Purpose
To summarize the current concepts and recent literature regarding the epidemiology, pathogenesis, imaging modalities, and treatment of acute hydrops in keratoconus.
Design
Perspective.
Methods
Review and synthesis of selected literature, with interpretation and perspective.
Results
Acute corneal hydrops is an incompletely understood complication of keratoconus, characterized by marked corneal edema caused by a break in Descemet membrane, allowing aqueous to enter the corneal stroma and epithelium. Although usually self-limiting, with clinical signs of edema typically resolving after 3 months, it often leaves a vision-impairing scar, necessitating and expediting the need for corneal transplantation. Studies have identified risk factors for developing acute hydrops. Modern imaging modalities such as ultrasound biomicroscopy, anterior segment optical coherence tomography, and in vivo confocal microscopy have enlightened us to the microstructural changes that take place during acute hydrops, the factors that influence its duration, and sequelae. Newer treatment regimens have seen a reduction in the duration of corneal edema during acute hydrops, and have improved the survival of corneal grafts after transplantation for resolved hydrops.
Conclusions
Effective management of acute corneal hydrops in keratoconus is based on recognizing and addressing the risk factors, treating the acute event effectively and promptly to reduce the duration of edema and its complications, and, ultimately, successful corneal transplantation with acceptable long-term graft survival rates. Improved in vivo imaging of the cornea during acute hydrops has led to an enhanced understanding of the pathogenesis and ultrastructural changes of the condition, and in turn has resulted in improved management of the disease.
Keratoconus is an ectatic corneal disorder, classically described as progressive, noninflammatory, and characterized by central corneal thinning, protrusion, and irregular myopic astigmatism. Although it was originally described in detail in 1854 by Dr John Nottingham, the substantial portion of our understanding of the condition has only been acquired in the last 50 years. Nonetheless, the earliest report of corneal edema related to keratoconus dates to a description by Terrien in 1906 ; however, the specific term “acute corneal hydrops” (hydrops corneae) was only popularized in 1940 following animal modeling and detailed clinical descriptions of focal disruption of Descemet membrane. Acute corneal hydrops remains an incompletely understood complication of keratoconus. It can also occur in other corneal ectasias, reportedly with higher frequency in pellucid marginal corneal degeneration (PMCD) (6%-11%) and keratoglobus (11%). Additionally it has been reported as a very rare late complication following penetrating keratoplasty for keratoconus.
Pathogenesis
Acute hydrops is the development of marked corneal edema caused by a break in Descemet membrane and endothelium, allowing aqueous to enter the corneal stroma and epithelium ( Figure 1 ). The elasticity of Descemet membrane means it retracts or coils when it breaks under tension. Basu and associates postulated that exact reapproximation of the displaced margins, either spontaneously or with C 3 F 8 , is not possible and resolution of corneal hydrops probably involves 2 steps. Firstly, the detached Descemet membrane has to reattach to the posterior stroma; the time for this depends on the depth of the Descemet membrane detachment. Secondly, the endothelium has to migrate over the gap between the 2 broken edges of Descemet membrane; the interval for this depends on the dimensions of the Descemet membrane break. Thus, insertion of C 3 F 8 can hasten the first step but not the second.
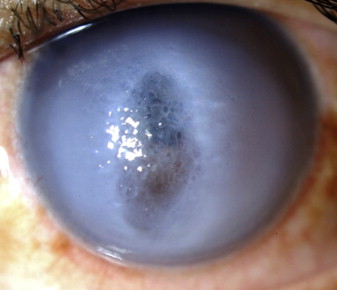
Although acute hydrops is usually self-limiting and clinical signs of edema typically resolve after 2-4 months, it often leaves a vision-impairing scar ( Figure 2 ), necessitating and expediting the need for corneal transplantation. In some cases, corneal neovascularization may occur ( Figure 2 , arrow), and this has significant implications for the patient’s future management and prognosis. Previous studies suggest that edema near the limbus and intrastromal cleft formation in cases of acute corneal hydrops may be considered risk factors for stromal neovascularization. Any associated inflammatory response, which may be greater in patients with atopy, has also been suggested as a potential stimulus to neovascularization.
Epidemiology and Risk Factors
The prevalence of keratoconus has been reported to vary in different studies internationally, from 8.8-54.4 per 100 000. Corneal hydrops is relatively uncommon and is estimated to occur in 2.6%–2.8% of patients with keratoconus. Interestingly, the mean age of onset of corneal hydrops is similar across studies, typically around 25 years of age, with a male preponderance.
Although an ethnic variation in prevalence of keratoconus is well established, few studies have specifically identified ethnic associations with the development of acute hydrops in keratoconus. In a recent study from New Zealand, where keratoconus is the most common indication for corneal transplantation, the authors noted that Pacific ethnicity is strongly associated with the development of acute hydrops, whereas New Zealand European ethnicity is negatively associated with hydrops development.
Although other risk factors have been identified in several studies, owing to the relatively small numbers of subjects in most studies it is difficult to isolate the risk factors of hydrops from those risk factors for keratoconus per se. However, 2 retrospective reviews attempted to determine the clinical factors associated with the development of acute hydrops, and each study included over 100 patients. Tuft and associates identified earlier age of diagnosis, steeper keratometry, and poorer Snellen visual acuity at the time of diagnosis of keratoconus to be strongly associated with subsequent development of corneal hydrops. Corneal hydrops also developed at a greater rate in eyes with severe allergic eye disease. Fan Gaskin and associates also identified poorer visual acuity at first presentation to the tertiary referral center for keratoconus to be associated with subsequent hydrops development. A history of eye rubbing was also associated with hydrops development—independent of a history of atopy and contact lens wear (which were not associated with greater likelihood of hydrops). Somewhat paradoxically, subjects who develop hydrops were found to be less likely to have a family history of keratoconus in this study.
Epidemiology and Risk Factors
The prevalence of keratoconus has been reported to vary in different studies internationally, from 8.8-54.4 per 100 000. Corneal hydrops is relatively uncommon and is estimated to occur in 2.6%–2.8% of patients with keratoconus. Interestingly, the mean age of onset of corneal hydrops is similar across studies, typically around 25 years of age, with a male preponderance.
Although an ethnic variation in prevalence of keratoconus is well established, few studies have specifically identified ethnic associations with the development of acute hydrops in keratoconus. In a recent study from New Zealand, where keratoconus is the most common indication for corneal transplantation, the authors noted that Pacific ethnicity is strongly associated with the development of acute hydrops, whereas New Zealand European ethnicity is negatively associated with hydrops development.
Although other risk factors have been identified in several studies, owing to the relatively small numbers of subjects in most studies it is difficult to isolate the risk factors of hydrops from those risk factors for keratoconus per se. However, 2 retrospective reviews attempted to determine the clinical factors associated with the development of acute hydrops, and each study included over 100 patients. Tuft and associates identified earlier age of diagnosis, steeper keratometry, and poorer Snellen visual acuity at the time of diagnosis of keratoconus to be strongly associated with subsequent development of corneal hydrops. Corneal hydrops also developed at a greater rate in eyes with severe allergic eye disease. Fan Gaskin and associates also identified poorer visual acuity at first presentation to the tertiary referral center for keratoconus to be associated with subsequent hydrops development. A history of eye rubbing was also associated with hydrops development—independent of a history of atopy and contact lens wear (which were not associated with greater likelihood of hydrops). Somewhat paradoxically, subjects who develop hydrops were found to be less likely to have a family history of keratoconus in this study.
Natural History of Corneal Hydrops
After the rupture of Descemet membrane, it may retract and curl anteriorly to form scrolls, ridges, or strands around attached fragments of stroma. This is thought to be the reason why acute hydrops takes longer to resolve than localized corneal edema caused by a breach of Descemet membrane during cataract surgery on a keratoconic eye.
The onset of acute hydrops is usually heralded by marked epiphora, followed by intense photophobia and pain, associated with markedly reduced visual acuity. Most cases of acute corneal hydrops resolve spontaneously over 2-4 months as the adjacent endothelial cells enlarge and migrate to cover the defect. Secondary flattening of the cornea may facilitate improved contact lens fitting, but central corneal scarring typically mandates corneal transplantation to restore visual function.
Unsurprisingly, greater area of corneal involvement by hydrops corresponds to a longer duration for the edema to resolve, increased risk of neovascularization, and ultimately a poorer visual outcome. Other complications of acute hydrops include infection, pseudocyst formation, malignant glaucoma, and corneal perforation. A history of hydrops may also predispose patients to greater likelihood of episodes of endothelial graft rejection after penetrating keratoplasty.
Imaging of Corneal Hydrops
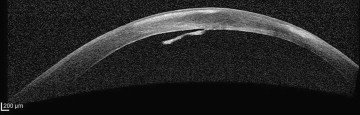
Traditional modalities of anterior segment imaging in corneal ectasia can be problematic in acute hydrops owing to the difficulty of imaging through an edematous cornea. The advent of ultrasound biomicroscopy (UBM), anterior segment optical coherence tomography (AS-OCT) ( Figure 3 ), and in vivo confocal microscopy (IVCM) ( Figure 4 ) has revealed some of the ultrastructural changes that occur during acute hydrops in vivo. Previously such ultrastructural changes could only be extrapolated from ex vivo observations on “resolved” tissue—obtained following corneal transplantation. These new technologies have also improved our ability to predict duration of edema and likelihood of neovascularization and to monitor response to therapy.
An ultrasound biomicroscopy study has confirmed that intrastromal edema is directly related to rupture of Descemet membrane. Direct visualization of Descemet tear revealed areas of deficiency under the location of maximum stromal edema. In the same study, following intracameral C 3 F 8 at week zero, ultrasound biomicroscopy demonstrated a complete unrolling and reapposition of Descemet membrane to the corneal stroma by 3 weeks, with complete resolution of edema in 92.3% of cases by 6 weeks.
In a similar study, Nakagawa and associates assessed a series of 13 consecutive hydrops corneas with ultrasound biomicroscopy and discovered that not only was rupture of Descemet membrane identified in all eyes, but all eyes also exhibited intrastromal clefts. In 11 of 13 eyes the clefts were connected with the anterior chamber on the ultrasound biomicroscopy images. The authors hypothesized that severe corneal edema results from the presence of intrastromal clefts by increasing the surface area exposed to the anterior chamber. The gap between Descemet membrane and the stroma may also delay the closure of Descemet membrane and ultimately the resolution of corneal edema.
Basu and associates examined 24 eyes with acute hydrops with anterior segment optical coherence tomography and published a retrospective study assessing the serial observations throughout the duration of hydrops. Interestingly, they identified 3 patterns of Descemet membrane appearance during acute hydrops on anterior segment optical coherence tomography: detachment with break and rolled ends, detachment with break and flat ends, and detachment with no break. However, they acknowledged that because anterior segment optical coherence tomography scans are taken at 45-degree intervals, a small planar break could have been missed, as detachment without break would question our present understanding of the mechanism of hydrops development. This study revealed that, without intervention, not only is the duration of edema affected by the size of Descemet membrane break, but also the depth of the Descemet membrane detachment. The third factor to influence duration of clinical edema is intervention with intracameral C 3 F 8 (see next section).
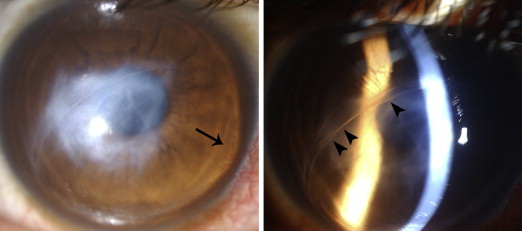
In vivo confocal microscopy is a technology that has revolutionized imaging of the cornea in recent years. Lockington and associates conducted a prospective study of acute hydrops in keratoconus as assessed by in vivo confocal microscopy. This study revealed the presence of presumed inflammatory cells, in 4 of the 10 eyes studied, and postulated these may be associated with neovascularization. These cells were hyper-reflective, with round cell bodies ( Figure 4 , Left) present in the epithelium and anterior to mid-stroma. In 2 eyes, these cells persisted throughout the duration of hydrops, beyond clinical resolution. In these 2 cases, other unique cellular structures were also identified: elongating cells with small cell bodies were noted in the anterior stroma at 2-3 months ( Figure 4 , Middle) and at 3 months after presentation, both of these corneas also exhibited unusual stromal cells with large, round, speckled cell bodies and elongated cells with branching cell processes ( Figure 4 , Right). Both corneas developed stromal neovascularization and were the only corneas to do so during the study.
Contemporary imaging modalities have enabled significantly greater insight into the pathophysiology of corneal hydrops. However, in a clinical setting, UBM and IVCM require more technical skills and operator experience. Notably, although the latter has increased our understanding of hydrops at the microstructural level, currently IVCM has a minimal role in management. In contrast, AS-OCT is easier to capture than UBM or IVCM and therefore might more readily assist in ascertaining whether certain interventions may be beneficial (see next section).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


