Purpose
To report and analyze long-term best-corrected visual acuity (BCVA) outcomes following a free autologous retinal pigment epithelium (RPE)-choroid graft translocation in patients with exudative age-related macular degeneration (AMD).
Design
Prospective cohort study.
Methods
setting: Institutional. study population: One hundred and thirty consecutive patients (133 eyes) with AMD underwent RPE-choroid graft translocation between October 2001 and February 2006. All patients had a subfoveal choroidal neovascular membrane with or without hemorrhage and/or an RPE tear. All were either ineligible for or nonresponsive to photodynamic therapy, the standard treatment at the time of surgery. observation procedures: Data collection included preoperative and postoperative visual acuity measurements, fundus photography, fluorescein and indocyanine green angiography, and microperimetry. main outcome measures: Postoperative BCVA.
Results
The mean preoperative BCVA was 20/250. Four years after surgery, 15% of the eyes had a BCVA of >20/200, and 5% had a BCVA of ≥20/40. One patient achieved a BCVA of 20/32, which was maintained at 7 years after surgery. Complications consisted of proliferative vitreoretinopathy (n = 13), recurrent neovascularization (n = 13), and hypotony (n = 2).
Conclusions
RPE-choroid graft transplantation may maintain macular function for up to 7 years after surgery, with relatively low complication and recurrence rates. Retinal sensitivity, BCVA data, and fixation on the graft suggest that the graft, rather than simply the removal of submacular hemorrhage and/or choroidal neovascular membrane, was responsible for the preservation of macular function. This surgery may be an alternative for patients with AMD who cannot undergo other standard treatment.
Age-related macular degeneration (AMD) is the third-leading cause of blindness globally and the leading cause of irreversible legal blindness in elderly people in industrialized countries. The current standard treatments for patients with exudative AMD are anti–vascular endothelial growth factor (anti-VEGF) injections, photodynamic therapy (PDT), or a combination of both modalities. However, anti-VEGF treatment might not be effective in restoring or improving visual acuity (VA) in patients with a retinal pigment epithelium (RPE) tear or a large submacular hemorrhage and in a small subset of patients with exudative AMD who fail to respond to anti-VEGF treatment. As potential alternative treatments for these patients, several surgical treatment modalities have been described, 4 of which are discussed below.
The first method, removal of the submacular choroidal neovascular membrane and/or hemorrhage, was studied in the Submacular Surgery Trials (SST). In the subgroup of patients in whom the hemorrhage was surgically removed, the percentage of patients who lost more than 6 lines of VA was statistically smaller compared to untreated controls. A second method is the displacement of an acute hemorrhage by gas and recombinant tissue plasminogen activator, with or without vitrectomy, and with subsequent treatment with PDT or anti-VEGF. Several case series suggest a benefit over natural course. The third method, macular translocation after 360-degree retinectomy, was first reported by Machemer and Steinhorst in 1993. This method is associated with several vitreoretinal complications such as a macular pucker, retinal detachment (RD), proliferative vitreoretinopathy (PVR), recurrence of choroidal neovascularization (CNV), choroidal hemorrhage, a tilted image, and diplopia. Despite an appreciable complication rate, stabilization or improvement of VA of this surgical technique has been demonstrated, with long-term (14−79 months) beneficial results in some patients with exudative AMD. Further, this surgical technique demonstrated superior 2-year VA results to PDT in a randomized controlled trial. The fourth method is the transplantation of an autologous graft of RPE, Bruch membrane, choriocapillaris, and choroid. This method was first described by Peyman and associates in 1991. In early studies, the graft was harvested from the edge of the macular RPE defect; later studies used tissue from the mid-peripheral retina. The most frequent complications of transplantation are recurrent CNV, RD, PVR, non-revascularization of the graft, macular pucker, and postoperative hemorrhages.
RPE and choroid graft surgery has been compared with macular translocation surgery. Both techniques seem to lead to better VA outcomes than no intervention. However, although macular translocation surgery might produce better VA outcomes than RPE and choroid graft surgery, it has a greater risk of complications. Moreover, there is a physical limit to the amount of translocation possible (even after 360-degree retinectomy), which makes it less suitable for large CNVs. Several centers have reported case series of free RPE and choroid grafts, albeit with relatively short follow-up periods. MacLaren and associates have presented patients with up to 6 years of follow-up, although this applied to only 4 of the 9 patients initially included.
The current study analyzes the primary outcome, defined as best-corrected visual acuity (BCVA), of a large group of 130 consecutive patients (133 eyes) who underwent a free RPE and choroid graft procedure. The follow-up period of these patients varies between 1 and 7 years. In addition, for some of these patients, BCVA results were correlated with macular sensitivity as measured by microperimetry.
Patients and Methods
At the Rotterdam Eye Hospital, RPE-choroid graft surgery was first performed in October 2001. As of February 2, 2010, 130 consecutive patients (133 eyes) had been included in this institutional prospective cohort study and had undergone this surgery before February 2, 2006. This resulted in registration of follow-up data of at least 4 years. The main outcome measure of this institutional prospective cohort study was postoperative BCVA up to 7 years after surgery. The inclusion criteria for RPE-choroid graft surgery were exudative AMD patients with a VA <20/80 (>0.6 logMAR [logarithm of minimal angle of resolution]) and a subfoveal choroidal neovascular membrane. The patients were not eligible for PDT, as they had a minimally classic lesion larger than 4 disc diameters and/or 1 of the following findings: a large hemorrhage, an RPE tear, or a 3-line VA loss after PDT. Exclusion criteria were underlying diseases other than AMD or anticoagulant drugs that could not be discontinued. All patients were operated before anti-VEGF therapy was routinely available at the Rotterdam Eye Hospital in February 2006.
Surgery
After the induction of a posterior vitreous detachment, a complete vitrectomy was performed. The macular retina was separated from the RPE and/or blood and/or CNV membrane by injecting a balanced salt solution into the subretinal space through a 28-gauge subretinal cannula . A paramacular temporal retinotomy was made, through which the CNV membrane and/or subretinal hemorrhage was removed from the subretinal space with Thomas subretinal forceps. After circular heavy diathermy in the midperiphery at the 6- or 12-o’clock position, vitreous scissors were used to cut a full-thickness graft of retina, RPE, Bruch membrane, and choroid of approximately 2 to 3 × 2 to 3 mm. Initially, the graft was loaded onto an aspiration-reflux spatula (Dutch Ophthalmic Research Center [DORC], Zuidland, The Netherlands), as described by Maaijwee and associates. Later on, the graft was grasped from the choroidal side by fine forceps. The retina was removed from the graft and was repositioned underneath the macula through the existing paramacular retinotomy. Perfluorocarbon liquid was injected over the macula to hold the graft in position during retraction of the instrument. A vibration device attached to the forceps facilitated the release of the graft. The midperipheral donor site was then encircled by laser photocoagulation, followed by intraocular tamponade with silicone oil.
In a second procedure, approximately 3 months later, the silicone oil was removed and the inner limiting membrane was peeled. This peeling was performed to prevent formation of a macular pucker. In phakic patients, lensectomy and intraocular lens (IOL) insertion was performed during either the first or second surgery. All surgical procedures were performed by 1 surgeon (J.v.M.).
Grading of Preoperative Images
Masked readers (H.H. and T.M.) independently assessed the preoperative color fundus photographs, fluorescein angiograms (FA), indocyanine green angiograms (ICGA), and red-free images of all 133 eyes. The images were imported into image analysis software (ImageJ; http://rsbweb.nih.gov/ij/ , last accessed April 21, 2010; National Institutes of Health, Bethesda, Maryland, USA) The fundus, FA, and ICGA images were used to determine disc area, lesion size (all lesion components taken together, measured in relation to disc area), composition, and size of hemorrhage (in disc area and % of total lesion size). If FA was not available, ICGA images were used in combination with red-free and/or color images for classification. The patients were then classified into 3 groups by lesion composition (predominantly classic, minimally classic, or occult), according to the Macular Photocoagulation Study (MPS) protocol. A fourth group, labeled “hemorrhagic lesion,” included all lesions masked for >50% by extensive hemorrhage.
Ophthalmic Examination and Follow-Up
Preoperative examination included BCVA in Snellen and logMAR equivalents, dilated fundoscopy, FA, and/or ICGA. Postoperative visits were scheduled at 1, 3, and 6 weeks; at 3, 6, 9, 12, and 18 months; and subsequently every year. During each visit, BCVA testing was performed. Visual acuity data was converted to logMAR for calculations and statistical analysis. The visual acuity data were reported as the percentage of patients with a BCVA of either >20/200 or ≥20/40.
Microperimetry
After surgery, 2 subgroups of patients were tested with the microperimeter (MP-1; Nidek Technologies, Padova, Italy) with an automatic eye tracker compensating for eye movements. Because of the inconsistent availability of the MP-1, not every patient could be tested. The first group consisted of 19 patients who were tested shortly after surgery. The second subgroup consisted of 10 patients who were tested at least 4 years after surgery. MP-1 software version 1.7.3 was used during the second period. Pupils were dilated with tropicamide 0.5% (Chauvin Pharmaceuticals Ltd, Kingston-Upon-Thames, United Kingdom) and phenylephrine 2.5% (Chauvin Pharmaceuticals Ltd). The stimulus size was Goldmann III with white stimuli of 200 ms projection time. A customized Cartesian pattern centered on the graft, with a staircase 4−2 threshold strategy, was tested automatically. The brightness of the test stimuli ranged from 0 to 20 decibels (400 to 4 asb). After performing these tests, a color image of the fundus was made. The infrared images of the microperimetry tests were matched to the color image using reference points, to display the tests on the color fundus image.
Results
Baseline Characteristics
In total, 130 consecutive patients (78 female, 52 male; 133 eyes) underwent RPE-choroid graft surgery. Three patients were operated in both eyes. The mean age was 79 ± 8 years (range: 50−95). The 60 patients who were on anticoagulant therapy before surgery received permission to discontinue this medication 2 weeks prior to surgery. Before surgery, 87 eyes were phakic and 46 were pseudophakic. The median duration of VA loss prior to surgery, as reported by the patients, was 61 days, with a range of 7 days to 3 years. The median duration of VA loss in patients with a hemorrhagic lesion was 39 days, ranging from 2 days to 183 days. However, in the few patients with a reported VA loss of ≤4 weeks, this loss was attributable to an aggravation of a pre-existing hemorrhagic lesion. Nine patients had undergone Argon laser therapy and 7 had undergone PDT before surgery.
Follow-Up
One-year follow-up BCVA data were available for 130 out of 133 eyes, 2-year data were available for 101 eyes, 3-year data for 72 eyes, 4-year data for 46 eyes, 5-year data for 27 eyes, 6-year data for 10 eyes, and 7-year data were available for 9 eyes ( Table 1 ).
| Years After Surgery | Number of Eyes | Median VA (logMAR) | Range (logMAR) | VA <1.0 logMAR (>20/200) (No. Eyes) |
|---|---|---|---|---|
| Preoperative | 133 | 1.00 | 0.40−2.20 | 52 |
| 1 year | 130 | 1.16 | 0.20−2.48 | 46 |
| 2 years | 101 | 1.30 | 0.30−2.64 | 32 |
| 3 years | 72 | 1.30 | 0.10−2.90 | 19 |
| 4 years | 46 | 1.10 | 0.20−2.90 | 20 |
| 5 years | 27 | 1.30 | 0.20−2.77 | 7 |
| 6 years | 10 | 1.20 | 0.40−1.78 | 4 |
| 7 years | 9 | 1.30 | 0.20−2.48 | 4 |
In total, during 7 years of follow-up, 24 patients were lost to follow-up with a last BCVA measurement of >20/200. Nine of these patients died, 2 patients were physically/mentally unable to come, 1 wished not to return, 1 patient did now show up for unknown reasons, and 11 patients did not yet reach the next-year follow-up visit (of 5 or 6 years after surgery). Of all 130 patients, 17 patients were lost to follow-up because they died from unrelated causes. Others did not return for follow-up because they were physically and/or mentally unable to do so, or for unknown reasons.
Grading of Preoperative Images
Of the 133 eyes, 108 were available and eligible for grading, as the image quality was insufficient to allow grading in 25 eyes. Ninety eyes were graded with fundus images and FA. For 18 eyes no FA was available and grading was carried out based on ICGA, color fundus pictures, and/or red-free images. According to MPS criteria, 16 patients had a minimally classic lesion, 11 had a predominantly classic lesion, and 35 had an occult subfoveal neovascularization. Forty-six of the eyes were classified as having a hemorrhagic lesion (>50% blood). Subretinal hemorrhage was present in 99 of the 108 eyes. The median size of the total lesion in disc areas was 6.22, ranging from 1.11 to 73.92 disc areas. The median size of the hemorrhage was 3.76 disc areas, ranging from 0.03 to 73.92 disc areas.
All patients had a subfoveal neovascular membrane before surgery, of which 122 contained blood and 11 did not. Three patients of the 133 had an RPE tear, 1 with and the other 2 without blood. The 11 patients without blood were 5 patients with an occult CNV, 2 patients with minimally classic CNV, 2 with predominantly classic CNV, and 2 patients with an RPE tear.
Visual Acuity
The median preoperative BCVA of all 133 eyes (130 patients) was 1. 10 logMAR (20/250). Table 1 shows the median BCVA of the eyes for each postoperative year with their range, while Figure 1 shows the percentage of patients with a BCVA of either ≤20/200 or >20/200. Three groups are depicted in this figure. Group 1 consists of the percentage of eyes with a BCVA >20/200 at each time point. Group 2 consists of the percentage of eyes with a BCVA of ≤20/200 at the moment the patients were lost to follow-up. Group 3 consists of eyes lost to follow-up and whose last measured BCVA was >20/200. Only in Figure 1 has the last measured data of this third group been carried forward.
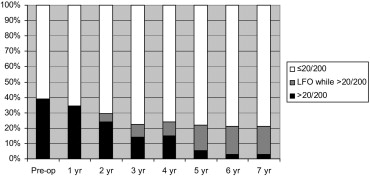
The results were divided into 2 patient groups ( Table 2 ); AMD patients with surgery for either predominantly classic, minimally classic, or occult neovascular lesions (group N), and AMD patients with surgery for hemorrhagic (>50% blood) lesions (group B). While no patients in either group N or B had a VA of ≥20/40 at baseline, 3 (3.2%) of the patients in group N, and 9 (8.7%) in group B, had a VA of ≥20/40, 4 years after surgery. Nineteen percent of the patients in group N and 13% of the patients in group B had, 4 years after surgery, a VA of >20/200.
| BCVA ≥20/40 | BCVA ≥20/40 | BCVA >20/200 | BCVA >20/200 | |
|---|---|---|---|---|
| Group N a (n c = 62) | Group B b (n = 46) | Group N (n = 62) | Group B (n = 46) | |
| Baseline | 0% | 0% | 50% | 37% |
| 1 year | 4.8% | 4.3% | 45% | 28% |
| 2 years | 4.8% | 4.3% | 34% | 22% |
| 3 years | 1.6% | 4.3% | 18% | 15% |
| 4 years | 3.2% | 8.7% | 19% | 13% |
a Group N: Surgery for subfoveal choroidal neovascularization, either minimally classic, predominantly classic, or occult, in age-related macular degeneration.
b Group B: Surgery for hemorrhagic (>50% blood) choroidal neovascular lesions of age-related macular degeneration.
Of all 133 operated eyes, 20 (15%) had a BCVA of >20/200 after 4 years. Six of these 20 patients had a BCVA of ≥20/40, which is 5% (6/133) of the total operated eyes. Their median BCVA was 0.56 logMAR (20/73), and the highest measured BCVA was 0.2 logMAR (20/32). Nine patients had a follow-up period of 7 years. Four of them had a BCVA of >20/200, with a median of 0.55 logMAR (20/71) and a highest measured BCVA of 0.20 logMAR (20/32).
Visual Acuity Course of a Subgroup
We further examined the VA course of the subgroup of 20 patients whose BCVA was >20/200 at 4 years after surgery. The median BCVA of these 20 patients was, at 3 months after surgery, 20/125 (0.80 logMAR, range 0.30−1.53 logMAR); at 6 months, 20/80 (0.6 logMAR, range 0.30−1.70 logMAR); at 1 year, 20/89 (0.65 logMAR, range 0.20−2.10 logMAR); and at 4 years after surgery, 20/73 (0.56 logMAR, range 0.20−0.90 logMAR). In 11 of these 20 patients an improvement in BCVA ranging from 1 to 7 Early Treatment Diabetic Retinopathy Study (ETDRS) lines was found between 3 and 6 months after surgery. After this initial increase, BCVA stabilized up to 4 years after surgery. Seven of these 20 patients, however, had a slowly and only slightly increasing BCVA, starting 3 to 6 months after surgery, up to 4 years after surgery. Two of these 20 patients had yet another visual acuity course: 1 had a slow but progressive increase of 7 ETDRS lines between 3 months and 4 years after surgery; the other had a more or less sudden increase of 6 lines at 3 years and an improvement of 2 more lines 1 year later.
In 17 these 20 patients (85%), fixation on the graft was documented. In 15 of these 20 patients the natural lens was removed and replaced in first surgery (n = 9) or at time of silicone oil removal (n = 6). In 6 of these patients a BCVA improvement was seen after IOL insertion at the time of silicone oil removal, which might also be attributed to this IOL insertion, but subsequent further improvement of BCVA was seen in 3 of them.
One year after surgery, 46 patients had a BCVA of >20/200. As described above, 20 of those preserved this BCVA up to 4 years after surgery. Eighty-seven patients had a BCVA of ≤20/200 1 year after surgery. Five of these 87 patients’ BCVA improved only temporarily to >20/200 during the follow-up period of the study.
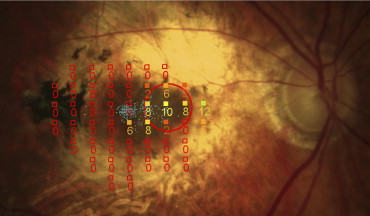
Microperimetry
Microperimetry data was available for a subgroup of 10 patients who were tested at least 4 years after surgery. Two of these patients were also included in the first subgroup of 19 patients who were tested shortly after surgery. The patient shown in Figure 2 was examined 8 months and 7 years after surgery. The preoperative BCVA was 20/200 (1.0 logMAR). At 8 months after surgery the BCVA was 20/80 (0.6 logMAR) and 7 years after surgery it was 20/32 (0.2 logMAR). Figures 3 and 4 show 2 patients who completed only 1 postoperative microperimetric evaluation. The first had a preoperative BCVA of 20/800 (1.6 logMAR) and a VA of 20/80 (0.6 logMAR) after 4 years and 10 months ( Figure 3 ). The second patient had a preoperative BCVA of 20/2400 (2.1 logMAR) and a VA of 20/50 (0.4 logMAR) after 6 years and 7 months ( Figure 4 ).