Purpose
To evaluate the ability of novel dyes to stain lens capsule (LC), internal limiting membrane (ILM), epiretinal membrane (ERM), and vitreous.
Design
Experimental study in animal and human donor eyes.
Methods
Thirteen dyes, methyl violet, crystal violet, eosin Y, sudan black B, methylene blue, toluidine blue, light green, indigo carmine, fast green, congo red, evans blue, brilliant blue, and bromophenol blue, were injected onto the LC and ILM of enucleated porcine eyes. The vitreous was stained with 2 mL of dyes for 1 minute. Six dyes (indigo carmine, evans blue, fast green, light green, bromophenol blue, and brilliant blue) were selected for experiments in human donor eyes and freshly removed ERM.
Results
In the porcine eyes, ILM staining with methylene blue, toluidine blue, indigo carmine, evans blue, bromophenol blue, and fast green was moderate, and methyl violet, crystal violet, brilliant blue, or sudan black resulted in strong staining. Methyl violet, crystal violet, sudan black, toluidine blue, and methylene blue caused histologic damage in porcine retinas. Vitreous examination revealed moderate staining with congo red, crystal violet, fast green, eosin Y, methylene blue, toluidine blue, brilliant blue, bromophenol blue, and methyl violet and strong staining with light green and evans blue. ERMs showed strong staining with 0.5% evans blue and moderate staining with 0.5% light green, fast green, brilliant blue, and bromophenol blue. Evaluation of donor eyes disclosed moderate staining with evans blue, light green, and bromophenol blue and strong staining with 0.5% brilliant blue. Moderate or strong staining of the vitreous occurred with most dyes. LC evaluation showed moderate staining with 0.5% evans blue, fast green, and brilliant blue, whereas 0.5% light green produced strong LC staining.
Conclusions
Brilliant blue shows the best ILM staining, whereas bromophenol blue, evans blue, and light green also stain ILM. Most dyes bind well to LC, vitreous, and ERM.
Intraoperative application of vital dyes for the visualization of intraocular membranes and tissues has facilitated surgical techniques and outcomes in recent years. For vitreoretinal surgery, indocyanine green (ICG) dye initially was introduced for staining the internal limiting membrane (ILM). This was followed by many publications reporting signs of retinal toxicity after intravitreous ICG injection. In cataract surgery, trypan blue has provided since the 1990s a better visualization of the anterior lens capsule (LC) when the red reflex is not possible. Following ICG and trypan blue, other dyes such as brilliant blue and patent blue were presented as newer alternative vital dyes for ocular surgery. However, concerns also were raised about the safety of brilliant blue, trypan blue, and patent blue, and their selective affinity for some intraocular membranes such as epiretinal membranes (ERMs), LC, or ILM, but not for all of them.
For this study, a total of 13 vital dyes were selected: light green, fast green, methyl violet, crystal violet, congo red, eosin Y, sudan black B, evans blue, brilliant blue, bromophenol blue, methylene blue, toluidine blue, and indigo carmine. In addition, ICG, trypan blue, and patent blue were included in our in vivo examinations for comparison. The goal of this study was to investigate the staining ability of these dyes regarding the retinal ILM, vitreous, ERM, and LC in a series of systematic experiments in freshly enucleated porcine eyes, freshly removed ERM, and enucleated human donor eyes.
Methods
Selection of Dyes and Preparation
A total of 50 mg of dye powder of light green, fast green, bromophenol blue, brilliant blue, patent blue, methyl violet, crystal violet, congo red, eosin Y, sudan black, evans blue, methylene blue, toluidine blue, indigo carmine, ICG, or trypan blue (Merck, Darmstadt, Germany, for all dyes, except for Sigma-Aldrich, Munich, Germany, in the case of bromophenol blue and Ophthalmos Ind, São Paulo, Brazil, for ICG and trypan blue) was weighed with an analytic balance (Mettler-Toledo, Inc, Columbus, Ohio, USA) and dissolved in 10 mL balanced salt solution (BSS plus; Alcon Laboratories, Inc, Fort Worth, Texas, USA) to obtain a concentration of 0.5% for the initial experiments. The mixtures were shaken for 5 minutes and sonicated (Unique Ind, Idaiatuba, Brazil) to obtain a homogeneous solution. For donor eye experiments, the selected dyes light green, ICG, trypan blue, patent blue, fast green, indigo carmine, evans blue, brilliant blue, and bromophenol blue were prepared as 0.5% and 0.05% solutions in balanced salt solution. The choice of agents was based on a rationale to validate the experimental method; thiazine, carbonyl, diazo, aminoarylmethane, and hydroxyxanthene dyes were analyzed to search for an optimal staining agent.
Staining of Porcine Internal Limiting Membrane, Vitreous, and Lens Capsule
All animal experiments were conducted according to the ARVO Statement for the Use of Animals in Ophthalmic and Vision Research. The experiments for ILM, LC, and vitreous staining were conducted on an ex vivo setup. Ninety-six pig eyes were obtained from a slaughterhouse and were prepared within 6 hours after death. The dyes initially were used to stain the LC of pigs. Immediately after eye removal, the eyes were placed on a glass support, and a 360-degree limbal incision was performed for corneal removal, followed by total iris removal. Next, 0.05 mL of 2 concentrations of 0.05% and 0.5% of each of the 16 dyes were placed onto the LC for 1 minute. Afterward, the anterior surface of the lens was irrigated carefully with saline solution. An anterior curvilinear continuous capsulorrhexis was performed within the stained capsule using forceps.
For evaluation of ILM and vitreous staining, the remaining eye cups had the anterior segment removed with a 3-mm incision behind the limbus, and eyes with funduscopic red reflex were included in the experiments. The vitreous was gently extracted completely with cotton buds and forceps and was placed in a glass vial for its evaluation. Next, 0.05 mL of 0.5% of the 16 dyes was injected slowly over the retinal surface to avoid dispersion. The dye was left on the retina for 2 minutes and the retinal surface was rinsed twice with 2 mL saline. Each experiment was repeated with 4 samples with each dye. In all eyes, peeling of the ILM was attempted with a bent 27-gauge needle.
To evaluate vitreous staining, the vitreous removed from fresh enucleated pig eyes was placed in a 5-mL vial for 1 minute with 2 mL of each of the 13 dyes at a concentration of 0.5%. The vitreous then was removed, rinsed thoroughly, and placed on a white background for staining evaluation under the microscope. Photographs of all experiments were obtained with a Canon camera (Canon PowerShot A2000 IS; Canon Electronics, Saitama, Japan) fitted to the operating microscope. The intensity of ILM, LC, and vitreous staining was graded in a masked fashion as no staining (0), faint staining (+), moderate staining (++), or strong staining (+++).
Histologic Evaluation of Porcine Eyes by Light Microscopy
LCs were placed in 10% buffered formaldehyde and 4% buffered glutaraldehyde (Karnowsky fixative) for a minimum of 48 hours. Specimens of LC then were evaluated macroscopically by light microscopy after preparation with routine hematoxylin and eosin staining.
Immediately after ILM removal, some specimens of both ILM and peeled retinas of porcine eyes were placed in a mixture of 4% buffered formaldehyde and 4% buffered glutaraldehyde (Karnowsky fixative) for 15 to 20 minutes to achieve rapid and proper fixation of the retina followed by storage in 10% buffered formaldehyde for 48 hours. Gross examinations of the tissues were performed. Semithin sections were cut along a superior–inferior plane, and they then were embedded in paraffin. Next, the retinas were sectioned at a thickness of 6 μm, stained using routine hematoxylin and eosin and examined by light microscopy. In screening for the most suitable dye for experiments in donor eyes, each dye was considered to be toxic to the retina if any of the following findings were observed: major signs of disorganization, cell loss, vacuolization, condensation, edema or swelling; necrosis of at least one retinal layer; or fragments, debris, as well as pieces of retinal cell elements within the removed ILMs.
Staining of Freshly Excised Human Epiretinal Membrane
This study was approved by local ethical committee and informed consent was obtained from each patient before surgery. Staining pattern of human ERMs was examined after exposure to 0.5% of the 16 dyes included in this study. The vital dyes (0.3 mL) were applied onto freshly surgically removed ERMs obtained from patients with proliferative diabetic retinopathy and proliferative vitreoretinopathy of rhegmatogenous retinal detachment. The staining intensity of ERMs was graded as for other membranes in as masked fashion. The stained ERM tissues also were fixed with 10% buffered formaldehyde followed by hematoxylin and eosin preparation for light microscopy to detect vitreous or ILM remnants and the composition of the tissue.
Staining of Lens Capsule, Vitreous, and Internal Limiting Membrane in Human Donor Eyes
Human donor eyes were obtained from the Eye Bank of the Federal University of São Paulo after institutional review board approval. Six dyes were chosen for this setup based on their safety and staining profile on the porcine eyes: light green, fast green, brilliant blue, indigo carmine, bromophenol blue, and evans blue. In addition, analysis of staining in these donor eyes was conducted with ICG, trypan blue, and patent blue to compare the currently used dyes with the novel future dyes. Initially, human cadaveric eyes up to 48 hours after death were examined in the laboratory to determine their integrity. To stain the LC, a 360–degree-limbal incision was performed for corneal removal, followed by segmented iris removal. Next, 0.05 mL of 0.05% and 0.5% concentrations of each dye were applied to the LC for 1 minute, followed by rinsing with saline solution. An anterior curvilinear capsulorrhexis was carried out with forceps, and selectively removed membranes were sent for histologic analysis after buffered formaldehyde fixation and periodic acid–Schiff staining.
The entire anterior segment of the eye was then excised to gain access to the vitreous cavity. After open sky vitrectomy (Accurus; Alcon Laboratories), 0.2 mL of dye solution was injected into the posterior vitreous cavity over the macula for 2 minutes and then was removed by mechanical aspiration, trying to leave the stained ILM clearly visible. A bent 27-gauge needle was used to create the initial ILM tear, followed by grasping with a 23-gauge forceps applied to peel the ILM.
Results
Selection of Dyes and Preparation
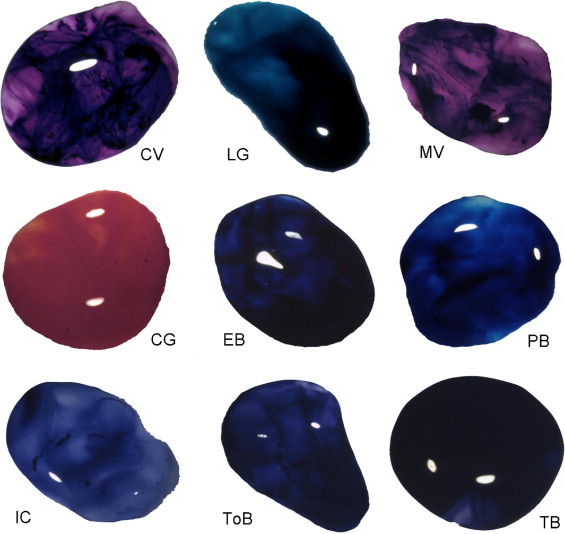
All dyes but ICG could be diluted successfully in balanced salt solution; ICG was diluted further in glucose 5% solution. Table 1 summarizes the chemical and staining properties of the vital dyes selected for investigation in this study. Four staining agents, evans blue, congo red, sudan black, and trypan blue, belong to the azo dyes, and 7 dyes, light green, crystal violet, fast green, methyl violet, patent blue, brilliant blue, and bromophenol blue, are in the group of the arylmethane agents. The mechanism of staining of the dyes varies and can involve binding, contrast, or reactivity, although some staining agents such as fast green and evans blue bind to living tissues through unknown mechanisms.
| Dye | Biochemical Properties and Principles of Biological Staining | Ex Vivo Staining in Porcince Eyes at 0.5% | Ex Vivo Histologic Results at 0.5% | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Chemical Group | Chemical Formula | Weight | Staining Principle | Lens Capsule Staining | ILM Staining | Color on the Retina | Vitreous Staining | Retina Histologic Damage | |
| Methylene blue | Thiazine | C 16 H 18 N 3 ClS | 373 | Absorptive | + | ++ | Bright blue | ++ | Toxic |
| Toluidine blue | Thiazine | C 15 H 16 N 3 SCl | 305 | Absorptive | ++ | ++ | Dark blue | ++ | Toxic |
| Indigo carmine a | Carbonyl | C 16 H 8 N 2 Na 2 O 8 S 2 | 466 | Contrast | + | ++ | Dark blue | + | Safe |
| Evans blue a | Anionic dis-azo | C 34 H 24 N 6 Na 4 O 14 S 4 | 960 | Unknown | ++ | ++ | Blue | +++ | Safe |
| Congo red | Anionic dis-azo | C 32 H 22 N 6 Na 2 O 6 S 2 | 696 | Contrast/reactive | + | + | Red | ++ | Safe |
| Light green Sf a | Anionic amino triarylmethane | C 37 H 34 N 2 Na 2 O 9 S 3 | 792 | Contrast | ++ | + | Light greenish | +++ | Safe |
| Eosin Y b | Acidic hydroxyxanthene | C 20 H 8 O 5 Br 4 | 648 | Unknown | + | 0 | Light reddish | ++ | Safe |
| Sudan black B | Anionic dis-azo | C 29 H 24 N 4 | 457 | Unknown | ++ | +++ | Black | + | Toxic |
| Crystal violet | Cationic amino arylmethane | C 25 H 30 N 3 | 372 | Contrast/reactive | ++ | +++ | Red violet | ++ | Toxic |
| Fast green a | Anionic amino arylmethane | C 37 H 34 N 2 Na 2 O 10 S 3 | 809 | Unknown | ++ | ++ | Dark green | ++ | Safe |
| Methyl violet | Cationic amino arylmethane | C 24 H 28 ClN 3 | 393 | Contrast | ++ | +++ | Red violet | ++ | Toxic |
| Patent blue | Anionic amino arylmethane | C 27 H 31 N 2 NaO 6 S 2 | 582 | Absorptive | + | + | Blue | ++ | Safe |
| Indocyanine green | Tricarbocyanine | C 43 H 47 N 2 NaO 6 S 2 | 774 | Unknown | ++ | +++ | Green | ++ | Safe |
| Trypan blue | Anionic dis-azo | C 34 H 24 N 6 Na 4 O 14 S 4 | 961 | Contrast | +++ | + | Dark-blue | +++ | Safe |
| Brilliant blue a | Triarylmethane | C 47 H 48 N 3 S 2 O 7 Na | 854 | Unknown | ++ | +++ | Violet blueish | ++ | Safe |
| Bromophenol blue a | Triarylmethane | C 19 H 10 Br 4 O 5 S | 670 | Reactive/contrast | ++ | ++ | Dark blue violet | ++ | Safe |
a Selected for further in vivo experiments in rabbits.
Staining of Porcine Internal Limiting Membrane, Vitreous, and Lens Capsule
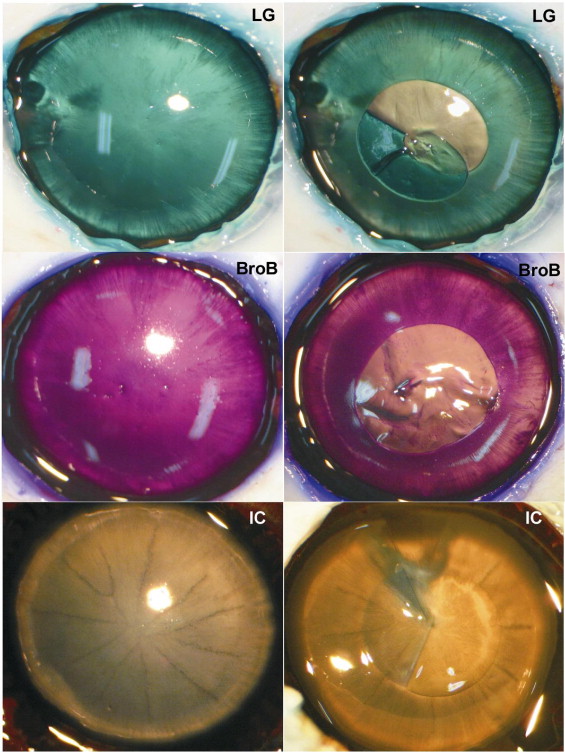
The grading of the staining effect of each dye in porcine eyes is shown in Table 1 . The effect in surgically stained LC varied for the different dyes and dye concentrations ( Figure 1 ). With the aid of most dyes, continuous capsulorrhexis was completed successfully, whereas adequate contrast and visibility was reported by the surgeon. Indigo carmine, congo red, brilliant blue, and methylene blue did not stain the LC sufficiently at a concentration of 0.05%; however, patent blue, trypan blue, evans blue, light green, sudan black, crystal violet, methyl violet, ICG, and bromophenol blue produced only light (+) staining of the LC, whereas fast green and trypan blue promoted moderate (++) staining. At the higher concentration of 0.5%, toluidine blue, evans blue, light green, sudan black, crystal violet, fast green, methyl violet, ICG, brilliant blue, and bromophenol blue provided moderate (++) and trypan blue provided strong (+++) staining of the LC ( Table 1 ).
Experiments with freshly enucleated porcine eyes with dyes at 0.5% demonstrated no (0) retinal ILM staining after eosin Y exposure, whereas congo red, light green, patent blue, and trypan blue generated only faint (+) retinal ILM staining. Exposure of the retinal ILM to methylene blue, toluidine blue, indigo carmine, evans blue, bromophenol blue, and fast green resulted in moderate (++) staining, and methyl violet, crystal violet, sudan black, brilliant blue, and ICG yielded strong (+++) retinal ILM staining in porcine eyes ( Figure 2 ). With most eyes, a continuous 1-piece ILM peeling was difficult to achieve, an expected finding in porcine eyes.
Most of the vital dyes used in this investigation at 0.5% stained the vitreous very well. Vitreous examination demonstrated moderate (++) staining with congo red, crystal violet, fast green, patent blue, eosin Y, methylene blue, toluidine blue, ICG, and methyl violet; demonstrated strong staining (+++) with trypan blue, light green, and evans blue; and demonstrated faint (+) staining with indigo carmine and sudan black ( Figure 3 ).
Histologic Evaluation by Light Microscopy
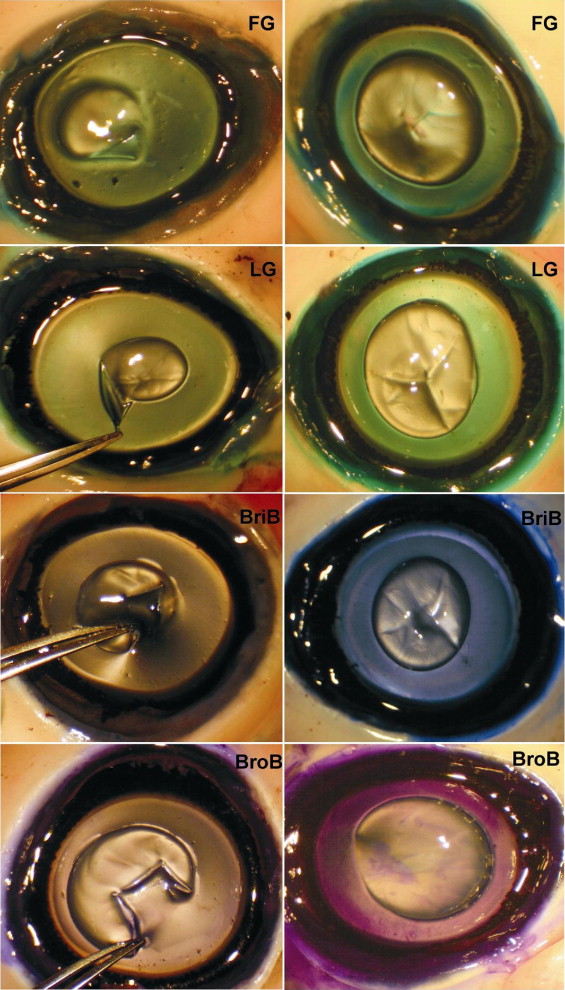
Light microscopy examination revealed the typical fibroblastic characteristics of the anterior LC. No anterior capsule or lens epithelium abnormality in the removed area was observed in any eyes except for surface-parallel intracapsular splits and delamination of the capsulorrhexis resulting from fixation artifact ( Figure 4 ).
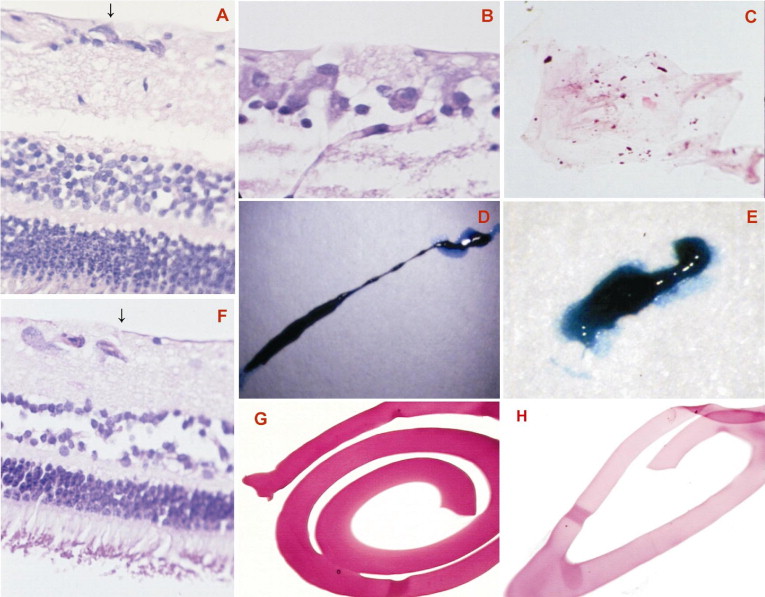
Gross examination of porcine retinal specimens showed no hemorrhage, inflammation, or detachment. In the cases of methyl violet, toluidine blue, and methylene blue staining, additional deeper layers of the retina also were removed during the ILM peeling maneuver, as cellular elements were present in the histopathologic examination ( Figure 4 ). In addition, the vital dyes crystal violet and sudan black at 0.5% were found to be toxic because they induced severe morphologic damage to the retina, such as producing regions of deformation and destruction of inner nuclear layers and outer nuclear layers, as well as focal areas of damage to photoreceptors. In contrast, eosin Y, evans blue, congo red, brilliant blue, bromophenol blue, indigo carmine, light green, patent blue, ICG, trypan blue, and fast green yielded no major morphologic alterations in porcine retinas, whereas the ILM could be peeled successfully. Histologic examination revealed that these dyes promoted no major toxicity changes in the inner and outer nuclear layer cells, nerve fiber layer, retinal pigment epithelium, and choriocapillaris. Histologic evaluation also demonstrated the successful separation of the ILM in cases of peeling with the aid of evans blue, light green, brilliant blue, bromophenol blue, patent blue, ICG, trypan blue, and fast green, without remnants of retinal cells ( Figure 4 ).
Staining of Freshly Excised Human Epiretinal Membrane
The results showed that most dyes possess at least some binding affinity to freshly enucleated human ERM. ERM examination revealed strong (+++) staining with evans blue and trypan blue; moderate (++) staining with 0.5% light green, fast green, brilliant blue, bromophenol blue, and patent blue; and faint (+) staining after exposure to indigo carmine and ICG ( Table 1 ). At the lower dose of 0.05%, evans blue, fast green, patent blue, ICG, brilliant blue, and bromophenol blue provided only faint (+) staining, and light green and trypan blue produced moderate ERM staining ( Figure 4 ). Histopathologic examination disclosed the highly cellular composition of the ERM with retinal pigment epithelium elements with some connective tissue components.
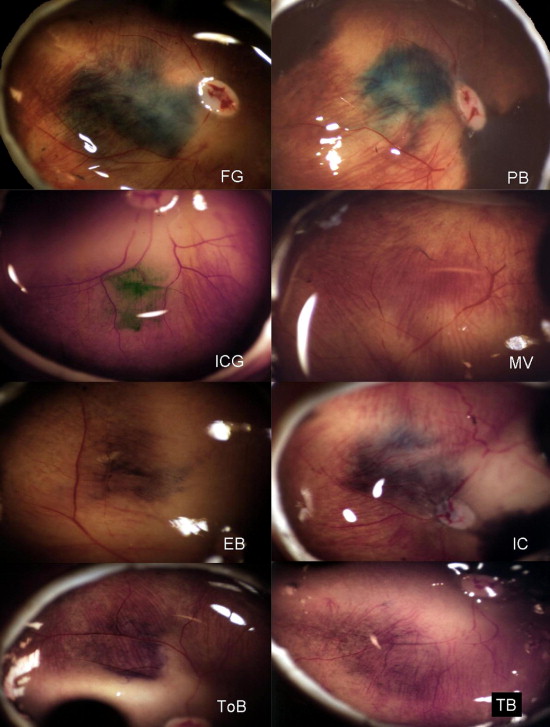
Staining of Lens Capsule, Vitreous, and Internal Limiting Membrane in Human Donor Eyes
Most of the dyes stained the LC homogenously, where its edge could be observed, and the circular capsulorrhexis could be performed in all cases. The concentration of 0.05% was enough to produce at least faint (+) staining with clear visualization with most dyes. Exceptionally, indigo carmine produced no staining, and no merit could be found in using this dye at this concentration. However, trypan blue enabled strong staining of the anterior LC. The staining was enhanced as the concentration of the dyes was increased to 0.5%, where mild (+) staining occurred with indigo carmine; moderate (++) staining occurred with evans blue, fast green, patent blue, ICG, and brilliant blue; and strong (+++) staining occurred with bromophenol blue, light green, and trypan blue ( Figure 5 ).