Purpose
To compare the functional and structural effects of ranibizumab versus macular laser therapy in patients with center-involving diabetic macular edema.
Design
Prospective, randomized, single-masked clinical trial.
Methods
Setting : Single center. Study Population : Thirty-three eyes of 33 patients with center-involving diabetic macular edema, with best corrected visual acuity of 55 to 79 Early Treatment Diabetic Retinopathy Study letters at baseline, completing the 48-week study period. Intervention : Subjects were randomized 2:1 to 3 loading doses of ranibizumab then retreatment every 4 weeks as required; or macular laser therapy at baseline, repeated as required every 12 weeks. Exploratory Outcome Measures : Structural imaging studies included greatest linear dimension and area of foveal avascular zone, perifoveal capillary dropout grade, and presence of morphologic features of diabetic macular edema on Spectralis optical coherence tomography (Heidelberg Engineering GmbH, Heidelberg, Germany). Functional measures : Visual acuity, retinal sensitivity in the central 4 and 12 degrees on microperimetry, color contrast sensitivity protan and tritan thresholds, pattern and full-field electroretinogram amplitudes and implicit times, and multifocal electroretinogram amplitude distribution. These were reported at 12, 24, and 48 weeks.
Results
Ranibizumab-treated subjects gained 6.0 vs 0.9 letters lost for laser, demonstrated improved tritan and protan color contrast thresholds, and improved retinal sensitivity. Electrophysiologic function also improved after ranibizumab therapy. No safety issues were evident. Better retinal thickness reduction and structural improvement in optical coherence tomography features of diabetic macular edema were seen with ranibizumab therapy than in the laser group. There was no evidence of progressive ischemia with ranibizumab therapy.
Conclusions
Ranibizumab therapy in the treatment of diabetic macular edema seems to improve retinal function and structure as demonstrated by this evaluation of different assessment methods.
Diabetic macular edema (DME) is a leading cause of visual loss in working-age populations ; knowledge of suitable prognostic indicators thus can be expected to have significant socioeconomic impact. The anti–vascular endothelial growth factor (VEGF) agents ranibizumab and bevacizumab have shown substantial improvements in visual acuity in recent clinical trials of the treatment of DME and were superior to modified Early Treatment Diabetic Retinopathy Study (ETDRS) macular laser therapy. The Diabetic Retinopathy Clinical Research Network protocol I trial showed that approximately 50% of patients in the ranibizumab with deferred laser arm gained 10 letters or more from baseline. The mean change in visual acuity was +3 ETDRS letters in the laser arm compared with +9 letters in the pooled ranibizumab arms. Ranibizumab is now licensed by the United States Food and Drug Administration and the European Medicines Agency for this indication and is used routinely in clinical practice in the United States and Europe. However, with expanded follow-up, the results show a subgroup who had minimal improvement despite repeated therapy; 29% of patients treated with ranibizumab plus deferred laser gained no more than 4 letters at 2 years.
Randomized clinical trials to date have used best-corrected visual acuity (BCVA) as the primary end point, whereas secondary end points focused on central retinal thickness reduction and safety aspects of the drug. No untoward side effects from ranibizumab specific to DME have been reported, with no proven increase in arterial thromboembolic events; the risks of injection are similar to those in neovascular age-related macular degeneration: an 0.04% risk of endophthalmitis per injection. Because of their size and purpose, the pivotal trials have not addressed the differences between ranibizumab and macular laser in terms of detailed functional and structural outcomes that include detailed visual function tests; they have done so only in terms of a foveal-dominated visual acuity test and central retinal thickness change on optical coherence tomography (OCT). Tests of generalized retinal function are lacking. In addition, although macular ischemia is a theoretical concern with anti-VEGF use, the larger trials did not perform routine masked grading of the foveal avascular zone by fluorescein angiography. However, a prospective randomized trial of intravitreal bevacizumab or laser therapy in the management of DME showed no evidence of progression of ischemia at 1 year after treatment with bevacizumab.
The Lucentis in Diabetic Macular Edema: A Treatment Evaluation (LUCIDATE) study performed detailed structural and functional evaluations, including full-field electroretinography (ERG) and microperimetry, after repeated ranibizumab therapy in DME compared with macular laser, with the goal of improving our understanding of its clinical effectiveness and safety profile in the treatment of DME.
Methods
Study Design
The study was a single-center, prospective, randomized clinical trial. It adhered to the tenets of the Declaration of Helsinki. Prospective ethical approval was obtained from the Central London Research Ethics Committee 4 of the United Kingdom National Research Ethics Service, and the trial was registered at www.clinicaltrials.gov (no. NCT01223612). Informed consent to participate in this research study was obtained from all patients before screening and after an explanation of the nature and possible consequences of participation. Study oversight was provided by an independent data and safety monitoring committee.
Participants
Adult patients with type 1 or 2 diabetes and BCVA of 55 to 79 ETDRS letters (Snellen equivalent, 20/30 to 20/80) resulting from center-involving DME, with Spectralis OCT (Heidelberg Engineering GmbH, Heidelberg, Germany) central subfield thickness of 300 μm or more in the study eye, were enrolled. Exclusion criteria included uncontrolled glaucoma; aphakia; cataract precluding fundus photography; external ocular infections; previous anti-VEGF or laser treatment in the preceding 3 months in both eyes; angiographic evidence of macular ischemia defined as foveal avascular zone greatest linear dimension of more than 1000 μm or severe perifoveal capillary loss; other causes for macular edema, for example, after cataract surgery; other causes of visual loss in the study eye; other diseases that may affect the course of macular edema in the study eye; or proliferative diabetic retinopathy, either active or treated within the previous 3 months.
Systemic conditions that precluded trial enrollment included glycosylated hemoglobin of more than 11.0%; past medical history of chronic renal failure requiring either dialysis or kidney transplantation; blood pressure of more than 170/100 mm Hg; an arteriothrombotic event within 6 months before randomization, including myocardial infarction, acute congestive heart failure or other cardiac event, and stroke or transient ischemic attack; planned surgery; and pregnancy or breastfeeding.
Randomization, Allocation, and Intervention
One eye per participant was included to avoid exposure of both eyes to the study drug. If both eyes were eligible, the eye with worse visual acuity became the study eye. Subjects were randomized with 2:1 probability to receive the intervention or standard care (ETDRS macular laser). The randomization list was created using permuted blocks of varying sizes, held by the trial statistician and concealed from the researcher who enrolled, assessed, and allocated treatment to participants.
Subjects in the intervention arm received intravitreal injections of ranibizumab (Lucentis, 0.5 mg in 0.05 mL solution for injection; Novartis Pharmaceuticals UK Ltd., Frimley, United Kingdom) at baseline, 4 weeks, and 8 weeks then every 4 weeks as required according to predefined retreatment criteria to a maximum of 12 injections. Retreatment occurred if BCVA was reduced by 5 letters or more from maximum acuity or if OCT central subfield thickness was more than 300 μm. Subjects in the laser arm received ETDRS macular laser at baseline guided by fluorescein angiography, OCT, and clinical examination. Laser retreatment occurred at 12, 24, and 36 weeks if clinically significant macular edema was still present, in accordance with standard clinical practice at the time; this was guided by the most recent fluorescein angiogram, OCT, and clinical examination results.
Follow-up Visits
All subjects were examined at baseline and at 12, 24, 36, and 48 weeks. Subjects in the ranibizumab arm also were examined at 4-week intervals between these visits for assessment and as-required retreatment.
Assessments
Subjects underwent detailed functional and structural evaluation at baseline and at 12, 24, and 48 weeks. Four-field color fundus photographs were obtained for ETDRS retinopathy grading at baseline and 48 weeks. BCVA (ETDRS), mesopic microperimetry (MP1; Nidek Technologies, Padova, Italy) using a customized 45-point radial grid covering the central 12 degrees of the macula, and color contrast sensitivity (ChromaTest; City University, London, United Kingdom) were performed according to standardized protocols in controlled lighting conditions. Electrophysiologic testing included full-field ERG, pattern ERG (PERG), and multifocal ERG and was performed to incorporate the recommendations of the International Society for Clinical Electrophysiology of Vision. Fundus fluorescein angiography and Spectralis OCT scans were obtained according to predefined protocols.
Blood pressure and glycosylated hemoglobin level were recorded at baseline and 48 weeks to identify any changes in systemic status that could affect retinopathy grade and macular edema. Cataract status was recorded at baseline and 48 weeks as a safety measure and as a possible confounding factor for visual acuity assessment.
Outcome Measures
The prespecified primary efficacy and safety evaluation was performed at week 48: 4 weeks after the final possible ranibizumab injection or 12 weeks after the final possible laser treatment. Exploratory functional and structural outcomes are reported for baseline and 12, 24, and 48 weeks.
Functional measures included change in ETDRS BCVA, mean microperimetric retinal sensitivity at 4 and 12 degrees, and protan and tritan color contrast thresholds. Electrophysiologic parameters included PERG positive component of waveform at 50 ms (P50) amplitude and peak time, PERG negative component of waveform at 95 ms amplitude, and multifocal ERG distribution of amplitudes. Full-field ERG evaluated generalized retinal rod and cone system function.
Structural outcome measures comprised changes in qualitative and quantitative OCT parameters, including macular thickness and volume in 9 ETDRS subfields, obtained from automated measures in the Heidelberg Eye Explorer software (Heidelberg Engineering GmbH, Heidelberg, Germany) without formally correcting for boundary detection error; these measures are highly reproducible. The change in ETDRS severity grade of diabetic retinopathy was reported from 4-field color fundus photographs.
Safety parameters included the reporting of ocular and nonocular adverse events. Changes to the greatest linear dimension and area of the foveal avascular zone on fluorescein angiography, together with the degree of perifoveal capillary loss, were reported.
The grading of photographs both for retinopathy grade and foveal avascular zone measurements was carried out by a trained and certified senior diabetic retinopathy grader at the Moorfields Eye Hospital Reading Centre with 100% adjudication by the clinical lead to ensure data integrity, in accordance with the protocol for another recent clinical trial. All eyes with change and 10% of eyes without change also were discussed and agreed on by an independent trained medical retinal clinician (S.S.). Ten percent of eyes were regraded independently of the adjudication and verification process by the same senior grader, and the intragrader reliability was substantial (κ, 0.76). The microperimetry and electrophysiologic assessors were masked to the patient treatment arm. Evaluation of OCT scans, fundus photographs and fluorescein angiograms was performed by masked Reading Centre graders. The protocol states that the visual acuity assessors were also masked to the patient treatment arm but due to a protocol deviation they had access to the source notes and were potentially unmasked.
Statistical Methods
This was an exploratory study investigating novel parameters. No formal sample size calculation was conducted, but 36 patients were deemed sufficient to examine the distributions of such parameters. Baseline characteristics of the patients were compared by treatment status to assess the adequacy of the randomization. Summary measures for the baseline characteristics were mean and standard deviation for continuous (approximate) normally distributed variables, median and interquartile ranges for non–normally distributed variables, and frequencies and percentages for categorical variables. Our assessment of normality was based on visual examination of histograms to ensure that there was no overt skewness or kurtosis. Because this was an exploratory trial, no intent-to-treat analysis was conducted. An available case analysis of data from all subjects who completed the 48-week follow-up period was conducted (as per a planned statistical analysis agreed on before analyzing the data). Summary statistics were computed for each outcome by treatment group. Analysis of covariance was used to compare continuous outcomes between treatment groups with baseline values of the outcome as a covariate. Any statistical tests conducted used a 2-sided P value of .05; P values were not formally adjusted for multiple testing. Stata statistical software (version 12, StataCorp LP, College Station, Texas, USA) was used for data analysis.
Results
Patient Disposition and Demographics
Subjects were recruited from November 2010 through July 2011. The final follow-up visit was in July 2012. Twenty-five patients were randomized to receive ranibizumab 0.5 mg, and 12 patients were randomized to receive ETDRS macular laser treatment (Supplemental Figure 1, available at AJO.com ). The 48-week study period was completed by 22 (88%) patients in the ranibizumab arm and 11 (92%) patients in the laser arm. Their baseline characteristics were broadly comparable ( Table 1 ), but in the laser group, there were no subjects with type 1 diabetes and a greater median duration of DME. There was no significant change in blood pressure or systemic diabetes control over the study period.
| Ranibizumab Group | Laser Group | |
|---|---|---|
| No. of patients | 22 | 11 |
| Demographics | ||
| Sex (female) | 7 (31.8%) | 5 (45.5%) |
| Age (y) | 64.9 (58.4 to 71.0) a | 67.4 (62.8 to 74.6) a |
| Ethnicity | ||
| White or white British | 15 (68.2%) | 7 (63.6%) |
| Black or black British | 0 (0%) | 1 (9.1%) |
| Asian or Asian British | 5 (22.7%) | 2 (18.2%) |
| Other | 2 (9.1%) | 1 (9.1%) |
| Systemic profile | ||
| Type of diabetes | ||
| 1 | 4 (18.2%) | 0 (0%) |
| 2 | 18 (81.8%) | 11 (100%) |
| Duration of diabetes (y) | 18.5 (10 to 26)* | 18 (14–25) a |
| Systolic BP | 130 ± 17 | 135 ± 17 |
| Diastolic BP | 75 ± 10 | 71 ± 9 |
| HbA 1C | 7.93 ± 1.31 | 7.25 ± 0.92 |
| Ocular profile | ||
| Study eye (left/right) | 17/5 | 5/6 |
| Duration of DME (mos) | 21 (14 to 27) a | 32 (15 to 60) a |
| Mean no. of previous macular laser treatments | 3 (1 to 4) a | 4 (3 to 5) |
| Cataract grade | ||
| 0 | 6 (27.3%) | 1 (9.1%) |
| 1 | 12 (54.5%) | 5 (45.5%) |
| 2 | 0 (0%) | 1 (9.1%) |
| 3 | 0 (0%) | 0 (0%) |
| IOL | 4 (18.2%) | 4 (36.4%) |
Treatments Received
In the ranibizumab arm, 24 subjects received injections at baseline and at 4 and 8 weeks. One subject did not agree to contraceptive use so did not receive ranibizumab treatment, withdrew from the trial, and received standard clinical care (laser treatment). All subjects in the laser arm received macular laser therapy at baseline. A summary of the total number of treatments given at each time point and the mean cumulative number of treatments given appears in Table 2 ; ranibizumab-treated subjects received a mean of 9 injections; laser-treated subjects underwent a mean of 2.6 sessions of macular laser. One subject in each arm underwent cataract surgery during the trial.
| Baseline | 4 | 8 | 12 | 16 | 20 | 24 | 28 | 32 | 36 | 40 | 44 | 48 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ranibizumab | |||||||||||||
| No. of treatments given (at each time point) | 22 | 22 | 22 | 16 | 17 | 16 | 13 | 14 | 15 | 15 | 12 | 13 | N/A |
| Mean cumulative no. of treatments | 1 | 2 | 3 | 3.7 | 4.5 | 5.2 | 5.8 | 6.5 | 7.1 | 7.8 | 8.4 | 9.0 | 9.0 |
| Laser | |||||||||||||
| No. of treatments given (at each time point) | 11 | 9 | 4 | 5 | N/A | ||||||||
| Mean cumulative no. of treatments | 1 | 1.8 | 2.2 | 2.6 | 2.6 |
Efficacy
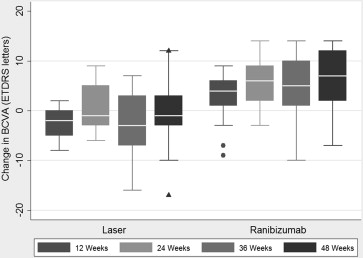
Mean BCVA at baseline in the ranibizumab group was 70.4 letters (standard deviation [SD], 4.9 letters) and 63.8 letters (SD, 5.7 letters) in the laser group. This improved to 76.4 letters (SD, 8.5 letters) in the ranibizumab group at 48 weeks, but decreased to 62.9 letters (SD, 10.6 letters) in the laser group ( P = .083, analysis of covariance). This represented a 6-letter gain for ranibizumab versus a loss of 0.9 letters for laser treatment ( Figure 1 ).
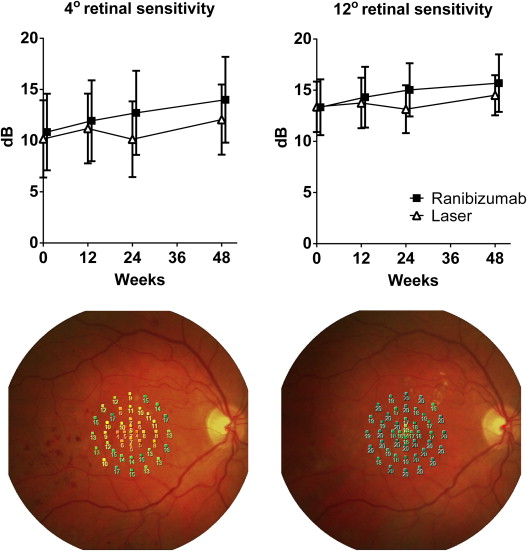
Retinal sensitivity in the central 4 and 12 degrees improved to a greater extent in the ranibizumab group (3.2 and 2.4 dB) than in the laser group (1.9 and 1.1 dB), shown in Figure 2 . Color contrast sensitivity improved in the protan and tritan axes for ranibizumab-treated subjects by 3.4% and 10.8%, whereas laser-treated subjects worsened in the protan axis by 8.1% and improved in the tritan axis by 3.1% ( Table 3 ).
| Data and Time Point (wks) | Ranibizumab Group (n = 22 All Time Points) | Laser Group (n = 11 All Time Points) |
|---|---|---|
| BCVA letter score | ||
| 0 | 70.4 ± 4.9 | 63.8 ± 5.7 |
| 12 | 73.1 ± 6.2 | 60.8 ± 5.6 |
| 24 | 75.7 ± 7.7 | 64.4 ± 7.5 |
| 36 | 75.3 ± 8.9 | 61.3 ± 9.0 |
| 48 | 76.4 ± 8.5 | 62.9 ± 10.6 |
| Tritan sensitivity (%) | ||
| 0 | 80.7 ± 29.6 | 88.9 ± 20.7 |
| 12 | 71.0 ± 33.5 | 92.4 ± 13.6 |
| 24 | 71.8 ± 34.8 | 84.7 ± 24.2 |
| 48 | 69.9 ± 34.5 | 85.8 ± 25.0 |
| Protan sensitivity (%) | ||
| 0 | 21.4 ± 22.5 | 22.9 ± 22.8 |
| 12 | 18.2 ± 15.7 | 32.5 ± 35.4 |
| 24 | 16.4 ± 15.9 | 18.0 ± 22.6 |
| 48 | 18.0 ± 16.9 | 31.0 ± 35.0 |
| Microperimetry 4-degree sensitivity (dB) | ||
| 0 | 10.8 ± 3.7 | 10.2 ± 3.8 |
| 12 | 11.9 ± 3.9 | 11.2 ± 3.4 |
| 24 | 12.7 ± 4.1 | 10.1 ± 3.7 |
| 48 | 14.0 ± 4.2 | 12.1 ± 3.4 |
| Microperimetry 12-degree sensitivity (dB) | ||
| 0 | 13.3 ± 2.7 | 13.4 ± 2.5 |
| 12 | 14.3 ± 3.0 | 13.8 ± 2.5 |
| 24 | 15.0 ± 2.6 | 13.1 ± 2.3 |
| 48 | 15.7 ± 2.8 | 14.5 ± 2.0 |
Amplitudes and, where appropriate, peak times of the major waveforms in each of the electrophysiologic tests performed are presented as mean ± standard deviation. Where technical difficulties such as eye closure or excessive blinking during the test occurred, the results for that subject at that visit are not included. When the PERG was undetectable despite a technically satisfactory examination, that is, severe macular dysfunction, those data were not included in group peak time means; the peak time of an undetectable wave should not be considered as 0. Laser-treated subjects experienced a decline of 0.13 μv (10.5%) in the P50 component from baseline to 48 weeks, whereas ranibizumab-treated subjects showed a minor increase (0.04 μv; 2.9%; Table 4 ).
| Time Point (wks) | Ranibizumab | Laser | ||||||
|---|---|---|---|---|---|---|---|---|
| No. Analyzed | P50 Amplitude (μv) | P50 Peak Time (ms) | N95 Amplitude (μv) | No. Analyzed | P50 Amplitude (μv) | P50 Peak Time (ms) | N95 Amplitude (μv) | |
| 0 | 17 | 1.37 ± 0.40 | 54.6 ± 5.6 | 2.17 ± 0.70 | 8 | 1.21 ± 0.41 | 55.8 ± 3.8 | 2.00 ± 0.51 |
| 12 | 14 | 1.21 ± 0.35 | 54.2 ± 3.9 | 2.16 ± 0.60 | 9 | 1.08 ± 0.32 | 53.7 ± 5.2 | 1.50 ± 0.38 |
| 24 | 17 | 1.23 ± 0.56 | 53.9 ± 4.2 | 2.16 ± 0.83 | 10 | 1.10 ± 0.41 | 51.9 ± 4.3 | 1.62 ± 0.56 |
| 48 | 18 | 1.41 ± 0.45 | 54.8 ± 3.7 | 2.13 ± 0.71 | 9 | 1.08 ± 0.36 | 53.5 ± 3.7 | 1.79 ± 0.72 |
Multifocal ERG responses at central and peripheral locations were compared between ranibizumab-treated and laser-treated groups and are reported qualitatively with reference to the investigators’ laboratory normal data. The changes in response amplitude were variable in both subject groups at 12, 24, and 48 weeks. All subjects from both ranibizumab-treated and laser-treated groups demonstrated moderate to severely reduced central responses, that is, moderate to severe central macular dysfunction, at baseline, defined as an amplitude reduction of 70% or more. Most subjects, approximately 60%, from the laser-treated group demonstrated additional mild to moderate peripheral macular dysfunction (defined as 25% to 70% reduction in amplitude) at baseline, compared with 10% in the ranibizumab-treated group. Thirty-four percent of ranibizumab-treated subjects and 18% of laser-treated subjects experienced mild to moderate increase in central responses (ie, central macular function) at 48 weeks. Fourteen percent of ranibizumab-treated subjects and 27% of laser-treated subjects experienced reduction in central responses at 48 weeks. The remaining subjects (52% of ranibizumab-treated subjects and 55% of laser-treated subjects) showed no change in central or peripheral responses from baseline to 48 weeks.
Structural Imaging Studies
Using automated measurements, ranibizumab-treated subjects showed a decrease in the central OCT subfield thickness from 455 μm (SD, 79 μm) to 324 μm (SD, 78 μm; reduction of 132 μm [SD, 98 μm]), whereas laser-treated subjects showed a decrease from 488 μm (SD, 96 μm) to 385 μm (SD, 98 μm; reduction of 103 μm [SD, 88 μm]; P = .06 at 48 weeks, ranibizumab vs laser). Retinal thickness data were recorded from the 9 ETDRS subfields with total macular volume. Details are shown in Table 5 , and the change in central subfield thickness over time is shown in Figure 3 . The prevalence of the morphologic features of DME identified by masked reading center graders is shown for the 2 groups in Table 6 . The prevalence of subretinal fluid in the central subfield decreased in the ranibizumab arm, but not the laser arm, by 48 weeks. There was no clear evidence of a treatment effect in either group on cysts or cystoid edema in the inner or outer retina either in the central subfield or in the 4 surrounding (inner) subfields. All subjects had an abnormal foveal depression at baseline. In the laser group, 9% of subjects (1/11) had a normal foveal depression by 48 weeks, but in the ranibizumab group, this was 40% (9/22 subjects). The 2 groups were comparable at baseline in terms of the prevalence of interruptions in the lines representing the external limiting membrane or inner segment–outer segment junction (ellipsoid layer), but at 48 weeks, the ranibizumab group showed a significantly lower prevalence of interrupted external limiting membrane compared with the laser group ( P = .01, Fisher exact test). The prevalence of interrupted inner segment–outer segment junction in the laser group increased, whereas in the ranibizumab group, it decreased ( P = .14). In the ranibizumab group, 5 subjects (22.7%) had an epiretinal membrane at baseline; 4 subjects had an epiretinal membrane at baseline in the laser group (36.4%). At 48 weeks, there was no significant difference in these figures. One subject in the ranibizumab group had an incomplete posterior vitreous detachment identified at baseline that persisted until 48 weeks. No subjects had lamellar hole, macular hole, or vitreomacular traction at any time point.
| Subfield | Ranibizumab | Laser | P Value (48 Weeks) a | ||||
|---|---|---|---|---|---|---|---|
| 0 Weeks | 48 Weeks | Change | 0 weeks | 48 Weeks | Change | ||
| Center | 455.4 ± 78.7 | 323.9 ± 78.1 | –131.5 ± 98.0 | 488.2 ± 96.3 | 385.3 ± 98.2 | –102.9 ± 88.4 | .06 |
| Inner | |||||||
| Superior | 458.7 ± 84.2 | 369.7 ± 61.0 | –89.0 ± 63.6 | 448.8 ± 96.6 | 390.0 ± 58.0 | –58.8 ± 67.3 | .37 |
| Nasal | 424.7 ± 56.6 | 356.4 ± 31.9 | –68.3 ± 53.1 | 440.1 ± 105.2 | 400.0 ± 78.0 | –40.1 ± 46.5 | .03 |
| Inferior | 413.9 ± 64.1 | 339.3 ± 26.5 | –74.6 ± 55.3 | 426.6 ± 49.8 | 385.8 ± 39.6 | –40.8 ± 37.2 | .0003 |
| Temporal | 455.5 ± 75.8 | 349.0 ± 49.3 | –106.5 ± 68.6 | 450.2 ± 70.3 | 384.9 ± 40.6 | –65.3 ± 65.1 | .05 |
| Outer | |||||||
| Superior | 366.3 ± 86.0 | 319.8 ± 45.6 | –46.5 ± 48.0 | 360.5 ± 73.4 | 336.7 ± 51.3 | –23.8 ± 31.1 | .34 |
| Nasal | 365.1 ± 62.9 | 322.7 ± 27.3 | –42.5 ± 46.4 | 370.0 ± 59.5 | 358.5 ± 53.5 | –11.5 ± 29.9 | .02 |
| Inferior | 334.7 ± 70.5 | 290.9 ± 26.6 | –43.8 ± 49.5 | 331.5 ± 42.2 | 319.9 ± 37.5 | –12.4 ± 17.5 | .02 |
| Temporal | 363.3 ± 76.8 | 303.5 ± 37.0 | –59.8 ± 46.3 | 369.3 ± 104.4 | 340.3 ± 74.4 | –29.0 ± 48.5 | .06 |
| Total macular volume | 10.53 ± 1.62 | 8.96 ± 0.81 | –1.57 ± 0.98 | 10.47 ± 1.69 | 9.77 ± 1.15 | –0.69 ± 0.82 | .03 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


