Purpose
Descemet membrane endothelial keratoplasty is a new technique for the replacement of diseased corneal endothelium with healthy donor endothelium. During this procedure, manipulation of the donor endothelium–Descemet membrane layer within the recipient anterior chamber can be associated with loss of the correct anterior-posterior orientation. Herein, we describe a simple method to keep the correct donor orientation during transplantation by marking the edge of the endothelium–Descemet membrane layer.
Design
Interventional case series.
Methods
Donor discs were created by stripping of the endothelium–Descemet membrane layer from corneoscleral buttons. Before completion of stripping, 3 circular marks were set in an identifiable order at the edge of the donor disc. After removal of the recipient Descemet membrane, the donor graft was inserted into the anterior chamber, unfolded, and attached to the posterior corneal stroma with an air bubble. Correct anterior-posterior orientation of the graft was identified by the clockwise order of the 3 marks.
Results
The marks allowed identification of the anterior-posterior orientation of the endothelium–Descemet membrane layer after unfolding in the anterior chamber and attachment to the recipient stroma in all cases. In 4 of 25 patients, orientation was upside down after unfolding of the roll, requiring inversion of the refolded donor graft.
Conclusions
Marking the edges of the endothelium–Descemet membrane layer helps to keep the correct orientation without adverse effects on donor detachment or corneal clarity during and after Descemet membrane endothelial keratoplasty.
After a century of penetrating keratoplasty, lamellar techniques recently have been improved, allowing for differentiated replacement of diseased corneal structures. Several techniques have been described to treat disorders of the corneal endothelium, most importantly deep lamellar keratoplasty and its variant, Descemet stripping automated endothelial keratoplasty. In deep lamellar keratoplasty and Descemet stripping automated endothelial keratoplasty, a thin graft consisting of posterior stroma, Descemet membrane, and endothelium is used that replaces diseased tissue and also changes the topography of the posterior surface of the cornea. More recently, Descemet membrane endothelial keratoplasty (DMEK) allows transplantation of the endothelium–Descemet membrane layer practically without alteration of the posterior stroma.
Although successful DMEK renders very good intermediate term outcomes, early graft failure frequently occurs and represents a significant problem prohibiting the widespread use of this technique. Graft failure mainly is the result of technical problems during unfolding and manipulation of the donor endothelium–Descemet membrane layer within the anterior chamber (AC). Although an endothelium–Descemet membrane layer stained with trypan blue typically displays a characteristic appearance with rolled edges indicating endothelial cells on the outside of the curved edge, this feature becomes less prominent after several attempts to manipulate the endothelium–Descemet membrane layer in the AC, especially in patients with corneal edema. Furthermore, immediately after preparation from the corneoscleral button, some grafts have a weak tendency to form a roll with typical appearance of the edges. Particularly with impaired visibility of the AC, the roll easily can flip and turn, and consequently orientation is lost. Misplacement of the endothelium–Descemet membrane layer adversely affects the corneal clarity, and in most cases, the endothelium–Descemet membrane layer finally detaches.
To overcome this problem, we developed a simple and reliable procedure that uses marks on the edge of the endothelium–Descemet membrane layer graft to allow permanent orientation of the endothelium–Descemet membrane layer within the AC.
Methods
Donor Preparation
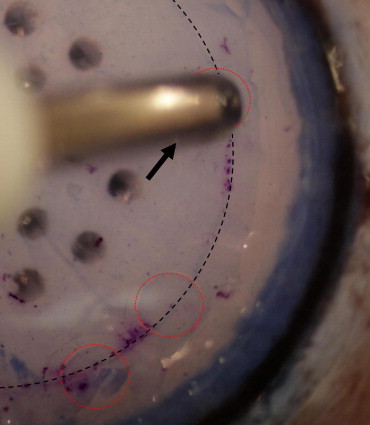
Corneoscleral buttons stored either in Optisol-GS (Bausch & Lomb Surgical, Irvine, California, USA) at 4 C or in Dulbecco’s Modified Eagle Medium containing streptomycin, penicillin (Biochrom, Berlin, Germany), and fetal calf serum (Linaris, Bettingen am Main, Germany) at 34 C were used for transplantation. Immediately before transplantation, an 8-mm endothelium–Descemet membrane layer graft was stripped from the corneal stroma. The peripheral endothelium–Descemet membrane layer was excised with a razor blade and the margin was lifted with a forceps. For identification of anterior-posterior orientation, a 1-mm trephine was used to excise 3 semicircles at the margin of the endothelium–Descemet membrane layer (2 close to one another and 1 with a recognizable distance; Figure 1 ). Then, stripping of the endothelium–Descemet membrane layer was completed, leading to an endothelium–Descemet membrane graft with 3 marks at the edge. The graft then was stained with a 0.06% trypan blue solution (VisionBlue; D.O.R.C. International, Zuidland, Netherlands) and was transferred into an injector cartridge (Acri.Tec GmbH, Hennigsdorf, Germany) that normally is used for intraocular lens delivery during cataract surgery. To allow easy unfolding, the marks were positioned near the opening of the Descemet roll and orientation of the roll in the shooter was chosen in a way that the marks entered the AC last ( Supplemental Video available at AJO.com ). Visibility of the marks thus is good, because the 12-o’clock position is nearest to the surgeon at work.
Surgical Technique
The recipient’s Descemet membrane was removed under air within the central 8 mm using an inverted hook. The graft then was inserted into the AC through a 3.5-mm corneal tunnel by use of the injector moved to a centered position, unfolded, and attached to the recipient stroma using weak phosphate-buffered saline jets and air bubble injections. The AC then was filled completely with air for 30 minutes. Afterward, the air was reduced to less than 20% of the AC volume. Postoperative endothelial cell density was analyzed using the SeaEagle Specular Microscope (RHINE-TEC, Krefeld, Germany).
Results
Herein, we report on 25 patients (14 male, 11 female; mean age ± standard deviation, 67 ± 9 years) who underwent DMEK for Fuchs endothelial dystrophy (22 patients) or pseudophakic bullous keratopathy (3 patients). In 7 patients, the DMEK was combined with phacoemulsification and intraocular lens implantation.
In all patients, the preparation of the donor endothelium–Descemet membrane layer was uneventful, and no tears in the graft occurred. However, 3 out of 25 stripped endothelium–Descemet membrane layers did not spontaneously form a roll. These endothelium–Descemet membrane layers had to be rolled manually using gentle jets of fluid and manipulation with a blunt spatula. In these cases, the marks were important to control the orientation of the endothelium–Descemet membrane layer when immersed after stripping.
Because most endothelium–Descemet membrane layers, especially from organ cultured grafts, exhibited very poor staining with trypan blue, the orientation marks were helpful to ensure correct orientation of the graft in the shooter. On insertion of the graft, the orientation marks were visible clearly and allowed correct positioning of the endothelium–Descemet membrane layer ( Figure 2 ). In cases where visualization of the marks was impaired, for example, by significant corneal edema, a second staining with trypan blue was performed in the AC.




