Purpose
To report 2-year follow-up data after Ahmed valve implantation (New World Medical, Inc) and Molteno single-plate implantation surgical treatment of refractory glaucoma.
Design
Prospective, randomized, comparative study.
Methods
Patients with refractory glaucoma, defined as uncontrolled intraocular pressure (IOP) of more than 21 mm Hg despite maximal antiglaucoma medication, previously failed nonseton surgical treatment, or a combination thereof were included. Ninety-two patients were allocated randomly to each of the study groups and underwent implantation of either the Ahmed valve implant (model FP7; 184 mm 2 surface area) or Molteno single-plate implant (134 mm 2 surface area) and were followed up for 24 months. Main outcome measures were IOP and surgical success rate. Other outcome measures were changes in visual acuity, number of ant-glaucoma medications, mean deviation of visual field, and rate of intraoperative and postoperative complications.
Results
Those who successfully completed the trial (28 in the Molteno group and 29 in the Ahmed group) achieved significantly less IOP and fewer glaucoma medications, but worse visual acuity 24 months after surgery. The Molteno group, compared with the Ahmed group, achieved significantly lower IOPs after the early postoperative period until the end of the study. Both groups reasonably maintained visual field during the follow-up. The rate of surgical failure was comparable in both groups. Median survival time was 24 months for both groups. There were no devastating intraoperative or postoperative complications in either group.
Conclusions
Both Ahmed and Molteno implants successfully preserved visual field, although IOP control was more pronounced in the Molteno implant group.
Historically, glaucoma drainage implants have been reserved for cases of refractory glaucoma deemed at high risk of failure (e.g., eyes with a previous history of failed trabeculectomy and with neovascular and uveitic glaucoma) with standard filtration surgery. Despite the high-risk profile of these patients, moderately good levels of success with various types of glaucoma drainage implants have been observed. Consequently, the use of these implants has increased in recent years, and they have become the preferred surgical procedure for complex cases.
For more than 2 decades, various types of glaucoma drainage implants have been developed. These implants may be subdivided into valved and nonvalved designs. Those with a nonvalved structure act as an open tube, which may result in immediate postoperative hypotony and its attendant complications (e.g., flat anterior chamber, suprachoroidal effusion, and suprachoroidal hemorrhage). Hypotony during the immediate postoperative period is less common with valved drainages, although it may still occur. We investigated the success of the Ahmed valve implant (New World Medical, Inc, Rancho Cucamonga, California, USA) and the Molteno single-plate implant (Molteno Ophthalmic Limited, Dunedin, New Zealand) in treating eyes with refractory glaucoma during a follow-up period of 24 months.
Methods
Study Subjects
A prospective, randomized trial was performed from January 2003 through August 2005 at 3 medical centers, namely, Imam Hossein Medical Center, Negah Eye Hospital, and Vanak Eye Surgery Center, all in Tehran, Iran. The eligibility criteria were refractory glaucoma, defined as uncontrolled intraocular pressure (IOP) despite maximal antiglaucoma medication, previously failed nonseton surgical treatment, or a combination thereof. The exclusion criteria included age younger than 40 years, a visual acuity of no light perception, lens opacity, elevated IOP associated with silicone oil, previous glaucoma drainage device implantation in the same eye, previous cyclodestructive treatment, increased risk of endophthalmitis (e.g., active adnexal and ocular surface infection, immunosuppression, or immunodeficiency, including the use of systemic steroids), posterior segment disorders, or pre-existing ocular comorbidities (e.g., pterygium, phacodonesis, corneal opacity, or corneal endothelial dystrophies). Only 1 eye per patient was included in this trial.
An estimation of sample size was performed considering a study power of at least 0.8 with an α error of 0.05 aiming to detect a difference of 1 mm Hg in mean IOP in the postoperative month 24 between the 2 groups. Mean IOP in the postoperative month 24 was estimated to be 15 mm Hg in the Ahmed group based on the results of the previous studies. Based on this estimation, a total of 64 study subjects was deemed adequate, and considering a total of 30% assumed dropout and failure rate during the follow-up, recruitment of at least 92 study subjects was targeted.
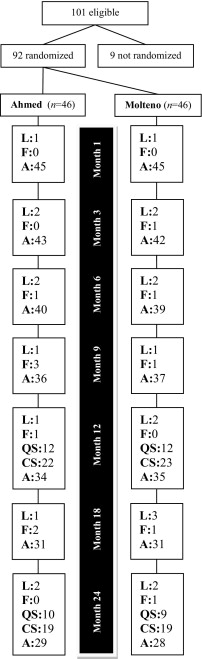
Of 101 eligible patients, 92 (91.0%) agreed to participate. Study subjects were assigned randomly to either the Ahmed or Molteno groups. Randomization was performed using a random permuted block design with a block size of 2, stratified for age, sex, and hosting medical center. Neither patients nor investigators were masked to study groups. Figure 1 shows a flow chart of the trial. Operations were all performed by the same senior right-handed surgeon (Nad.N.). To reduce the risk of potentially permanent visual loss, all patients underwent surgery within 4 weeks of enrolling in the trial. Before operation, the exclusion criteria again were reviewed by the surgeon.
Preoperative and Postoperative Assessments
Presurgical assessments included slit-lamp biomicroscopic examination, funduscopy, Snellen visual acuity (VA), baseline IOP, number of antiglaucoma medications, and Humphrey perimetry results. Follow-up visits were scheduled at day 1, week 1, and months 1, 3, 6, 9, 12, 18, and 24, and more often when necessary. Postoperative evaluations included assessment of visual acuity, IOP (in millimeters of mercury), number of antiglaucoma medications, lens opacity, and postoperative complications. Humphrey perimetry was performed at postoperative months 3 and thereafter. A formal motility evaluation was performed in all patients at baseline and at the 1-year and 2-year follow-up visits, as well as during any visits after 3 months if the patient reported diplopia.
The best-corrected visual acuity was measured using a Snellen chart (CP-690; Nidek Co, Ltd, Gamagori Aichi, Japan) calibrated for a 20-foot (approximately 6 m) distance by the line assignment method within the month before the surgery; the figures were converted to logarithm of the minimal angle of resolution notation by the standard conversion table. IOP was measured using a Goldman applanation tonometer (AT-900; Haag-Streit AG, Koniz, Switzerland) mounted on a slit lamp; if required, gonioscopy (Haag-Streit AG) also was performed. The preoperative IOP for each eye was considered as the last measure before operation.
24–2 threshold visual field assessment was performed with the Humphrey Field Analyzer (Allergan-Humphrey Instruments, San Leandro, California, USA). The mean deviation—a visual field index provided by Statpac-2 (Allergan-Humphrey Instruments) and determined from a comparison with an age-matched normal population—was available as a global assessment of visual field damage. We considered the phenomenon of learning effect in our analysis of mean deviation; this is a phenomenon whereby the baseline visual field is worse than in subsequent tests, so using baseline tests may result in a false-positive defect being compared with actual glaucomatous progression or the development of a cataract, and not stable fields. To avoid this, we included only results where consecutive field tests did not show an improvement of 2 dB or more in the mean deviation over the baseline reading.
Outcome Measures
IOP (primary outcome measure), number of antiglaucoma medications, visual acuity, visual field, and surgical failure were considered as measures assessing efficacy; intraoperative and postoperative complications were considered as measures assessing safety. Surgical failure was defined as persistent IOP of more than 21 mm Hg on maximally tolerated medications or IOP less than 6 mm Hg on 2 consecutive visits, phthisis bulbi, reduction of vision to no light perception, removal of the shunt implant, reoperation for glaucoma, or any devastating intraoperative or postoperative complication. Reoperation was defined as additional glaucoma surgery requiring a return to the operating room, such as placement of a tube shunt. Interventions performed at the slit lamp, such as needling procedures, were not considered reoperations. Surgical revision of the glaucoma shunt implants for other postoperative reasons (e.g., tube repositioning or repair of conjunctival wound dehiscence) were recorded, but were not considered surgical failures. Devastating complications included suprachoroidal hemorrhage that threatened visual fixation, was likely to develop a kissing appearance, or both; malignant glaucoma; endophthalmitis; retinal detachment; chronic hypotony; or serous choroidal effusions necessitating surgical drainage or that had a kissing appearance.
Treatment success was subdivided into complete and qualified. A complete success was defined as IOP between 6 and 21 mm Hg without any antiglaucoma medication, and qualified success was defined as IOP between 6 and 21 mm Hg with 1 medication or more. Hypotony was defined as an IOP of less than 6 mm Hg on 2 consecutive visits; a hypertensive phase was defined as an IOP of more than 21 mm Hg during the first 3 postoperative months (with or without medications) after a reduction in IOP to less than 22 mm Hg during the first postoperative week and not caused by tube obstruction, retraction, or valve malfunction. Resolution of this phase was IOP of less than 22 mm Hg along with a reduction of the IOP by 3 mm Hg or more with the same or fewer number of medications, or reduction of at least 1 medication with a change in of IOP less than 3 mm Hg.
Procedure
The tube shunts used were either the valved 184-mm 2 surface area Ahmed Glaucoma Valve (AGV Model FP7) or the 134-mm 2 surface area single-plate Molteno implant. Before the trial, the surgeon made a transition from the polypropylene (model S2) to the silicone (model FP7) Ahmed valve. Recent studies have reported significantly lower failure rates with silicone valves. General anesthesia was given in most cases; in some cases, a peribulbar block consisting of lidocaine 2% (Xylocaine; AstraZeneca, Cheshire, United Kingdom) with hyaluronidase (Hyalase; Wockhardt Ltd, Wrexham, United Kingdom) was used. Immediately before surgery in the operating room, povidone–iodine 5% was applied to the skin of the eyelids of the operative eye, nose, and forehead. The operative eye was draped in the usual manner for ophthalmic surgery. In both types of shunts, a clear corneal traction suture was placed with silk 7-0, and the fornix-based conjunctival flap and Tenon capsule were fashioned supratemporally. To prime the valve, the tube of the shunt was irrigated with a balanced saline solution (BSS; Alcon, Fort Worth, Texas, USA). The plate was secured 8 to 9 mm posterior to the surgical limbus with interrupted 7-0 nylon sutures using tapered cutting needles.
In the Molteno implant group, the anterior chamber drainage tube was occluded by tying a 7-0 Vicryl suture (Ethicon, Norderstedt, Germany) around its exterior, where it joined the plate of the implant, to restrict the flow through the device temporarily until encapsulation of the plate occurred. Fenestrations were placed anterior to the suture using a 28-gauge needle to allow leakage of the aqueous, providing initial IOP control. Next, in both groups, the tube was trimmed to extend approximately 2 to 3 mm beyond the surgical limbus with the bevel facing up; it then was entered into the anterior chamber by a 23-gauge needle parallel to the iris plane. If a peripheral iridectomy was present from a previous surgery, the needle tract was placed slightly to one side of the iridectomy. Sodium hyaluronate (Healon, 10 mg/mL; Advanced Medical Optics, Santa Ana, California, USA) was injected to prevent sudden collapse of the anterior chamber. The tube was inserted with smooth forceps through the needle tract, ensuring that no iris or corneal touch occurred. In patients with aphakic and pseudophakic glaucoma who had vitreous in the anterior chamber, an anterior vitrectomy was performed before the placement of the tube. The tube then was anchored to the sclera using a 10-0 nylon suture and was covered with a rectangular piece of donor scleral patch graft. The conjunctiva was closed using 10-0 interrupted nylon sutures. A clear cornea paracentesis then was created to remove the Healon as much as was possible, and the anterior chamber then was reformed with BSS. At the end of surgery, subconjunctival injections of antibiotics and corticosteroids were administered away from the surgical site. No adjunctive antimetabolite was used in any of the cases. Postoperative management consisted of combination topical antibiotic and steroid eye drops, which were tapered over the course of 6 to 8 weeks. Antiglaucoma medications were removed or added as needed, depending on the IOP and clinical status of the operated eye.
Statistical Analysis
Preoperative and postoperative measures were compared within and between study groups. Quantitative variables were compared using nonparametric tests, that is, the Mann–Whitney U test or Wilcoxon signed-rank test, whichever appropriate. The chi-square and Fisher exact tests were used for qualitative variables. To determine factors independently associated with IOP in postoperative month 24, multivariate analysis was performed using a stepwise linear regression model of several variables. The cumulative probability of success was analyzed by Kaplan-Meier life-table analysis. As soon as an eye reached a failure end point, it was censored from further analysis. Cox proportional hazards analysis was used to identify independent predictors of surgical failures. Post hoc study power for significant comparisons was calculated using G*Power 3, a power analysis software. P values less than .05 were considered statistically significant.
Results
Baseline variables were comparable in both groups ( Table 1 ). Those who successfully completed the trial in both groups (28/46 [60.9%] in the Molteno group and 29/46 [63.0%] in the Ahmed group; Figure 1 ) achieved significantly lower IOP and fewer glaucoma medications, but worse visual acuity 24 months after operation in comparison with their preoperative measures ( Table 2 ).
| Characteristics | Ahmed Valve Implant (n = 46) | Molteno Single-Plate Implant (n = 46) | P Value |
|---|---|---|---|
| Demographic characteristics | |||
| Mean age (SEM), yrs | 59.4 (1.51) | 63.3 (1.62) | .08 |
| Male/female, n | 25/21 | 22/24 | .67 |
| Background conditions, n (%) | |||
| Diabetes mellitus | 11 (23.9) | 11 (23.9) | 1.00 |
| Hypertension | 13 (28.3) | 13 (28.3) | 1.00 |
| Family history of glaucoma | 11 (23.9) | 10 (21.7) | 1.00 |
| Bilateral glaucoma | 12 (26.1) | 14 (30.4) | .82 |
| Previous glaucoma treatments, n (%) | |||
| Penetrating iridectomy | 8 (17.4) | 9 (19.6) | 1.00 |
| Trabeculectomy | 17 (37.0) | 18 (39.1) | 1.00 |
| No. of trabeculectomy procedures | .98 | ||
| 1 | 1 (2.2) | 1 (2.2) | |
| 2 | 14 (30.4) | 14 (30.4) | |
| 3 | 2 (4.3) | 3 (6.5) | |
| Enrolled eye baseline features | |||
| Side, right/left, n | 24/22 | 20/26 | .53 |
| Lens group, n (%) | .69 | ||
| Phakic | 31 (67.4) | 33 (71.7) | |
| Aphakic | 6 (13.0) | 7 (15.2) | |
| Pseudophakic | 9 (19.6) | 6 (13.0) | |
| Subtypes of glaucoma, n (%) | .82 | ||
| Failed filtration | 17 (37.0) | 19 (41.3) | |
| Pseudoaphakia | 9 (19.6) | 6 (13.0) | |
| Neovascular | 9 (19.6) | 12 (26.1) | |
| Aphakia | 6 (13.0) | 6 (13.0) | |
| Uveitic | 5 (10.9) | 3 (6.5) | |
| Mean IOP (SEM), mm Hg | 32.11 (1.32) | 32.41 (1.27) | .87 |
| Mean BCVA (SEM), logMAR units | 0.64 (0.09) | 0.61 (0.09) | .86 |
| Visual field mean deviation (SEM), dB | –18.57 (0.82) | –19.11 (0.79) | .64 |
| Mean no. of glaucoma medications (SEM) | 2.7 (0.08) | 2.7 (0.09) | .72 |
| Ahmed Valve Implant (n = 29) | Molteno Single-Plate Implant (n = 28) | |||||
|---|---|---|---|---|---|---|
| Preoperative | Month 24 | P Value a | Preoperative | Month 24 | P Value a | |
| Mean IOP (SEM), mm Hg b | 30.81 (1.67) | 17.00 (0.23) | <0.001 | 33.06 (1.66) | 15.36 (0.33) | <.001 |
| Mean BCVA (SEM), logMAR units | 0.66 (0.12) | 0.78 (0.12) | 0.001 | 0.59 (0.12) | 0.70 (0.11) | <.001 |
| Visual field mean deviation (SEM), dB c | −19.70 (0.96) | −19.67 (0.95) | 0.09 | −18.45 (1.11) | −19.49 (1.17) | <.001 |
| Mean no. of glaucoma medications (SEM) | 2.8 (0.11) | 1.03 (0.27) | <0.001 | 2.7 (0.11) | 1.41 (0.19) | <.001 |
a Wilcoxon signed-rank test comparisons of postoperative month 24 and preoperative figures in each study group, post hoc study powers for all significant comparisons were >80% (based on an α error of 0.05).
b P < .001, Mann–Whitney U test comparison of postoperative month 24 figures between the 2 study groups.
c Three subjects in the Ahmed group and 5 in the Molteno group were excluded from visual field calculations because of the learning effect, reducing total number to 26 in the Ahmed group and 23 in the Molteno group.
Intraocular Pressure and Other Eye Features
IOP was significantly lower in the Ahmed group than in the Molteno group in postoperative day 1 and week 1. Nevertheless, the Molteno group showed markedly lower IOPs thereafter up to the end of the study in postoperative month 24 ( Figure 2 ). The Molteno group, compared with Ahmed group, showed significantly higher mean (± standard deviation) percentages of change from preoperative IOP to postoperative month 12 IOP (−50.7 ± 11.6% vs −41.4 ± 13.9%; P = .004; post hoc study power, 91%) and month 24 IOP (−49.7 ± 11.6% vs −41.9 ± 14.6%; P = .049; post hoc study power, 71%). Meanwhile, the number of glaucoma medications after operation was not significantly different between the 2 groups ( Figure 3 ).
Visual acuity deteriorated in both groups during the 2-year follow-up and there were no significant between-group differences at any point during the study ( Table 2 ; Figure 4 ). In postoperative month 24, the mean deviation of visual field results remained comparable with the preoperative value in those who successfully completed the trail in the Ahmed group. However, the Molteno group experienced worsened mean deviation of the visual field results in postoperative month 24 than before surgery ( Table 2 ).

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


