Age-Related Macular Degeneration
Jie Jin Wang
Ronald Klein
Age-related macular degeneration (AMD), also termed age-related maculopathy (ARM), is the leading cause of irreversible blindness and visual impairment in older persons, and will remain a major threat to vision in coming decades (1). Research on AMD has progressed rapidly in recent decades (2, 3, 4, 5, 6, 7, 8 and 9). In particular, the identification of genetic variants responsible for AMD susceptibility (6, 7, 10, 11 and 12) and environmental risk or protective factors for AMD, has pointed to likely pathogenetic mechanisms for this condition (7, 9, 13, 14 and 15).
PREVALENCE AND INCIDENCE OF AGE-RELATED MACULAR DEGENERATION
In the last two decades there has been a growing number of large population-based studies conducted in different countries, providing estimates of AMD prevalence (16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32 and 33) and incidence (34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47 and 48).
AGE-RELATED MACULAR DEGENERATION PREVALENCE IN WHITES
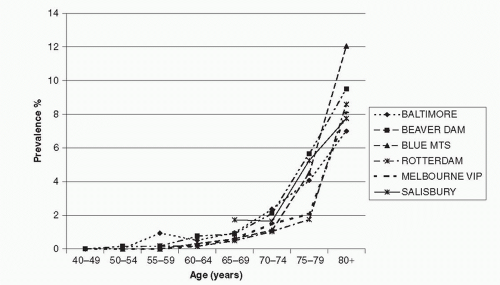
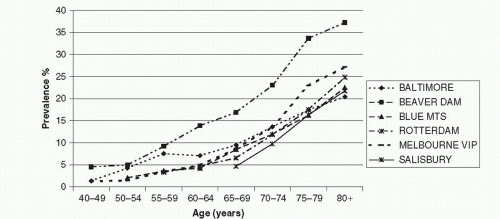
The meta-analysis conducted by the Eye Disease Prevalence Research Group (49), using data collected from multiple populations, including the Baltimore Eye Survey (20), the Beaver Dam Eye Study (BDES) (16) and the Salisbury Eye Evaluation (SEE) project (50) in the USA, the Rotterdam Study (RS) (17) in the Netherlands, and the Blue Mountains Eye Study (BMES) (18) and Visual Impairment Project (VIP) (22) in Australia, showed high consistency in the age-specific prevalence of late AMD (Fig. 30-1) and large soft drusen (≥125 μm in diameter) (Fig. 30-2) in whites. The prevalence of late AMD (geographic atrophy and neovascular AMD) increased exponentially with increasing age, rising from less than 0.5% at age 60 years to around 10% at age 80+ years (49) (Fig. 30-1). The prevalence of large drusen showed a similar age-related increasing trend but the association with age is more linear than exponential (49) (Fig. 30-2). The natural course of AMD (involving the disappearance of large drusen associated with the progression to a more advanced AMD stage with aging (36) could explain the observed age-related linear (rather than exponential) pattern in the prevalence of large drusen.
AGE-RELATED MACULAR DEGENERATION PREVALENCE IN OTHER RACES AND ETHNICITIES
Ethnic variation in the prevalence of AMD and AMD lesions was observed in studies with multiracial groups such as the Baltimore Eye Survey (20), National Health and Nutrition Examination Survey (NHANES III) (51) and the Multi-Ethnic Study of Atherosclerosis (MESA) (33), and was previously summarized from studies conducted prior to this century (52).
Blacks
Findings from the Baltimore Eye Survey (20), and the Barbados Eye Study (53) which examined a sample of blacks, have suggested that the prevalence of some AMD lesions, particularly late AMD, is lower in blacks than in whites. The same observation has been consistently reported by a number of studies conducted in populations in the USA (21,23,33 and 50). Possible selection bias may have occurred in some studies due to either low participation rate (21), a high proportion with ungradable
retinal photographs (20,21 and 23), or poor survival in the eldest among black participants (20). Other factors may also be responsible for the racial variations in AMD frequency, including choroidal melanin density (23) and gene polymorphisms that may predispose whites to a more severe AMD than blacks (19,20).
retinal photographs (20,21 and 23), or poor survival in the eldest among black participants (20). Other factors may also be responsible for the racial variations in AMD frequency, including choroidal melanin density (23) and gene polymorphisms that may predispose whites to a more severe AMD than blacks (19,20).
Mexican Americans
Mexican Americans (Hispanic whites) have been found to have a relatively low prevalence of late AMD but a similar prevalence of signs of early AMD compared to non-Hispanic whites (19,51). This observation was confirmed by data from the Los Angeles Latino Eye Study (LALES) (25) and the Proyecto Vision Evaluation and Research (VER) (32), both with large population-based samples of Hispanic whites, and the MESA Study (33), which used the same study methods, grading protocol and graders to assess AMD lesions in participants of four racial/ethnic groups (blacks, Hispanic, Chinese and whites).
Asians
Relatively well-conducted population-based studies of AMD prevalence in Asian populations have emerged in recent years, including studies conducted in Japan (28) (Funagata Study, unpublished data), India (29,31), and China (30).
The prevalence of early and late AMD in Japanese and Indian populations does not appear to be substantially different from that in whites of the Western world, according to the two studies that used retinal photographic grading to diagnose AMD and AMD lesions (28,31). However, the prevalence of soft drusen and retinal pigmentary abnormalities varies across studies (28,31) and differs from the corresponding prevalence reported in white populations of similar age (soft distinct drusen 10-16%, soft indistinct drusen 5-8%, and pigmentary abnormalities 7-13%)(16,18). While soft indistinct drusen were relatively infrequent in both Japanese (0.5%) (28) and Indians (2.2%) (31), the predominant early AMD lesion in the Japanese population was soft distinct drusen (8.4%) and not retinal pigmentary abnormalities (3.2%) (28), whereas in the Indian population, both soft distinct drusen (34.0%) and retinal pigmentary abnormalities (10.8%) were frequent (31). In a Chinese population aged 40+ years, the Beijing Eye Study team reported a very low prevalence of both early (1.4%) and late AMD (0.2%) (30).
Comparability of study findings from Asian populations to those of Western countries is still limited, due to different methods used across studies. Although the Wisconsin Age-Related Maculopathy Grading System has been used in these recent studies in Asia, the AMD grading used has not been validated either internally or externally (28,30 and 31). Standardized methodology in retinal photography and AMD grading, together with the use of uniform AMD definitions, are critical for new studies in Asia to confirm whether the differences in AMD frequency between Asian and Western populations are real.
There are other possible reasons that could explain the observed differences in AMD prevalence across studies and racial groups. It is worth noting that two Japanese studies both reported high prevalence rates of neovascular AMD in Japanese men, similar to those observed in whites but sharply contrasting with the much lower prevalence in Japanese women (28) (Funagata Study, unpublished data). Possible explanations for this gender difference may include a high prevalence of smoking among Japanese men and a higher frequency of polypoidal choroidal vasculopathy (PCV) among Japanese men compared to Japanese women (54). The complement factor H (CFH) polymorphism (the Y402H) has consistently been documented as a major AMD genetic marker in white patient samples. However, in Japanese and Chinese with late AMD, the frequency of the variant high risk genotypes (CC) or the risk allele (C) of CFH was much lower than that reported in whites with late AMD (55, 56 and 57). This may partly explain the low prevalence of late AMD observed in the Japanese and the Chinese. Different AMD-related gene variants may be manifest in Asian with AMD. The lack of an association of CFH Y402H polymorphism with late AMD in two independent case studies of Japanese patients suggest that different gene polymorphisms may play a role in the pathogenesis of AMD in Asians compared to non-Asian cohorts (55,56).
AGE-RELATED MACULAR DEGENERATION INCIDENCE IN WHITES
After the turn of the century a number of population-based studies, following the BDES (34,37), have provided AMD incidence in older white populations (36, 38, 39, 40, 41, 42, 43, 46 and 47).
The cumulative 5-year, person-specific incidence of early AMD has been reported to range from 7.9% to 8.7%, and the corresponding incidence of late AMD ranges from 0.9% to 1.1% in the three predominantly white populations across three continents, the BDES (34), RS (38) and the BMES (36), all of which used similar study protocols in AMD phenotype ascertainment. Similar to AMD prevalence, incidence of this condition is strongly age-related (34,36,38 and 39), showing an exponential increase with age (see Fig. 30-2 of the report by the RS (38).
The long-term (10 or more years) cumulative incidence and progression of AMD has been reported by the BDES (37,48), BMES (47), and the Copenhagen City Eye Study (42). Estimates from the two studies with reasonable follow-up rates (37,47) indicate that the person-specific 10-year incidence of early AMD ranged from 10.8% to 12.1%, and the corresponding incidence of late AMD ranged from 2.1% to 2.8%, after agestandardization of the BMES to the BDES population (47). The 15-year cumulative incidence of late AMD was 3.1% and of early AMD was 14.3% in the BDES (48). In addition to age, incidence of late AMD was also strongly related to the severity of early AMD lesions at baseline (37,47 and 48).
A substantially higher incidence of neovascular AMD has been observed in the second eye of clinic patients presenting unilateral neovascular AMD, observed not only in whites (58, 59, 60 and 61) but also in Japanese (62). In the BMES population, 29% of second eyes developed end-stage AMD (neovascular AMD or geographic atrophy involving the fovea) in subjects with unilateral end-stage AMD (36).
PROGRESSION
Progression of AMD from soft (distinct or indistinct) drusen, reticular drusen, or retinal pigmentary abnormalities to geographic atrophy or neovascular AMD has been well documented in clinical case series (60,63 and 64). In the BDES and BMES, early ARM lesion characteristics have been studied in detail in terms of distribution, location, size, lesion type, and area involved in relation to the risk of subsequent development of late AMD (37,65 and 66). Large soft drusen (≥125 μm in diameter), soft drusen with indistinct margins (indistinct soft or reticular drusen), or pigmentary changes involving large macular areas plus close proximity to the fovea indicate a much higher risk of subsequent late AMD development (37,65).
The Age-Related Eye Disease Study (AREDS) developed an ordinal AMD severity for risk prediction from pre-existing early AMD lesions defined from baseline retinal photographs (67,68). The BMES 10-year AMD incidence data validated this AREDS AMD Simplified Severity Scale and provided further evidence supporting that incidence of late AMD strongly relates to the severity of early AMD lesions at baseline (47).
AGE-RELATED MACULAR DEGENERATION INCIDENCE IN OTHER ETHNICITIES
Blacks
The Barbados Eye Study reported 4-year (40) and 9-year incidence (46) of AMD in a black population aged 40-84 years at baseline. At the 4-year visit (40), incident neovascular AMD was observed in one of the 2362 persons at risk (0.04%) and incident geographic atrophy in none of the 2419 at risk (0%). The incidence of early AMD lesions, including intermediatesized drusen, was also low (5.2%, 60/1160). However, at the 9-year visit, the long-term incidence of early AMD was 12.6% and of late AMD 0.7% (46). This 9-year incidence of early AMD is similar to that reported from the BDES and BMES (37,47), but the 9-year incidence of late AMD is lower than that observed in whites (37,47). In the AREDS participants who had early or intermediate AMD at baseline, the risk of progression to neovascular AMD over a median follow-up period of 6.3 years was nearly 7-fold in whites than in blacks (odds ratio 6.8, 95% confidence interval 1.2-36.9) (69).
Other Ethnicities
To date, there are no Hispanic white population-based data available on AMD incidence, and only one report from a Japanese population on the 5-year incidence of AMD (44). Early AMD incidence was 8.5% and late AMD incidence was 0.8% in Japanese aged 50+ years at baseline, with 1.9% in men but only 0.2% in women. The nearly 10-fold difference in incidence rates between Japanese men and women is consistent with findings of AMD prevalence among Japanese, possibly due to high frequency of smoking and high incident PCV cases in Japanese men (54).
EMERGING AMD RISK AND PROTECTIVE FACTORS
Smoking
Smoking is a major modifiable risk factor for AMD, consistently found not only in cross-sectional population-based studies (70, 71, 72 and 73) (Fig. 30-3A), but also in longitudinal studies (69, 74, 75, 76, 77, 78, 79 and 80) (Fig. 30-3B). Pooled data estimated that risk for 5-year incidence of late AMD was more than double (odds ratio [OR] 2.35, 95% confidence interval [CI] 1.30-4.27) in current smokers compared to persons who never smoked (77) (Fig. 30-3B). Overall, the magnitude of risk for late AMD in current versus noncurrent smokers, or in past versus nonsmokers, ranges between 2- and 5-fold (80). In the BMES population (76), current smokers developed late AMD at a mean age of 67 years,
while past smokers developed late AMD at a mean age of 73 years and nonsmokers at a mean age of 77 years. This 10-year average difference in the mean age of developing late AMD in smokers versus nonsmokers implies a substantial increase in the burden on affected individuals, their families, and aged disability care. Evidence supporting a causal role for smoking in AMD includes its dose-response relationship with pack-years of smoking (81,82), reduced risk after smoking cessation (the earlier smoking cessation occurs, the lower the risk of developing AMD later in life (71, 81, 82, 83 and 84), and evidence from animal models (85). Passive smoking exposure has also been documented to be associated with AMD in a case control study (81), but not in a population-based study (86). Smoking as a major AMD risk factor has also been documented in Mexican American (87), Indian (88), and Japanese (44) populations.
while past smokers developed late AMD at a mean age of 73 years and nonsmokers at a mean age of 77 years. This 10-year average difference in the mean age of developing late AMD in smokers versus nonsmokers implies a substantial increase in the burden on affected individuals, their families, and aged disability care. Evidence supporting a causal role for smoking in AMD includes its dose-response relationship with pack-years of smoking (81,82), reduced risk after smoking cessation (the earlier smoking cessation occurs, the lower the risk of developing AMD later in life (71, 81, 82, 83 and 84), and evidence from animal models (85). Passive smoking exposure has also been documented to be associated with AMD in a case control study (81), but not in a population-based study (86). Smoking as a major AMD risk factor has also been documented in Mexican American (87), Indian (88), and Japanese (44) populations.
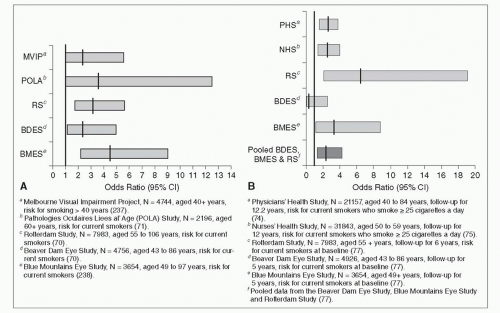 Figure 30-3. A: Smoking and prevalence of late age-related macular degeneration. B: Smoking and incidence of late age-related macular degeneration. |
Evidence of synergistic effects of history of smoking with other factors contributing to high AMD risk has been emerging recently (82, 89, 90, 91, 92, 93 and 94). Carriers of the APOE ε2 allele had a much higher risk of neovascular AMD if they smoked, compared to APOE ε4 or APOE ε3 carriers who also smoked (89). For the two confirmed AMD-related gene loci (CFH and LOC387715), smoking exerts a much greater effect on AMD risk in persons homozygous for the risk alleles of either of these two gene polymorphisms (Y402H in CFH and rs10490924 in LOC387715), compared to subjects homozygous for the nonrisk alleles (90, 91, 92, 93, 94 and 95) (Figs. 30-4 and 30-5). The odds ratio (OR) for AMD among individuals homozygous for the risk alleles who also smoked was two- or three-fold compared to subjects with either factor alone (nonsmoking, or absence of the risk alleles) (91,92,94 and 96). In the BMES 10-year follow-up data, biological synergism of smoking with the lowest level of serum high-density lipoprotein (HDL) cholesterol, the highest total to HDL cholesterol ratio, or history of low fish consumption with AMD risk was suggested (82).
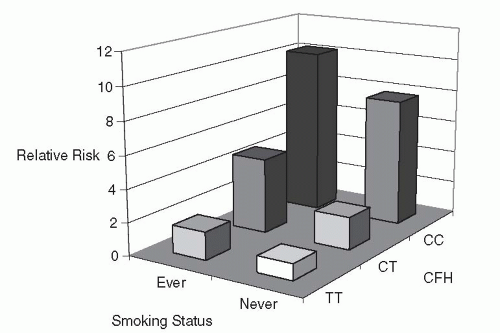 Figure 30-4. Joint effect of CFH-Y402H gene and smoking on risk of age-related macular degeneration (93). CFH, complement factor H; CC; CT; TT. |
Based on currently available evidence, cigarette smoking is considered an important trigger or promoter in the course of the development of late AMD (80




Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree