Fig. 7.1
Flood imaging adaptive optics funds camera developed by Imagine Eyes
7.3 Results
7.3.1 AO Imaging of Drusens and Geographic Atrophy
We examined patients with variable severity of GA. Drusens appeared as hyperreflective, round- or doughnut-shaped lesions with often a hyporeflective center. The photoreceptor mosaic remains visible over the drusen in most cases (Fig. 7.2).
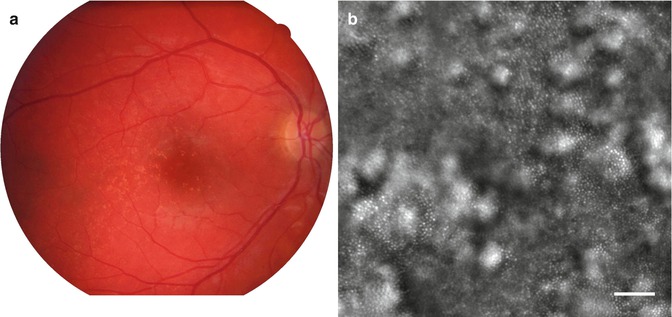

Fig. 7.2
Typical aspect of drusens by AO imaging. (a) Left, color photograph; (b) right, AO image of the area indicated in the square (bar, 100 μm)
In areas of GA, as compared to scanning laser ophthalmoscope imaging, AO NIR imaging dramatically improved the resolution of the limits of atrophic areas as well as the redistribution of melanin-loaded cells (Fig. 7.3). A striking feature was indeed the presence of multiple hyporeflective clumps within and around GA areas, which gave a “salt and pepper” appearance to the posterior pole [2]. Of particular interest are the cases with foveal sparing, for which AO can give a very precise delineation of the central island of preserved RPE cells (Fig. 7.4).
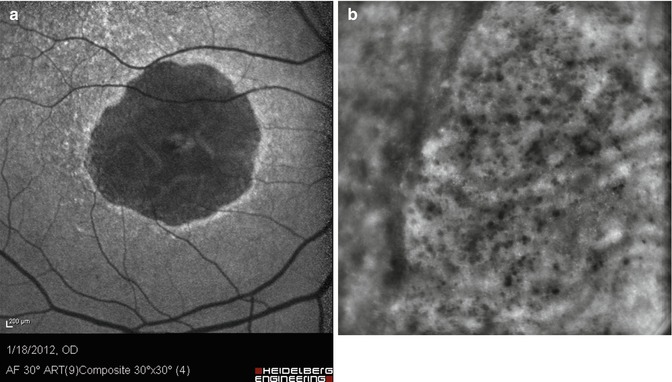
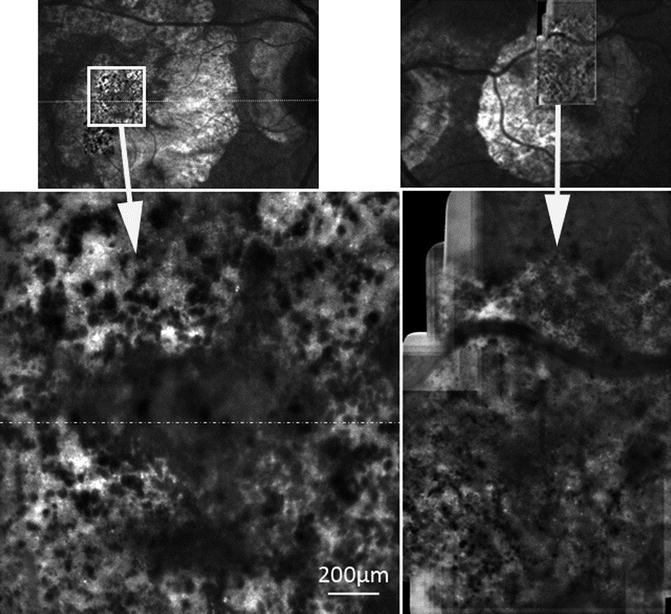

Fig. 7.3
Case of geographic atrophy. (a) Left, 488 nm autofluorescence image; (b) right, AO image of the area indicated in square. Note the better resolution of the limits of the GA area as well as the observation of a myriad of melanin clumps (bar, 250 μm)

Fig. 7.4
GA case with foveal sparing. Note in the AO image the clear delineation of the area of preserved fovea
We also investigated by time-lapse imaging the kinetics of the progression of atrophy and of pigment redistribution. Successive AO images taken several weeks apart were registered, which allowed the observation of the emergence and progression of atrophic spots. Time-lapse observation also showed the dynamic aspect of the redistribution of melanin-loaded cells such dynamic changes were observed within as well as outside atrophic areas. While limited changes were noted in some cases, many showed a more complex figure, with appearance of new clumps, change in their size and/or shape, rearrangement of large melanin deposits or abrupt disappearance.
7.3.2 AO Imaging of Retinal Vessels
Arterial hypertension, diabetes, and age affect the structure of retinal vessels. An increase of the wall-to-lumen ratio (WLR) of small arteries is a hallmark of hypertensive microangiopathy and is predictive of end-organ damage such as stroke [3]. Using conventional fundus photographs, several large-scale epidemiological studies reported that arteriolar narrowing and the incidence of focal arteriolar narrowing (FAN) and arteriovenous nicking (AVN) correlates with past and incident arterial pressure and with end-organ damage such as coronary diseases [4, 5] and stroke [6]. The clinical evaluation of hypertensive retinopathy is however impaired by technical limitations, mostly because fundus photographs or fluorescein angiography do not enable visualizing the arteriolar wall.
By AO NIR imaging, a linear structure is visible along both sides of the blood column of arteries (Fig. 7.5). In order to extract morphometric parameters from these images, we designed a software to automatically segment parietal structures (only images taken in diastole were considered for such analysis in order to minimize motion blur). The ratio of total parietal thickness over the lumen diameter (D) defined the WLR. This parameter was measured in the superotemporal artery in a cohort of 49 treatment-naïve patients [7]. In multivariate analysis taking into account age, gender, lumen diameter, body mass index, and systolic, diastolic, mean, and pulse blood pressure, only mean blood pressure and lumen diameter were independently correlated to WLR, which is in agreement with the concept that a myogenic reflex accounts for parietal thickening [8].
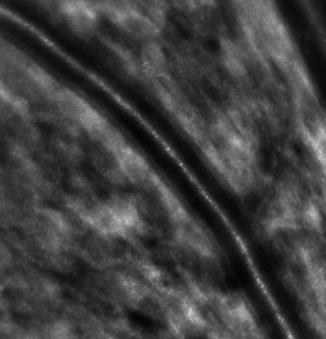

Fig. 7.5
AO imaging of a retinal artery showing the vessel wall
While it is commonly assumed that diffuse parietal thickening is initiated by pressure-driven myogenic response, the pathogenesis of focal changes remains debated. By AO, FANs and AVNs showed distinct anatomical features. At sites of FANs, the inner and outer limits of the arteriolar wall maintained their parallelism, and there was no evidence of parietal growth, suggesting that FANs were caused by focal vasoconstriction. In the microenvironment of AVNs, AO revealed a combination of loss of retinal transparency (Fig. 7.6) and presence of focal venous narrowings upstream and downstream of the arteriovenous crossing. The latter findings are in accordance with histology reports, which emphasized the role of the periarteriolar retina in the venous damage [9–11].




