FIGURE 53.1 Persistent anterior TVL vessels. These vessels are persistent and not newly developed, and therefore are best referred to as rubeosis iridis.
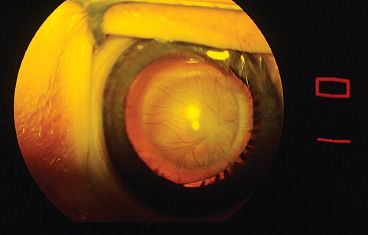
FIGURE 53.2 Vessels extending from the lens surface to the ciliary processes. These vessels can be divided with little bleeding after the central vessel is treated with diathermy.
PFVS has a spectrum of presentations depending on the degree of involution of the hyaloid (see also Chapters 1 and 2) and TVL. PFVS can be associated with varying amounts of retinal dysplasia, which frequently is the rate-limiting step for postoperative vision beyond that from anisometropic aphakic amblyopia following lens removal in this predominantly unilateral process.
PFVS can present with predominant features of the TVL without much or any posterior hyaloid component (Fig. 53.3) (4). Alternatively, the hyaloid vasculature can persist with little in the way of regressed TVL (Fig. 53.4). Eyes with predominantly hyaloid changes often are referred to as posterior PFVS and those with predominantly tunica vasculosa changes as anterior PFVS.
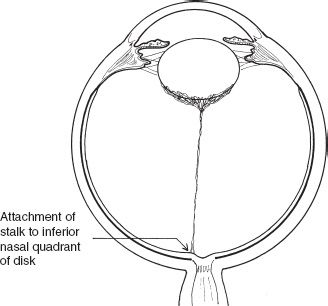
FIGURE 53.3 Illustration of the opaque posterior lens tissue, elongated ciliary processes, and fine stalk extending to the disc. This is predominantly anterior PFV syndrome.
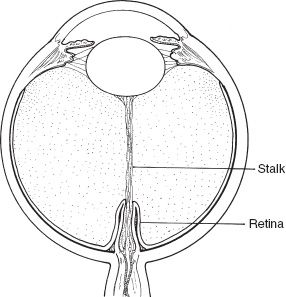
FIGURE 53.4 Illustration of the stalk attachment to the lens, obstructing the visual axis. Retina is draped around the stalk or hyaloid tissue. Bending the stalk can pinch retinal vessels.
Although PFVS has been recognized for many years, a single gene for PFVS has not been identified. Certainly, the PFVS process, which involves an interrupted genetically programmed involution of embryonic vessels and the assembly of the lens and retinal tissue, is considered likely to have a genetic etiology. A six-generation consanguineous Pakistani family showing linkage to chromosome 10q11–q21 has been reported (5). The abnormal gene in this family has not yet been found, and whether or not it will explain other PFVS cases remains to be determined. Additionally, mutations in the PAX6 gene were detected recently in patients with optic nerve malformations that included PFVS (6).
When bilateral PFVS is present, Norrie disease must be ruled out (7). Although Norrie disease is commonly thought to mimic persistent fetal vascular syndrome, it often has a more severe hemorrhagic and dysplastic retinal detachment. In some circumstances of bilateral PFV (<27%), hearing or central nervous system abnormalities are present (8,9). Other systemic associations have been reported with PFVS, most often in association with central nervous system problems (10). However, many of these reports do not rule out mutations in the Norrie disease gene. Associations have also been reported with oculo–palatal–cerebral syndrome (11), intrauterine herpes simplex virus infection (12), intrauterine exposure to clomiphene, oral–facial–digital syndrome, anterior and posterior colobomas or even cystic globes (13,14), and tuberous sclerosis (15). Another recently discovered syndrome associated with PFVS is MPPC (microcornea, posterior lenticonus, persistent fetal vasculature, and coloboma).
PFVS is customarily described as the involved eye being a smaller eye with a posterior lens opacity, elongated ciliary processes, and, if visible, a stalk that extends from the lens posterior to the optic disc (Fig. 53.5). There is variability in clinical presentation. In our experience, the severity of anterior segment appearance, globe size, severity of lens involvement, or vascular appearance does not predict the amount of retinal dysplasia, which in turn often determines the eye’s visual result.
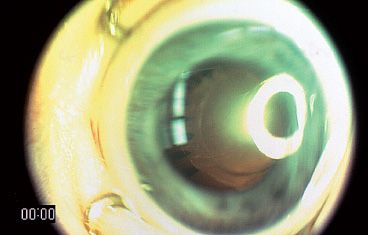
FIGURE 53.5 Typical PFV syndrome with a smaller eye, elongated ciliary processes, and media opacity.
Ultrasound, computed tomographic scanning, magnetic resonance imaging, and visual evoked potential testing have not been reliable in predicting retinal dysplasia. However, of these modalities, visual evoked potential testing is perhaps the most helpful (16,17). If a response is present, then it can be helpful in the decision whether to recommend surgery for a retinal detachment. This is most helpful when the other eye is uninvolved, and the waveforms can be compared.
The problem of PFVS can be divided into two components that affect vision: the media opacity and retinal changes (both dysplasia and traction). The stalk also can cause traction on the posterior lens capsule leading to posterior lenticonus (Fig. 53.6). In addition, traction on the ciliary body can lead to hypotony.
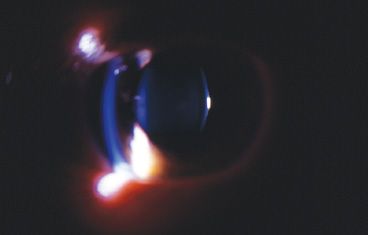
FIGURE 53.6 Slitlamp photograph showing the bowing of the posterior lens surface in the child with an eccentric stalk who did not have surgery as an infant and developed posterior lenticonus.
Media opacities are addressed similarly to congenital cataract and, for the most part, are handled by lens removal, refraction, and amblyopia therapy (18). The tractional effects and retinal dysplastic features are not as well understood. The stalk itself can be a simple column-like structure that extends from the lens to the optic nerve or posterior retina or can be an inverted Y shape with a second or even third arm that attaches to the disc and elsewhere on the retinal surface. The stalk can exert traction on the retina, leading to areas of tractional retinal detachment. The stalk can insert anteriorly on the lens either centrally, involving the visual axis, or eccentrically, sparing the visual axis of the lens. If the stalk is eccentric, no change in visual acuity is noticed in the very young child, but strabismus can result. If there is a visual axis opacity, the problem usually is discovered at the newborn screening. The child with a stalk eccentric to the visual axis often presents with strabismus later in life, at age 9 to 10 months (19).
The tractional component on the lens is perhaps even less well known. The eccentric stalk that leaves the visual axis clear does not always need surgical intervention. Tractional effects on the lens or retina determine whether surgical intervention with division of the stalk is necessary. Retinoscopy, giving a scissors-like reflex, can help to identify progressive posterior lenticonus as a result of the traction of the fixed stalk on the posterior lens surface during eye growth.
The traction on the ciliary body is primarily a function of the TVL and must be relieved in order to avoid one of the complications of the long-term PFVS, hypotony, which we believe is due to prolonged ciliary body traction resulting in either ciliary process damage or ciliary body detachment. In addition, several forms of glaucoma can occur in some cases of PFVS.
Retinal dysplasia has a wide range of clinical presentations and can involve the retinal circulation and the cellular retina. We divide retinal dysplasia into macroscopic and microscopic types. Macroscopic dysplasia is defined as changes easily visible with the operating microscope or indirect ophthalmoscope. Microscopic dysplasia occurs at the cellular or vascular level. Although at present we have no way to determine cell dysplasia, vascular dysplasia can be assessed by fluorescein angiography. An example of vascular dysplasia is the lack of a capillary-free zone. This finding can be missed without angiography and might account for some of the poor vision after successful surgery. It is not unusual for a surgeon to anticipate good vision after surgery for PFVS based on the clinical appearance of the retina. After a reasonable course of amblyopia therapy, the doctor might find that vision better than 20/100 is not possible. The converse also can be true: that eyes with unusual appearing posterior poles can sometimes achieve better vision. Recently, we found that alterations in the foveal circulation detected by fluorescein angiography predicted foveal hypoplasia and poor central visual acuity. However, such changes may not always be detectable. We use fluorescein angiography and a full examination under anesthesia to determine if the foveal structure is good enough to merit continued amblyopia therapy. If the child is older, optical coherence tomographic testing can be performed to further define foveal architecture and foveal thickening speaking to a microscopic form of foveal dysplasia.
The surgeon should be aware that intrinsic retinal vessels can be pulled onto the stalk tissue and occasionally can be dragged one to two-thirds the length of the stalk toward the lens. In addition, retinal tissue can cloak the stalk, giving it more substance. This requires great care to divide only stalk tissue, not retina or intrinsic retinal vessels. This could lead to a retinal hole or bleeding, followed by a retinal ischemic event (Fig. 53.7).
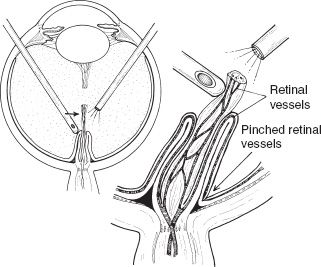
FIGURE 53.7 Illustration of moving the stalk tissue to determine if intrinsic retinal circulation is in the stalk tissue. If moving the stalk changes the retinal vascular flow, then the surgical goal is to only minimally dissect the stalk.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


