Purpose
To compare the effects of 2 corticosteroids on corneal thickness and visual acuity after cataract surgery.
Design
Multicenter, randomized, contralateral-eye, double-masked trial.
Methods
Fifty-two patients (104 eyes) underwent bilateral phacoemulsification. The first eye randomly received difluprednate 0.05% or prednisolone acetate 1%; the fellow eye received the alternative. Before surgery, 7 doses were administered over 2 hours; 3 additional doses were given after surgery, before discharge. For the remainder of the day, corticosteroids were administered every 2 hours, then 4 times daily during week 1 and twice daily during week 2. Corneal pachymetry, visual acuity, and corneal edema were evaluated before surgery and at days 1, 15, and 30 after surgery. Endothelial cell counts were evaluated before surgery and at 30 days after surgery. Retinal thickness was evaluated before surgery and at 15 and 30 days after surgery.
Results
Corneal thickness at day 1 was 33 μm less in difluprednate-treated eyes ( P = .026). More eyes were without corneal edema in the difluprednate group than in the prednisolone group at day 1 (62% vs 38%, respectively; P = .019). Uncorrected and best-corrected visual acuity at day 1 were significantly better with difluprednate than prednisolone by 0.093 logMAR lines ( P = .041) and 0.134 logMAR lines ( P < .001), respectively. Endothelial cell density was 195.52 cells/mm 2 higher in difluprednate-treated eyes at day 30 ( P < .001). Retinal thickness at day 15 was 7.74 μm less in difluprednate-treated eyes ( P = .011).
Conclusions
In this high-dose pulsed-therapy regimen, difluprednate reduced inflammation more effectively than prednisolone acetate, resulting in more rapid return of vision. Difluprednate was superior at protecting the cornea and reducing macular thickening after cataract surgery.
As the technology of cataract surgery has progressed, so too, has the increased patient demand for excellent vision and safety after the procedure. Innovative lens design and improved phacoemulsification techniques have assisted the ophthalmic surgeon in meeting these demands, but visual rehabilitation after cataract surgery is sometimes delayed by the inflammatory processes that are induced by phacoemulsification. Inflammation is induced in all cataract surgery by the mechanical transmission of energy into the eye, disruption of cell membranes, and the normal healing process. Inflammation results in disruption of the blood-aqueous barrier with the clinical findings of cell and flare, as well as cystoid macula edema. Postoperative topical corticosteroids and preoperative and postoperative topical nonsteroidal anti-inflammatories are used routinely to reduce inflammation and improve visual outcomes after cataract surgery. Despite the use of these pharmacologic agents, transient corneal edema has emerged as one of the major factors hindering the improvement of vision in the first days after surgery, and cystoid macula edema may reduce quality of vision for weeks and months after the procedure. Therefore, reducing inflammation and its potential damage to the corneal endothelium and retina is a high priority for the ophthalmic surgeon.
Difluprednate ophthalmic emulsion 0.05% (Durezol; Alcon Laboratories, Fort Worth, Texas, USA) is a new prednisolone derivative classified as a very strong steroid in dermatology. Difluprednate has a high glucocorticoid receptor binding affinity as a result of fluorination at the C-6 and C-9 positions, with superior tissue penetration as a result of the addition of an acetate ester at position C-21. In addition, there is increased bioavailability and dose uniformity resulting from the formulation of difluprednate as an emulsion, rather than a suspension. Difluprednate has been shown to be similar to (Ohji M, et al. Efficacy and safety results of a phase III study of difluprednate ophthalmic emulsion, 0.05%, in postoperative inflammation. Poster presented at Annual Meeting of the Association for Research in Vision and Ophthalmology. May 6–10, 2007, Fort Lauderdale, Florida) or more potent than (Ohno S, et al. Phase III non-inferiority study of difluprednate ophthalmic emulsion, 0.05% in the treatment of anterior uveitis. Poster presented at Annual Meeting of the Association for Research in Vision and Ophthalmology. May 6–10, 2007, Fort Lauderdale, Florida) betamethasone sodium phosphate 0.1% in terms of efficacy. Betamethasone has been shown to be approximately 6 times more potent than prednisolone acetate 1% on a molecular basis. A trial in which difluprednate was used 4 times daily compared with prednisolone acetate 8 times daily in the treatment of endogenous uveitis demonstrated that difluprednate was at least equal in reducing inflammation. Additionally, the use of difluprednate treatment 4 times daily was associated with a statistically significant more rapid return of visual acuity than seen with the use of prednisolone acetate 8 times daily.
High-dose corticosteroids have proven to be protective of neural function after injury. In animal models, high-dose corticosteroids have been shown to reduce posttraumatic neuronal degeneration and to improve outcomes in animals with head injury. Corticosteroids also have been found to protect cochlear hair cells after trauma, photoreceptors after subretinal hemorrhage, and ganglion cells after optic nerve crush. In humans, traumatic spinal cord injuries routinely are treated with high-dose corticosteroids to improve neuronal function and to prevent neuronal cell death. It has been proposed that the mechanism of neural protection is the induction of glucocorticoid modulatory element-binding protein 1, which disrupts the autoactivation of procaspase 2 and the induction of apoptosis.
In addition to providing a level of protection after injury, steroids administered before surgery have been a mainstay in many surgeries because of their ability to regulate the so-called stress response induced by surgery. The stress response is a combined endocrine and inflammatory response to trauma, and often can contribute to postoperative morbidity. In this setting, corticosteroids—which are known immunomodulators and reduce the amount of circulating proinflammatory cytokines—play an important role in limiting potentially detrimental effects of this innate response. The use of preoperative steroids has been studied extensively outside of ophthalmology, and a systematic review of randomized controlled clinical trials of major surgical procedures revealed several important points: (1) the steroid decreased production of proinflammatory mediators and increased the release of anti-inflammatory mediators; (2) administration of the steroid reduced local edema and pain in many of the procedures; and (3) the timing of steroid administration was critically important: steroids dosed less than 1 to 2 hours before surgery seemed to have less effect, potentially because the onset of action for steroids (which is mediated by altered protein synthesis) typically takes 1 to 2 hours, and inflammatory responses to surgery are initiated immediately at the time of the incision. Although most of these surgeries were considered more invasive than cataract surgery and most used intravenously delivered steroids (as opposed to topical), the inciting trauma of all surgeries result in the same inflammatory response. Based on this, we believe that it is plausible that the preadministration of steroids may have a protective effect after cataract surgery. In addition, because the corneal endothelium and retina are of the same neuroectodermal origin as the brain and spinal cord, the corneal endothelium and retina also may benefit from pulsed high-dose steroids to minimize the effects of surgical trauma. In this article, we report on the use of a pulse-dosed steroid regimen that is used in the perisurgical period and compare treatment with difluprednate and prednisolone in this novel pulse schedule.
Methods
Study Design
This prospective, multicenter, double-masked, randomized, contralateral eye study compared treatment with the corticosteroids difluprednate 0.05% and prednisolone acetate 1% in phacoemulsification cataract surgery. Patients were enrolled at 3 clinical sites in 1 country.
Patient Selection
Men and women (21 years of age and older) were selected on the basis of requiring bilateral cataract surgery. For inclusion, patients needed to be in good health, to be scheduled to undergo standard cataract surgery with topical anesthesia in both eyes within 6 to 25 days between surgeries, and to have best-corrected visual acuity better than 20/100 in both eyes with a visual potential of 20/25 or better. Patients were screened for their willingness and ability to administer the eye drops, to record the times, to complete the study, and to comprehend and sign the informed consent form. Exclusion criteria included the use of eye medications other than study drugs; a need for regional or general anesthesia during surgery; inability to dilate pupils more than 5 mm before surgery; complicated cataract surgery, including use of iris hooks or iris stretchers; any surgical complication; history of uveitis, iritis, or intraocular inflammation; confirmed or suspected active viral, bacterial, or fungal keratoconjunctival disease; glaucoma; macular pathologic features; lack of an intact corneal epithelium; diabetes mellitus; and any condition requiring use of a systemic steroidal or nonsteroidal anti-inflammatory drug during the study period. A history of steroid-related intraocular pressure (IOP) rise and previous intraocular surgery also resulted in exclusion. Women who were pregnant, nursing, or planning a pregnancy in the study period were excluded from the study.
Randomization and Treatment
For each patient, surgery on one eye preceded surgery on the other eye by at least 6 days and no more than 25 days. After presurgical evaluation and screening, patients were assigned randomly to receive either difluprednate or prednisolone for treatment of the first eye; the second eye was assigned the alternative medication. Allocation of the medication was concealed from the investigators based on a random number list generated using randomizer.org. Both investigators and patients were masked to the treatment condition. Study medication (obtained from commercial sources) was relabeled in a manner so as to obscure the bottle shape and contents.
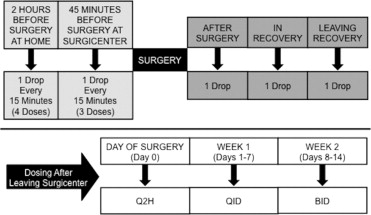
The dosing schedule for prednisolone and difluprednate is shown in Figure 1 . On the day of surgery (day 0), patients instilled 1 drop of topical steroid medication every 15 minutes into the eye scheduled for surgery during the hour before arrival at the surgery center (4 drops total). On arrival, staff administered 1 drop every 15 minutes (3 drops total). After surgery, a single drop was instilled immediately on entry to surgical recovery, another drop was administered while in surgical recovery, and a final drop was given on leaving surgical recovery. After discharge, patients administered 1 drop of medication every 2 hours for the remainder of day 0. Starting on the day after surgery (day 1), patients administered medication to the operated eye 4 times daily for 1 week and twice daily for the subsequent week.
Additional medications or devices administered during the study included an anti-infective based on surgeon preference (moxifloxacin 0.5%, Vigamox; Alcon Laboratories; or gatifloxacin 0.3%, Zymar; Allergan, Inc, Irvine, California, USA) 4 times daily starting 3 days before surgery and continuing for 10 days after surgery; a nonsteroidal anti-inflammatory drug based on surgeon preference (nepafenac 0.1%, Nevanac; Alcon Laboratories; or ketorolac tromethamine 0.4%, Acular LS; Allergan, Inc), beginning 3 days before surgery and continuing for 4 weeks after surgery. All patients were dilated with tropicamide 1% and phenylephrine 2.5% before surgery. During surgery, all patients received the same viscoelastic sodium hyaluronate (Healon; Abbott Medical Optics, Abbott Park, Illinois, USA).
Study End Points and Assessments
Primary end point
The primary end point evaluated was the change from baseline in corneal thickness between the difluprednate- and prednisolone acetate-treated groups at day 1.
Secondary end points
The following exploratory secondary end points also were compared between the difluprednate 0.05% and prednisolone acetate 1% groups: corneal thickness at day 15 ± 2 days and at day 30 ± 3 days; uncorrected visual acuity (UCVA) at all time points; best-corrected visual acuity (BCVA) at all time points; corneal edema (epithelial and stromal) at all time points; endothelial cell counts at day 30 ± 3 days; optical coherence tomography-central retinal thickness (OCT-CRT) at day 15 ± 2 days and at day 30 ± 3 days; and IOP at all time points.
Corneal thickness
Central corneal thickness was measured by ultrasonic corneal pachymetry under topical anesthetic (Sonomed Model 200P+ used for all patients). The value recorded at each time point was the mean of 3 measurements obtained. Measurements were obtained at the preoperative visit and on postsurgical days 1, 15, and 30.
Visual acuity
UCVA and BCVA were measured using the Early Treatment Diabetic Retinopathy Study chart under identical testing conditions. Visual acuity was assessed at the preoperative visit and on postsurgical days 1, 15, and 30. Visual acuity was converted to logarithm of the minimal angle of resolution (logMAR) units for numerical analysis and comparison.
Corneal edema
Corneal edema (stromal folds and epithelial edema) was measured by slit-lamp examination at the preoperative visit and on postsurgical days 1, 15, and 30. Stromal corneal edema was graded on a 0-to-3 scale: 0 = no edema, 1 = mild edema, 2 = moderate edema, and 3 = severe edema. Epithelial corneal edema was graded on a 0-to-3 scale: 0 = no edema, 1 = epithelial bedewing, 2 = small bullae, and 3 = large bullae.
Endothelial cell density
The density of central corneal endothelial cells (cell per square millimeter) was determined by cell counts obtained under specular microscopy (Konan Cellchek; Konan Medical USA, Irvine, California, USA). Readings for all patients were performed by the same masked observer who performed a manual test. Measurements were obtained at the preoperative visit and on day 30.
Retinal thickness
Central retinal thickness (6-mm diameter circle or 6-mm square centered on the fovea) was measured by either Cirrus or Stratus OCT (Carl Zeiss, Dublin, California, USA) at the preoperative visit and on postsurgical days 15 and 30. In all cases, each patient had the same OCT used on both eyes.
Intraocular pressure
IOP was measured by Goldmann applanation tonometry in millimeters of mercury. Measurements were obtained at the preoperative visit and on days 1, 15, and 30.
Statistical Analyses
Eyes were the analytical unit, and the outcome measurement was treated as paired data for each patient. Between-treatment comparisons were conducted using the paired t test for numeric variables where data were normally distributed, and the McNemar test for dichotomized variables where data were not normally distributed. All tests were 2 sided with a confidence level set to 95%. Statistical computing was performed with SAS software version 9.1 (SAS Institute, Cary, North Carolina, USA). Data collected at the clinical trial sites on an ad hoc basis suggested that an increase in corneal thickness of 37 μm occurred with prednisolone acetate 4 times daily compared with 20 μm with difluprednate 0.05% 4 times daily. The normal range for corneal thickness is approximately 555 μm. Therefore, a sample size of 67 was required for a difference of 17 μm to be statistically significant ( P < .05), assuming a standard deviation of 30 μm.
Results
Study Population
A total of 63 patients were enrolled in the study between July 28, 2009, and June 30, 2010. The mean age at enrollment was 70.47 years (range, 51 to 105 years). Most patients (38/52, 73%) were female. Surgeries were performed under topical anesthesia with limbal incisions. Eleven patients (17.5%) were not included for the efficacy end point analysis because of protocol violations (the most common was the use of a dispersive viscoelastic); however, these patients were included in the safety analysis. The remaining 52 patients (104 eyes) were included in the efficacy evaluations, which were carried out on a per-protocol basis. Values for the parameters measured before and during surgery are shown in Table 1 . There were no significant differences between treatment groups on any preoperative or intraoperative measures.
| Difluprednate | Prednisolone | P Value b | |
|---|---|---|---|
| Uncorrected visual acuity (logMAR) | 0.874 ± 0.496 | 0.888 ± 0.477 | .66 |
| Best-corrected visual acuity (logMAR) | 0.379 ± 0.315 | 0.345 ± 0.292 | .46 |
| Central corneal thickness (μm) | 561.58 ± 36.01 | 562.24 ± 38.79 | .66 |
| Retinal thickness (μm) | 258.38 ± 37.41 | 254.92 ± 30.16 | .23 |
| Endothelial cells (cells/mm 2 ) | 2299.77 ± 340.73 | 2279.06 ± 352.10 | .46 |
| Intraocular pressure (mm Hg) | 15.05 ± 3.28 | 14.95 ± 3.00 | .71 |
| Lens density, n (%) | .33 | ||
| Soft | 8 (15%) | 14 (27%) | |
| Medium | 37 (71%) | 33 (63%) | |
| Hard | 7 (14%) | 5 (10%) | |
| Phacoemulsification energy per eye (J) | 10.43 ± 10.37 | 8.92 ± 7.65 | .40 |
| Range | 0 to 60 | 0 to 34 |
a n = 52 per treatment group, except intraocular pressure, where n = 63.
b Student t test for numeric variables; Pearson chi-square test for categorical variables.
Study Outcomes
Corneal thickness
At day 1, the mean central corneal thickness of difluprednate-treated eyes increased by 28 μm (from 562 to 590 μm), roughly half the increase seen in the prednisolone-treated eyes, in which the increase was by 57 μm (from 562 to 619 μm). By day 15, the corneal swelling largely had subsided, and by day 30, it had resolved in both treatment groups. At day 1, the central corneal thickness of the eyes given difluprednate averaged 32.59 μm less than the corneal thickness of the fellow eyes given prednisolone ( P = .026; Table 2 ). At subsequent time points, there was no statistically significant difference between treatments ( Figure 2 ).
| Difluprednate Eyes | Prednisolone Eyes | Fellow-Eye Difference a | |||||
|---|---|---|---|---|---|---|---|
| Outcome, Time | No. | Mean ± SD | No. | Mean ± SD | No. | Mean ± SD | P Value b |
| Uncorrected visual acuity (logMAR) | |||||||
| Baseline | 52 | 0.874 ± 0.496 | 52 | 0.888 ± 0.477 | 52 | −0.015 ± 0.239 | .6614 |
| Postoperative day 1 | 51 | 0.231 ± 0.198 | 51 | 0.322 ± 0.250 | 50 | −0.093 ± 0.314 | .0416 |
| Postoperative day 15 | 51 | 0.156 ± 0.144 | 52 | 0.173 ± 0.187 | 51 | −0.004 ± 0.154 | .8423 |
| Postoperative day 30 | 52 | 0.128 ± 0.115 | 51 | 0.146 ± 0.131 | 51 | −0.016 ± 0.120 | .3496 |
| Best-corrected visual acuity (logMAR) | |||||||
| Baseline | 52 | 0.379 ± 0.315 | 52 | 0.345 ± 0.292 | 52 | 0.034 ± 0.334 | .4632 |
| Postoperative day 1 | 51 | 0.100 ± 0.130 | 51 | 0.231 ± 0.207 | 50 | −0.134 ± 0.244 | .0003 |
| Postoperative day 15 | 51 | 0.045 ± 0.107 | 52 | 0.038 ± 0.077 | 51 | 0.005 ± 0.090 | .6655 |
| Postoperative day 30 | 52 | 0.015 ± 0.076 | 51 | 0.062 ± 0.151 | 51 | −0.047 ± 0.154 | .0319 |
| Central corneal thickness (μm) | |||||||
| Baseline | 52 | 561.58 ± 36.01 | 52 | 562.24 ± 38.79 | 52 | −0.66 ± 10.80 | .6613 |
| Postoperative day 1 | 50 | 589.69 ± 48.04 | 51 | 619.28 ± 72.75 | 49 | −32.59 ± 99.08 | .0257 |
| Postoperative day 15 | 51 | 571.51 ± 44.46 | 52 | 567.60 ± 39.19 | 51 | 3.24 ± 20.36 | .2609 |
| Postoperative day 30 | 52 | 563.49 ± 37.06 | 51 | 562.97 ± 39.84 | 51 | 0.49 ± 10.64 | .7435 |
| Endothelial cell density (cells/mm 2 ) | |||||||
| Baseline | 52 | 2299.77 ± 340.73 | 52 | 2279.06 ± 352.10 | 52 | 20.71 ± 202.31 | .4638 |
| Postoperative day 30 | 51 | 2088.14 ± 364.04 | 51 | 1894.61 ± 431.84 | 50 | 195.52 ± 377.33 | .0006 |
| Retinal thickness (μm) | |||||||
| Baseline | 52 | 258.38 ± 37.41 | 52 | 254.92 ± 30.16 | 52 | 3.46 ± 20.57 | .2306 |
| Postoperative day 15 | 50 | 260.68 ± 30.26 | 51 | 268.73 ± 33.05 | 50 | −7.74 ± 20.74 | .0111 |
| Postoperative day 30 | 52 | 259.12 ± 33.68 | 51 | 263.65 ± 32.47 | 51 | −4.94 ± 27.49 | .2052 |
| Intraocular pressure (mm Hg) | |||||||
| Baseline | 63 | 15.05 ± 3.28 | 63 | 14.95 ± 3.00 | 63 | 0.10 ± 2.08 | .7181 |
| Postoperative day 1 | 61 | 18.56 ± 4.29 | 62 | 17.82 ± 4.33 | 60 | 0.72 ± 4.18 | .1896 |
| Postoperative day 15 | 59 | 15.44 ± 4.11 | 60 | 14.58 ± 2.88 | 58 | 0.93 ± 3.96 | .0790 |
| Postoperative day 30 | 60 | 13.78 ± 2.39 | 58 | 13.83 ± 3.07 | 57 | −0.09 ± 3.40 | .8462 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


