over time, but glycerin is an effective preserver of potency and sterility at strengths of 50% for up to 3 years. Glycerin also prevents “walling” in glass vials. For these reasons, extract concentrates used by most clinicians are placed by the manufacturer in vials containing 50% glycerin.
As one prepares dilutions #1 through 6, however, both glycerin concentrations and antigen concentrations become weaker and weaker. Thus, the potency-preserving properties of the glycerin dissipate, and the potency of most dilutions prepared on a test board becomes unpredictable after 6 to 8 weeks, if properly refrigerated, and even earlier if not properly refrigerated. Therefore it is necessary to replace all dilutions with newly prepared dilutions for intradermal testing every 6 to 8 weeks, and these dilutions should be refrigerated as much as possible .
Single-dilution intradermal testing
Intracutaneous (intradermal) testing using a single dilution of an antigen has been practiced for many decades. Following appropriate responses from the placement of intradermal positive and negative controls, a 4-mm intracutaneous wheal is created by injecting a small amount of antigen (usually 0.01–0.02 mL) using a one-piece 27-gauge syringe. The wheal is remeasured within 10 to 20 minutes. If the wheal has grown to 7 mm or greater, the test is positive. The dilution used depends on variable factors; but commonly used single dilutions include 1:500 (the #2 dilution in a 5-fold system), 1:1000 (the #2 dilution in a 10-fold system), 1:10,000 (the #3 dilution in a 10-fold system), and 1:12,500 (the #4 dilution in a fivefold system) ( Table 1 ).
| 10-fold system | 5-fold system | ||
|---|---|---|---|
| Concentrate | 1:100 w/v a | Concentrate | 1:20 w/v a |
| #1 Dilution | 1:100 w/v | #1 Dilution | 1:100 w/v |
| #2 Dilution | 1:500 w/v | ||
| #2 Dilution | 1:1,00 w/v | ||
| #3 Dilution | 1:2,500 w/v | ||
| #3 Dilution | 1:10,000 w/v | ||
| #4 Dilution | 1:12,500 w/v | ||
| #5 Dilution | 1:62,500 w/v | ||
| #4 Dilution | 1:100,000 w/v | ||
| #6 Dilution | 1:312,500 w/v | ||
| #5 Dilution | 1:1,000,000 w/v | ||
a Concentrate w/v is commonly 1:10, 1:20, or 1:100. 1:10 represents 1 gram extracted in 10 mL of solution .
Testing with a single dilution is quick and efficient; however, testing with one dilution by itself does not give the quantitative or relational information that would be provided by testing responses to additional dilutions of different strengths. In addition, the use of a single-dilution intradermal test with concentrated antigen can be accompanied by an increased risk of significant adverse systemic or local responses. To that end, some clinicians couple a single-dilution test with either another intradermal test using a weaker dilution or with a prick/puncture test. Many clinicians, however, use only one dilution to qualify the presence of IgE-mediated allergy; others also may quantitate sensitivity roughly based on the size of the wheal growth from that one single dilution.
Multiple-dilution intradermal testing using serial dilutions
Intracutaneous (intradermal) testing using multiple serial dilutions of the same antigen has been practiced for many decades. This method of intradermal testing was introduced in the 1930s but probably was most popularized in the 1950s by Herbert Rinkel .
The historical terms for intradermal testing using multiple serial dilutions were “serial dilution testing” and “skin endpoint titration.” These names have been replaced by the term “intradermal dilutional testing.”
Proponents of this technique point out that patients vary in sensitivity to individual antigens and therefore vary in tolerance to initial antigen doses when starting immunotherapy . Assessing test accuracy over a range of antigen strengths serves both to confirm allergy and to quantify the intensity of the response. The latter information allows more precise tailoring of beginning immunotherapy doses, which in theory will bring relief in a more efficient and timely manner without compromising safety. In addition, the premise that some patients’ allergies are less sensitive than others suggests that some patients may not always test positive to single-dilution tests containing weaker dilutions or to prick/puncture tests; thus they falsely will be labeled negative.
Whether or not one chooses actually to perform intradermal dilutional testing on patients, an understanding of its principles facilitates a better understanding of all other forms of testing. The clinician who understands the relationship of intradermal dilutional testing to other testing can move with facility between these methods and thus diversify diagnostic options .
In intradermal dilutional testing, consecutive dilutions of antigenic extracts are applied in a sequential manner from weakest to strongest. Positive whealing demonstrates the presence of allergy (qualitative test), and comparison of whealing responses at different dilutions demonstrates the degree of sensitivity (quantitative test). Quantitation of sensitivity allows the safe initial starting dose for desensitization immunotherapy to be determined.
The first technical step in intradermal dilutional testing is the application of positive, negative, and glycerin controls. If these control tests do not yield appropriate results, skin testing should be suspended, because the results of further tests probably will not be reliable. The second step is to create skin wheals containing test antigens for titration testing. All wheals produced should be 4 mm in diameter and should be read 10 to 20 minutes after the wheal is created. Begin with a test dilution that is anticipated to be weak enough (usually #6 dilution) to produce a negative response. Then apply progressively stronger dilutions to the point at which either a positively reacting strength is confirmed with the next strongest dilution or all wheals are negative down to a #2 dilution. If the patient is allergic to the test antigen, there will be a series of wheals that progresses from negative to positive and shows increasingly larger wheals with stronger dilutions .
For positive responses, the end point of titration is the first or weakest antigen dilution that produces a positive wheal ( Fig. 1 ). It is the intradermal dilution at which the patient’s response turns from negative to positive. This end point represents the level (ie, the intensity) of the patient’s sensitivity to that particular antigen. The end point represents the antigen dilution that may be used safely to begin immunotherapy. To confirm that the end point is authentic, Rinkel advocated the placement of a “confirming” skin wheal using an antigen strength one dilution stronger than the one that produced the end point wheal. Because this confirming dilution is five times stronger than the end point dilution, it should produce a wheal that is at least 2 mm greater in diameter than the preceding wheal. This confirming wheal validates the end point (see Fig. 1 ).
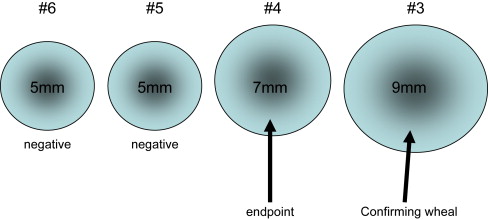
Despite the application of appropriately responding controls, unusual whealing responses that make interpretation challenging still may occur. Occasionally, the next stronger wheal following the first positive wheal grows only to the same size as the first positive wheal. This phenomenon is called a “plateau” response because there is no progression of wheal growth, as should occur with the confirming wheal. Ideally one then would apply the next stronger dilution in hopes of identifying a confirming wheal. If a confirming wheal results, the positive wheal that immediately preceded the confirming wheal is the end point because it is the dilution that initiated progressive whealing. Occasionally a “flash” whealing response will occur, which is a negative wheal followed by a very large wheal (usually ≥ 10–12 mm) in response to the next stronger dilution. The cause of the “flash response” is unknown but is postulated to be the ingestion of a cross-reacting food (eg, melons in patients allergic to ragweed) or the age/potency of the antigen being tested. Repeating the test in a few days generally yields a more conventional response .
In a series of negative responses for one antigen, how far does one titrate intradermal dilutional tests to look for a positive result? As long as titrations are negative, antigens are tested down to the #2 dilution. If no positive wheal occurs by that point, most clinicians end the test for that antigen and consider the patient to be negative to that antigen. Clinicians should not test with a #1 dilution if a #2 test was negative. The only time to place a #1 dilution for inhalant testing is to confirm a #2 end point. There are no objective data supporting the clinical significance of a positive #1 dilution if that dilution is the only positive dilution. Furthermore, performing intradermal tests with full-strength (undiluted) extract concentrates to confirm a positive #1 dilution poses unacceptable risk of anaphylaxis. Therefore, one should not perform intradermal testing with full-strength (undiluted) extract concentrates .
There is no definite, universally followed pattern in the number of dilutions applied at one time for each antigen or which dilution strength is placed first. There are, however, some general guidelines that should be followed. First, one should start with relatively weak dilutions and work toward stronger dilutions. As a general practice, it is safe to begin testing most patients at a #4 dilution to limit the number of needle sticks necessary for testing. Because of the potential for increased sensitivity, however, weaker dilutions (#6 dilutions) should be used when testing (1) patients who have asthma (especially steroid-dependent asthma), (2) patients receiving beta-antagonist medication, (3) patients who have a history of previous significant reactions to skin testing or immunotherapy, (4) patients who have had a recent large exposure to suspect antigens, and (5) for antigens that are in season .
Intradermal dilutional testing can be time consuming, painful, labor intensive, and expensive. In an effort to save time, needle sticks, and costs, some shortcuts that are acceptable. One can save a small but significant amount of time and number of steps by using “simultaneous dual consecutive dilutions.” This testing is accomplished by first applying a #6 dilution of the antigen in question. If the result is negative, place the next two stronger dilutions (#5 and #4). If these results are negative, place the next two stronger dilutions (#3 and #2). If any dilution is positive, allow pattern and size to determine the next step in an effort to identify the end point . Another time- and cost-saving variation on classical intradermal dilutional testing is using extrapolation. Begin at either a #4 or #6 dilution (in low-risk or high-risk patients, respectively) as a screen and then, depending on positive or negative wheal responses, test only even-numbered dilutions (#4 and #2) and extrapolate the end point.
There are several benefits of intradermal titration testing. It is both qualitative and quantitative. Because allergy sensitivities change with antigen exposure , multiple samplings over a continuum of dilution strengths beginning with weak dilutions give an accurate and safe appraisal of allergic sensitivity, regardless of whether the allergen is in season. The test is highly sensitive, because patients who have relatively low sensitivity to certain antigens and who may react only to stronger dilutions are identified as the skin titration progresses. It also provides a methodical approach to determining a safe starting dose for immunotherapy, regardless of the patient’s current ambient exposure to those antigens . Although circumstances may make it impractical to perform a full titration on every patient, the principles of quantitative testing and development of a treatment regimen tailored to each individual patient remain valid.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


