Purpose
To conduct a cost-utility analysis and determine the cost-effectiveness of the Boston Keratoprosthesis (Boston Kpro).
Design
Retrospective cohort study.
Methods
setting: The Massachusetts Eye and Ear Infirmary corneal service. patients: Inclusion required a minimum 2-year follow-up. Patients with autoimmune diseases and chemical burns were excluded. Eighty-two patients were included with various indications for surgery. intervention: The keratoprosthesis is a collar button–shaped polymethylmethacrylate (PMMA) device consisting of 2 curved plates sandwiched around a corneal donor (allo)graft. The device is assembled intraoperatively and sutured to a patient’s eye after removing the diseased cornea. main outcome measures: Average cost-effectiveness of the keratoprosthesis was determined by cost-utility analysis, using expected-value calculations and time-tradeoff utilities. The comparative effectiveness, or gain in quality-adjusted life years (QALYs), was also sought. Cost-effectiveness was compared to recently published data on penetrating keratoplasty (PK).
Results
A total discounted incremental QALY gain for the Boston Kpro of 0.763 correlated with a conferred QALY gain of 20.3% for the average patient. The average cost-effectiveness of the keratoprosthesis was $16 140 per QALY.
Conclusions
Comparable to corneal transplantation, with a cost-effectiveness between $12 000 and $16 000 per QALY, the keratoprosthesis can be considered highly cost-effective.
The global burden of visual impairment is substantial. In a report published by the World Health Organization in 2009, there were an estimated 314 million (about 4.6% of the world population) visually impaired people in the world, of whom 45 million were considered bilaterally blind (visual acuity worse than 10/180). Approximately 8 million are bilaterally blind because of corneal disease, including 1.5 million children. In many parts of Africa, nearly 60% of blindness is caused by corneal etiologies, primarily cataract and trachoma. In the United States, 3.7 million are either visually impaired or blind with greatly disparate corneal etiologies, such as trauma, infection/ulceration attributable to chronic contact lens wear, Herpes simplex virus, dystrophies, autoimmune diseases, and graft failure. The annual cost of adult vision problems in the United States is estimated at $51.4 billion.
In 1993, the World Bank Report entitled Investing in Health described cataract surgery as a highly cost-effective intervention. A study assessing the global cost-effectiveness of cataract surgery in 2007 confirmed this worldwide. Penetrating keratoplasty, although comparatively more expensive, was also found to be cost-effective in the United States in patients with bilaterally poor vision. The general term cost-effective requires defining, as it can include several disparate analyses. The first is cost-minimization analysis . Typically, such an analysis compares 2 interventions of comparable effectiveness to determine the less costly. In contrast, a cost-benefit analysis compares the costs of an intervention against the money saved as a result and is widely used in healthcare policy decisions. In both of these examples, however, quality-of-life issues are ignored. The cost-effectiveness analysis measures the output (benefit) on a different scale, rather than in monetary terms. Output units are usually described in terms of life-years gained or saved, for example, quality-adjusted life years (QALYs) gained. In ophthalmology, vision years can be substituted for life years. A cost-effectiveness analysis that accounts for the perceived value of an intervention or health state is termed a cost-utility analysis (CUA).
To calculate utility, a scale from 0 to 1 is used. Normally, 0 represents death and 1 represents perfect health. In ophthalmology, for example, perfect (20/20) vision in the better seeing eye has been given a utility value of 0.92 while no light perception (NLP) vision is assigned a utility value of 0.26. Several methods have been used to determine utility values, the most notable being the standard-gamble and time-tradeoff methods. The former has long been considered the gold standard. It has, nevertheless, been perceived as cognitively demanding and overestimating risk aversion. Moreover, it has been shown that as visual acuity in the better-seeing eye decreases, the corresponding time-tradeoff utility value, and not the standard-gamble utility value, decreases at every visual stratification level. This correlation assumes vision in the worse eye is 20/40 or less. In the time-tradeoff approach, an individual is asked how many years she is expected to live, followed by how many years she would be willing to trade in return for perfect vision. Thus, if expected to live another 20 years and willing to trade 5 of them for perfect vision, the utility value for her current visual acuity would be calculated as 1.0 − (5/20) = 0.75.
To our knowledge, no cost-utility analysis has been performed on the use of the Boston Keratoprosthesis. It is the intention of this study to objectively assess 1) the comparative effectiveness (gain in QALYs) and 2) the average cost-effectiveness (compared to no further treatment or current visual state) of the Boston Keratoprosthesis procedure for the treatment of severe corneal disease and blindness.
Methods
The Boston Keratoprosthesis Type I
The Boston Keratoprosthesis (Kpro) was developed at the Massachusetts Eye & Ear Infirmary over the last 40 years. It is a collar button–shaped polymethylmethacrylate (PMMA) device consisting of 2 curved plates sandwiched around a corneal autograft or allograft. A soft bandage contact lens is used indefinitely after surgery to prevent evaporative losses and thus minimize the risk of corneal melts. For severe dry eye and cicatricial diseases, a “Type II” Boston Keratoprosthesis can be implanted through the eyelids. Implantation of either the type I or type II device is performed in 1 stage, and is technically similar to standard corneal transplantation. The Boston Keratoprosthesis is by far the most commonly used prosthetic cornea in the world: more than 3000 devices had been implanted worldwide by December 2008 (Boston Keratoprosthesis Update Newsletter V:2008).
Patients
In a retrospective cohort study chart review, patients who underwent keratoprosthesis surgery at the Massachusetts Eye and Ear Infirmary between the years 2003 and 2008 were identified. Patients had to have a minimum of 2 years of visual acuity follow-up data recorded. Because patients with autoimmune diseases and chemical burns are known to be poor prognostic groups for keratoprosthesis surgery, they were excluded from this study. Eighty-two patients were included.
Patient characteristics with underlying diagnoses prior to keratoprostheis surgery are given in Table 1 . Infectious causes included: fungi, 3 (3.6%); idiopathic but presumed microbial ulceration, 3 (3.6%); Pseudomonas , 1 (1.2%); Herpes simplex virus, 2 (2.4%); and Varicella zoster virus, 1 (1.2%). A complete ophthalmic exam was performed prior to Kpro surgery. Median preoperative best-corrected visual acuity (BCVA) in the treated eye was logMAR 2.12 ± 0.38 (Snellen equivalent Hand Motions). Visual acuity values were normally distributed per Shapiro-Wilk testing. The IRB/Ethics Committee decided approval was not required for this study. The project does not fall under HIPAA requirements because no protected health information is recorded or linked by code to data.
| Characteristic | Amount | Qualifier |
|---|---|---|
| Patients | n = 82 | |
| Gender | Male = 45 | |
| Female = 37 | ||
| Age | Mean = 65 years | Range = 23–97 years |
| Diagnostic group: | ||
| Penetrating keratoplasty graft failure | n = 59 | 71.1% |
| Infectious | n = 10 | 12.0% |
| Trauma | n = 4 | 4.8% |
| Primary (high risk for graft failure) | n = 3 | 3.6% |
| Oral Surg Oral Med Oral Pathol neovascularization | n = 2 | 2.4% |
| Pseudophakic bullous keratopathy | n = 1 | 1.2% |
| Salzman’s degeneration | n = 1 | 1.2% |
| Silicone oil keratopathy | n = 1 | 1.2% |
| Limbal stem cell deficiency | n = 1 | 1.2% |
| Aniridia | n = 1 | 1.2% |
Decision Analysis
In order to make cost-utility calculations, several model assumptions were made. Average cost-effectiveness was determined, comparing keratoprosthesis surgery to no further intervention (the current visual state). The typical risks of keratoprosthesis failure include infection, endophthalmitis, extrusion, and retinal detachment. Complications and additional procedures were incorporated into a weighted cost decision tree. A list of the most common postoperative complications and treatments is given in Table 2 .
| Complication/Procedure | Number of Patients | Number of Treatments | Percent of Total Cohort (Based on Treatment) |
|---|---|---|---|
| No complications after 24 months | 35 | n/a | |
| Retroprosthetic membranes | 26 | 42 YAGS | 51.22 |
| 5 membranectomy | 6.10 | ||
| Inflammation | 14 | 13 Kenalog | 16.05 |
| 1 pars plana vitrectomy | 1.22 | ||
| 5 IV solumedrol | 6.10 | ||
| High IOP | 13 | 13 shunts | 16.05 |
| 2 pars plana vitrectomy | 2.44 | ||
| 4 revised shunts | 4.88 | ||
| PCO | 7 | 7 YAG | 8.54 |
| BCL early replacement | 4 | 4 replacements | 4.88 |
| CME | 4 | 4 Kenalog | 4.88 |
| Tarsorrhaphy | 3 | 3 surgery | 3.66 |
| Ptosis repair | 2 | 2 surgery | 2.44 |
| Retinal detachment | 2 | 1 silicone oil | 1.22 |
| 1 pars plana vitrectomy | 1.22 | ||
| Iris prolapse | 1 | 1 surgery | 1.22 |
| Strabismus surgery | 1 | 1 surgery | 1.22 |
| Kpro replacement | 1 | 1 replacement | 1.22 |
| Total optic cupping | 1 | n/a |
In our sample, 2-year anatomical retention was 99% and 5-year anatomical retention in a sub-sample (n = 27) was 89%. Based on available literature, the multicenter retention rate at 8.5 months is purported to be 95%. In a separate sample, 2-year retention rate decreased to 61%, but this figure included patients with autoimmune diseases, chemical injury, and severe glaucoma. Furthermore, unpublished multicenter data report 2-year retention in 350 non-autoimmune patients to be as high as 94% (Ament J, Todani A, Ciolino J, Belin M. Multicenter KPro retention data. KPro Study Group, Albany, New York. Ongoing study.). Because of the variability in reported retention rates the following model assumptions were made: 1) 2-year anatomical retention was set at 94%, a value lower than reported in this sample but more representative of the larger multicenter data; 2) since 5-year survival data exist for the most updated version of the Boston Keratoprosthesis, the time frame chosen for the CUA was 5 years; 3) by French Curve extrapolation, we conservatively estimated 85% anatomical retention for our entire sample at 5 years; 4) a yearly transplant survival probability of 97% was interpolated from the extrapolation curve; 5) since all patients had a minimum 2-year follow-up, comparing best spectacle-corrected visual acuity (BSCVA) preoperatively and at 2 years was utilized to calculate incremental utilities; 6) in patients without 5-year follow-up, BSCVA was assumed to be stable throughout the remaining study period; 7) we assumed surgery in only 1 eye and the outcome was ignored in cases where the keratoprosthesis had been implanted in the second eye; 8) since the mean patient age was 65, we postulated that all patients would survive the 5 study years; and 9) follow-up for all patients was assumed to be similar.
Utility Assessment
As described, the time-tradeoff method was used for patient-based utility assessment in the CUA. For each patient, the appropriate utility value based on past studies was matched to the corresponding BCVA both preoperatively and 2 years postoperatively. The mean (±standard deviation) preoperative utility value was 0.449 (±0.113), increasing to 0.630 (±0.174) postoperatively. For each patient, the difference between preoperative and postoperative utility was calculated and the mean of these differences obtained. Decreases in vision were accounted for by incorporating negative utilities into the mean incremental utility calculation. The mean incremental utility in our study population was 0.182. The total QALY gain (comparative effectiveness) was also determined by multiplying the years of utility gain by years of benefit duration and comparing it to the preoperative utility (quality of life) state.
Identification of Costs
The costs for the Kpro, surgical procedure, and hospitalization were obtained from the Massachusetts Eye and Ear Infirmary CPT diagnosis and procedure codes. The costs associated with postoperative care were obtained from clinic billing sheets with corresponding CPT codes. Costs were initially obtained based on these codes; however, the CUA was conducted based on the current Medicare reimbursement rates in 2009. Reimbursement for keratoplasty done at the time of keratoprosthesis insertion has typically been at 50% of the allowable. There are special circumstances where the patient’s own cornea can be harvested for keratoprosthesis implantation to mitigate costs, but this is rarely done. Ophthalmologic visits were conducted every 3 months for the first year, every 6 months over the second year, and then annually. Maintenance costs included bandage contact lenses, medications, and patient travel. An overview of all costs is given in Table 3 .
| Item or Service | CPT Code | Cost (USD) |
|---|---|---|
| Evaluation | 92004 | 98 |
| Kpro surgery | 65770 | 1328 a |
| Facility fee | n/a | 3500 |
| Pseudophakic PK | 65755 | 593 |
| Aphakic PK | 65750 | 587 |
| ECCE with IOL | 66840 | 328 |
| ECCE without IOL | 66984 | 360 |
| Glaucoma shunt | 66180 | 548 |
| Tarsorrhaphy | 67880 | 175 |
| Follow-up visit | 99211 or 99212 | 200/year |
| BCL fitting and replacements | 92070 | 550 year 1; 300/year thereafter |
| BCL early replacement | 92326 | 100 |
| Medications | vanc/pf/vigamox b | 600/year |
| Contact lens cleaning | 92325 | 150/year |
| Patient travel | n/a | 40 year 1; 20 year 2; 10 thereafter |
| Procedures related to complications | ||
| YAG | 67031 | 333 |
| Membranectomy | 65865 | 453 |
| Kenalog injection | 67500 | 75 |
| Shunt revision | 66185 | 700 |
| Silicone oil | 67042 | 1417 |
| Vitrectomy | 67036 | 905 |
| Iris repair | 66680 | 483 |
| Strabismus surgery | 67331 | 287 |
| Kpro replacement | 65770 | 1328 |
| Blepharoplasty | 15822 | 376 |
a Some procedures (ie, vitrectomy, silicone oil injection) are bundled into the Kpro or PK procedural code and are at no additional cost.
b Vancomycin, prednisolone acetate 1%, vigamox − the standard postoperative regimen.
The expected value decision tree is illustrated in the Figure . Future costs, such as follow-up visits and possible complications requiring procedures, are weighted based on normal clinical practice and complication rates observed in our sample (refer to Table 2 ). Total costs were calculated by the summation of initial costs with appropriately discounted future costs. Discounted future costs included inevitable costs and the average of weighted probable/possible future costs.
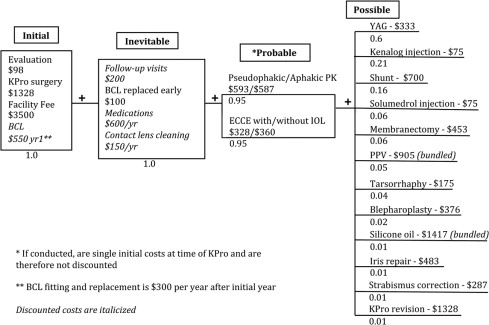
Discounting
There is a consensus that both utilities and costs should be discounted in health care economics analyses. We decided that the commonly used 3% discount rate was appropriate.
Calculation of Quality-Adjusted Life Years
The quality-adjusted life years for the 5-year period was calculated using the following formula:
where t is the yearly transplant survival rate (0.97), u is the average incremental utility (0.182), and d q is the discounting rate for QALYs (3%).
Calculation of Cost
The equation for the total discounted cost associated with Kpro surgery is:
$ 5931 + ∑ x = 1 5 $ 600 ( 1 + d c ) x + $ 550 ( 1 + d c ) 1 + ∑ x = 1 4 $ 300 ( 1 + d c ) x + ∑ x = 1 5 $ 200 ( 1 + d c ) x + ∑ x = 1 5 $ 150 ( 1 + d c ) x + $ 396 ( 1 + d c ) 1
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


