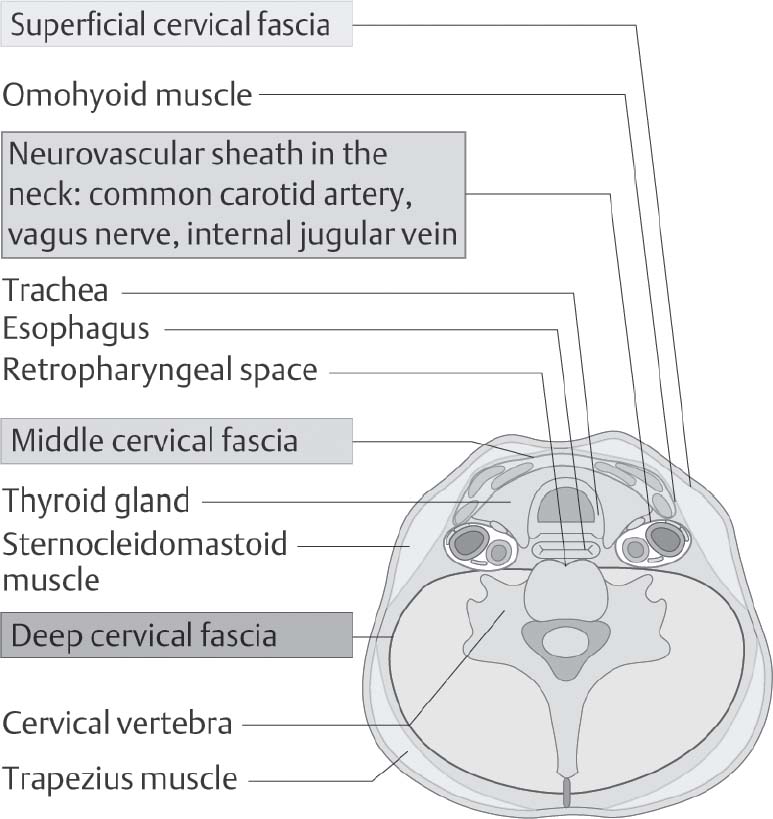
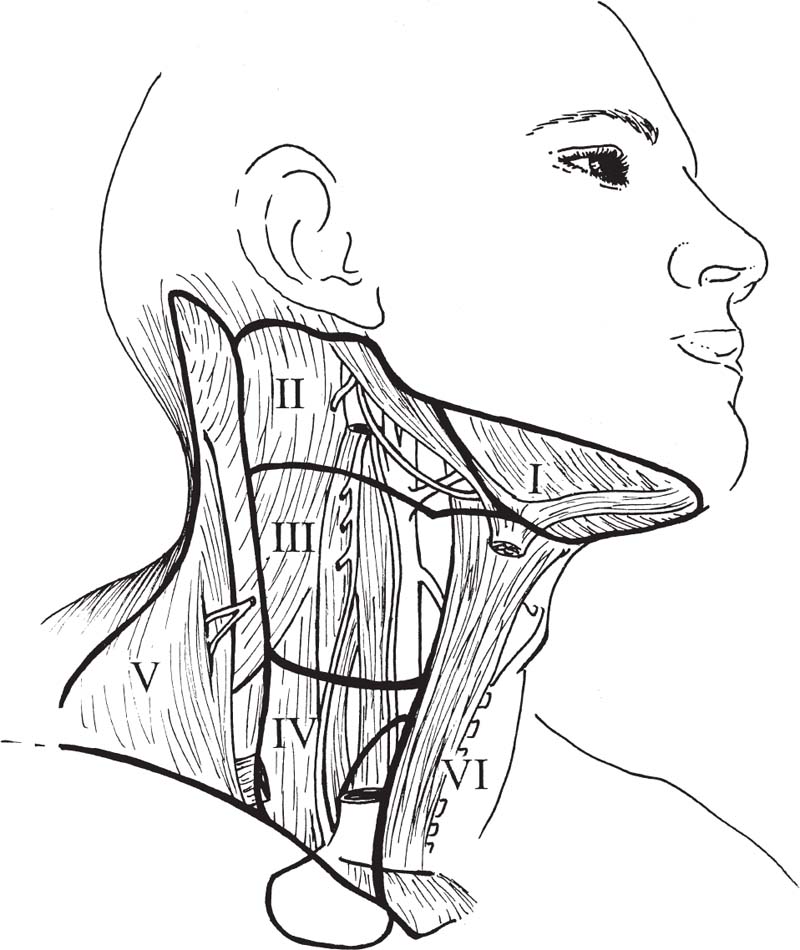
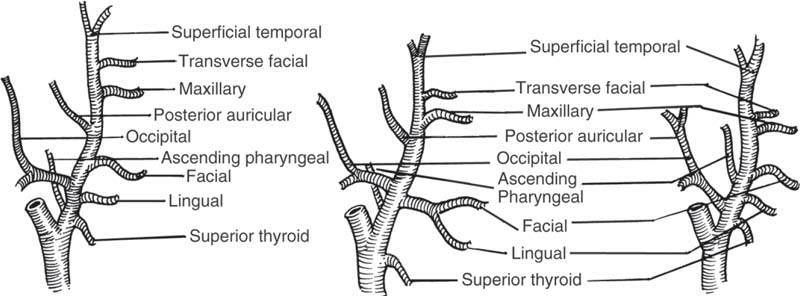
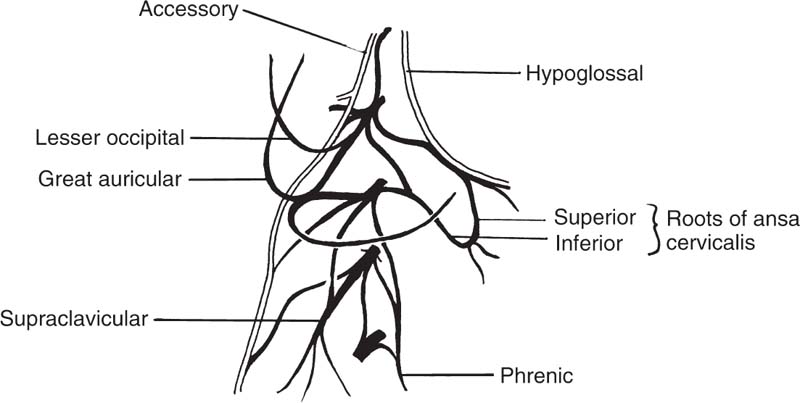
5 Head and Neck Section Editors Contributors Surgically, it is useful to consider the neck anatomy in terms of compartments or levels and to consider fascial layers. The neck is covered by a layer of superficial fascia, and the deep cervical fascia divides into superficial, middle, and deep layers (Fig. 5.1). This is relevant in terms of spread of infection and lymphatic spread of cancer. The visceral structures of the neck are included in the central compartment and include the laryngotracheal complex, the thyroid and parathyroids, and the cervical esophagus. Posteriorly, of course, are the cervical spine and paraspinal muscles. Anterolaterally, the neck contents is considered in terms of nodal levels I though VI (Fig. 5.2). Each level contains a compartment of fibrofatty lymph node-bearing tissue that is addressed for removal during neck dissection procedures for cancer. Level I is the submental region and the submandibular area containing the submandibular gland. Level II is the upper jugular chain lymph node region, level III is the middle jugular chain, and level IV is the inferior jugular chain region. Level V is the posterior triangle from the anterior border of the trapezius to the posterior border of the sternocleidomastoid muscle. Level VI is the node-bearing tissue of the central compartment. Fig. 5.1 Cervical fascial planes. (From Probst R, Grevers G, Iro H. Basic Otorhinolaryngology: A Step-by-Step Learning Guide. Stuttgart/New York: Thieme; 2006:313.) Fig. 5.2 Nodal levels I through VI. (From Van de Water TR, Staecker H. Otolaryngology: Basic Science and Clinical Review. Stuttgart/New York: Thieme; 2006:606.) The common carotid artery ascends into the neck and bifurcates into the internal and external carotid artery. The internal carotid has no branches in the neck and delivers important blood supply to the brain, along with the vertebral arteries. The external carotid supplies the head and neck structures via multiple branches (in order, starting inferiorly): the superior thyroid, ascending pharyngeal, lingual, facial, occipital, posterior auricular, superficial temporal, and maxillary arteries (Fig. 5.3). Venous drainage tends to parallel major arteries, with variable anterior and external jugular branches superficially, and large common facial veins joining into the internal jugular vein, which feeds the subclavians. The carotid sheath, formed by all three layers of the deep cervical fascia, contains the carotid artery, jugular vein, and the vagus nerve. Important delicate lymphatics from the thoracic duct carry chyle into the jugular vein near its junction with the subclavian in the left neck; however, accessory thoracic ducts commonly occur on the right. Fig. 5.3 The branching of the arteries originating from the carotid artery can be quite variable. (From Van de Water TR, Staecker H. Otolaryngology: Basic Science and Clinical Review. Stuttgart/New York: Thieme, 2006:603.) The 12 cranial nerves (CNs) supply the head and neck region with motor, sensory, and special sensory fibers. They are, in order, the olfactory, optic, oculomotor, trochlear, trigeminal, abducens, facial, vestibulocochlear, glossopharyngeal, vagus, spinal accessory, and hypoglossal. In the neck there are surgically important aspects of CN anatomy. The hypoglossal emerges from the skull base at the hypoglossal canal and courses beneath the digastric and submandibular contents as motor supply to the tongue. The pars nervosa of the jugular foramen contains the glossopharyngeal, vagus, and accessory nerves. The vagus runs within the carotid sheath and the recurrent laryngeal branches course in the tracheoesophageal groove to supply the vocal folds; the superior laryngeal nerves arise from the vagus in the upper neck. The accessory nerve courses posteroinferiorly through the neck entering the anterior border of the sternocleidomastoid, emerging from the posterior border near Erb’s point, and continuing into the trapezius. The facial nerve exits the skull base at the stylomastoid foramen and its main motor branches course within the parotid gland to innervate the muscles of facial expression, while the lingual nerve supplies parasympathetics to the submandibular gland and sensation to the anterior tongue and is encountered deep to the submandibular gland. Important cervical nerves are encountered in the neck as well. The great auricular nerve (it is not named great er, since there is no lesser auricular nerve) transmits sensory fibers of C2–3 to the auricular area (Fig. 5.4). This nerve is encountered along the sternocleidomastoid coursing superiorly from Erb’s point. The phrenic nerve is deep to the floor fascia along the anterior scalene muscle, supplying motor fibers to the diaphragm. Also, the brachial plexus is in the same plane deep to the fascia on the middle scalene. The sympathetic trunk runs deep to the carotid. CN anatomy is reviewed in Appendix B of this book. Fig. 5.4 Cervical plexus. (From Van de Water TR, Staecker H. Otolaryngology: Basic Science and Clinical Review. Stuttgart/New York: Thieme; 2006:603.) Schuenke M, Schulte E, Schumacher U. Thieme Atlas of Anatomy: Neck and Internal Organs. Stuttgart/New York: Thieme; 2006 Smith RV, Frenz D. Surgical anatomy of the neck and classification of dissections. In: Van de Water TR, Staecker H, eds. Otolaryngology: Basic Science and Clinical Review. Stuttgart/New York: Thieme; 2006:598–609 • Necrotizing fasciitis is a soft tissue infection that causes necrosis of fascia and subcutaneous tissue, but initially spares skin and muscle. • It is rare in the head and neck. • Half of patients develop systemic bacteremia. • Necrotizing soft tissue infections require aggressive treatment to combat the associated high morbidity and mortality. Necrotizing soft tissue infections rarely may involve the face and neck, scalp, and eyelids. In the head and neck, dental infections are the most common etiology, followed by trauma, peritonsillar and pharyngeal abscesses, and osteoradionecrosis. Immunocompromised patients are most susceptible. Predisposing conditions include diabetes mellitus, obesity, arteriosclerosis, alcoholism, chronic renal failure, hypothyroidism, malignancy, and poor nutrition. Gas-producing wound infections are usually produced by bacteria of the Clostridia group. Onset of symptoms is usually 2 to 4 days after the insult. The signs of a necrotizing soft tissue infection include a low-grade fever, and the skin becomes smooth, warm, tense and shiny with no sharp demarcation. The infected skin also develops a dusky discoloration with poorly defined borders. Soft tissue crepitus is common from gas formation. Later in the disease process, bullae may develop. Systemic symptoms get more severe with typical signs of septicemia. The last stage of the disease is characterized by cyanotic skin discoloration typical of necrosis. There is sudden severe pain and swelling. There may be anesthesia of the involved skin. The differential diagnosis includes other deep neck space infections, pyoderma gangrenosum, radiation necrosis, cellulitis, or erysipelas. When clinical hard signs are present (i.e., crepitus, skin necrosis, bullae, hypotension), a physical exam may be helpful. Unfortunately, the signs and symptoms are often very subtle on presentation. Plain soft tissue films of the neck looking for gas in soft tissue may be obtained. Computed tomography (CT) is the most useful study to detect gas in areas inaccessible to palpation and to identify areas where the infection has spread. In addition, it can detect vascular thrombosis, erosion of vessels, or mediastinitis. Routine blood work is needed to look for metabolic abnormalities such as hyponatremia and hypoproteinemia due to fluid sequestration and hypocalcemia as a result of subcutaneous fat saponification. Group A hemolytic streptococci and Staphylococcus aureus, alone or in synergism, are frequently the initiating infecting bacteria. However, other aerobic and anaerobic pathogens may be present, including Bacteroides, Clostridium, Peptostreptococcus, Enterobacteriaceae, coliforms, Proteus, Pseudomonas, and Klebsiella. The pathology shows a localized necrosis of skin that is secondary to thrombosis of nutrient vessels as they pass through involved fascia. Tissue hypoxia resulting from small vessel vasculitis and thrombi and impaired host defenses help facilitate anaerobic bacterial growth. Prompt measures offer hope for survival of the patient. High-dose intravenous (IV) broad spectrum antibiotics are recommended. Hyperbaric oxygen therapy is sometimes used in addition to surgical and antimicrobial treatment. Aggressive surgical débridement until there are bleeding viable borders is necessary, and sometimes repeated excisions to remove necrotic tissue. If the necrotizing process involves the neck, avoid tracheotomy through the infected area. The overall mortality rate is close to 70%.
5.1 Anatomy of the Neck
Blood Supply
Innervation
Further Reading
5.1.1 Neck Emergencies
5.1.1.1 Necrotizing Soft Tissue Infections of the Head and Neck
Key Features
Epidemiology
Clinical
Signs
Symptoms
Differential Diagnosis
Evaluation
Physical Exam
Imaging
Labs
Microbiology
Pathology
Treatment Options
Medical
Surgical
Outcome and Follow-Up
ICD-9 Codes
728.8.6 | Necrotizing fasciitis |
Use additional code to identify:
041.00–041.89 | Infectious organism |
785.4 | Gangrene, if applicable |
Further Reading
Beck HJ, Salassa JR, McCaffrey TV, Hermans PE. Life-threatening soft-tissue infections of the neck. Laryngoscope 1984;94(3):354–362
Goldenberg D, Golz A, Netzer A, Flax-Goldenberg R, Joachims HZ. Synergistic necrotizing cellulitis as a complication of peritonsillar abscess. Am J Otolaryngol 2001;22(6):415–419
Wenig BL, Shikowitz MJ, Abramson AL. Necrotizing fasciitis as a lethal complication of peritonsillar abscess. Laryngoscope 1984;94(12 Pt 1):1576–1579
5.1.1.2 Ludwig Angina
Key Features
• Ludwig angina is a rapidly expanding, diffuse inflammation of the submandibular and sublingual spaces.
• It is most often caused by dental infections.
• The condition is often found in immunocompromised patients such as those with diabetes or human immunodeficiency virus/acquired immunodeficiency syndrome (HIV/AIDS), or drug abusers.
Ludwig angina is a rapidly spreading bilateral cellulitis of the sublingual and submaxillary spaces. Before the advent of antibiotics, the mortality associated with Ludwig angina approached 50%. Today, mortality rates are in the range of 8 to 10%. The most common cause of death is respiratory compromise.
Epidemiology
Ludwig angina represents up to 13% of all deep neck infections. Typically, young adults present with Ludwig angina. It is unusual in children. The infection generally spreads from a dental or periodontal infection. Other causes include upper respiratory infections, floor-of-mouth trauma, mandibular fractures, and sialadenitis, IV drug abuse, trauma and tonsillitis, and immunocompromised states such as diabetes or HIV/AIDS. Forty percent of Ludwig angina cases involve anaerobes.
Clinical
Signs
The signs of the infection include an inability to close the mouth, trismus, drooling, sitting upright, inability to swallow and dysphonia, dyspnea, stridor, fever, chills, and tachycardia. Note that strider, dyspnea, decreased air movement, or cyanosis suggests impending respiratory compromise.
Symptoms
The symptoms of Ludwig angina are severe neck pain and tenderness, submandibular and submental swelling, fever, malaise, and dysphagia.
Differential Diagnosis
The differential diagnosis involves other deep neck space infections, an infected cyst, tumor, and cellulitis.
Evaluation
Physical Exam
On physical examination, the patient will often present with carious molar teeth, neck rigidity, and drooling. There is a “woody” or “brawny” induration of involved spaces with little to no fluctuance. The patient’s mouth floor is swollen, and the tongue is swollen or elevated.
Imaging
A CT scan is most useful.
Labs
Blood cultures are usually negative; if the swelling is aspirated or drained, samples should be sent for Gram stain, culture, and sensitivity.
Treatment Options
Airway control is the first priority of treatment, followed by IV antibio tics and timely surgical drainage. Blind oral or nasotracheal intubation or attempts with neuromuscular paralysis are contraindicated in Ludwig angina as they may precipitate an airway crisis.
Medical
Aggressive empiric high-dose, IV antibiotics are recommended—cefuroxime plus metronidazole. If the patient is allergic to penicillin, prescribe clindamycin plus a quinolone. Once culture and sensitivity results have been obtained, antibiotic therapy may be changed accordingly.
Surgical
To establish airway control, a tracheotomy may be indicated. Surgical drainage was once universally required but now may be reserved for cases in which antibiotic treatment fails. On external incision and drainage, straw-colored material and not frank pus is often found.
Complications
A spontaneous rupture may lead to asphyxia, aspiration, or pneumonia. The infection may spread to other deep neck compartments.
Outcome and Follow-Up
The offending tooth should also be removed if the infection’s origin is odontogenic.
ICD-9 Code
528.3 | Cellulitis and abscess Cellulitis of mouth (floor) Ludwig angina Oral fistula |
Further Reading
Har-El G, Aroesty JH, Shaha A, Lucente FE. Changing trends in deep neck abscess: a retrospective study of 110 patients. Oral Surg Oral Med Oral Pathol 1994;77(5):446–450
5.1.1.3 Deep Neck Infections
Key Features
• Deep neck infections are most commonly caused by tonsillar, peritonsillar, or odontogenic infections.
• They may involve surrounding nerves, vessels, bones, and other soft tissue.
• Microbiology typically reveals mixed bacterial flora, including anaerobic species.
• Deep neck spaces have avenues of communication with each other: infection in one space can spread to adjacent spaces.
Deep neck infections hold the potential for severe complications. Complex head and neck anatomy often makes early recognition of deep neck infections challenging, and a high index of suspicion is necessary to avoid delay in treatment. Aggressive monitoring and management of the airway are the most urgent aspects of care, followed by appropriate antibiotic coverage and surgical drainage, as needed. Risk factors for deep neck infection include diabetes mellitus, HIV, steroid therapy, chemotherapy, and other sources of immune compromise.
Clinical
Signs and Symptoms
Pain and swelling of the neck are the most prevalent symptoms. Fever, malaise, and dysphagia may occur. Other common symptoms are deep space specific and include dysphagia, trismus, dysphonia, otalgia, and dyspnea. In the pediatric population, fever, neck mass, and stiffness are most prevalent, followed by sore throat, poor oral intake, drooling, and lymphadenopathy. Stridor, dyspnea, decreased air movement, or cyanosis suggest impending respiratory compromise.
Differential Diagnosis
The differential diagnosis includes infected congenital cysts, lymphangitis, tumor, cellulitis, and necrotizing fasciitis.
Evaluation
History, physical examination, laboratory work, and diagnostic imaging each provide important clues when assessing a patient for a deep neck infection.
Physical Exam
Initial evaluation of the airway is always the first priority and any signs of respiratory distress or impending airway compromise should be immediately and aggressively managed.
Imaging
A contrast-enhanced CT scan is most useful. Ultrasound may be more accurate than CT in differentiating a drainable abscess from cellulitis. Magnetic resonance imaging (MRI) provides better soft tissue definition than CT. MRI also avoids exposure to radiation and interference from dental fillings.
Labs
Labs should include a complete blood count (CBC) with differential, serum glucose, and electrolytes; coagulation studies; HIV screening in adults; blood cultures; and appropriate cultures of aspirates obtained before antibiotics are instituted, if possible.
Microbiology
Commonly polymicrobial and reflect the oropharyngeal flora. Frequently isolated aerobes include Streptococcus viridans, Klebsiella pneumoniae, and Staphylococcus aureus, and less frequently, Streptococcus pneumoniae, Streptococcus pyogenes, Neisseria species, and Haemophilus influenzae. Common anaerobic isolates include Peptostreptococcus, Bacteroides fragilis, pigmented Prevotella and Porphyromonas spp, Fusobacterium spp, and Eikenella corrodens.
Treatment Options
Airway control is the first priority of treatment, followed by IV antibiotics and timely surgical drainage. Airway management, if necessary, should be undertaken under controlled conditions, if possible in the operating room, with either awake fiberoptic intubation or awake tracheotomy. Blind oral or nasotracheal intubation or attempts with neuromuscular paralysis may precipitate an airway crisis.
Medical
Every patient who has a deep neck infection should be given empiric antibiotic therapy until culture and sensitivity results are available. Empiric therapy should be effective against the aerobic and anaerobic bacteria that are commonly involved.
Either penicillin in combination with a -lactamase inhibitor (e.g., amoxicillin with clavulanic acid) or a -lactamase–resistant antibiotic (e.g., cefoxitin, cefuroxime, imipenem, or meropenem) in combination with a drug that is highly effective against most anaerobes (e.g., clindamycin or metronidazole) is recommended for optimal empiric coverage. Once available, the results of the culture and sensitivity tests can allow for tailoring of adequate antibiotic therapy.
In select cases, an uncomplicated deep neck abscess or cellulitis can be effectively treated with antibiotics and careful monitoring, without surgical drainage. Simultaneous medical treatment for associated comorbidities such as diabetes mellitus can improve the overall immune status of a patient.
Surgical
Indications for surgery include airway compromise, critical condition, septicemia, complications, descending infection, diabetes mellitus, or no clinical improvement within 48 hours of the initiation of parenteral antibiotics. In addition, abscesses 3 cm in diameter that involve the prevertebral, anterior visceral, or carotid spaces, or that involve more than two neck spaces, should be surgically drained.
Surgical drainage (Table 5.1) can be performed in several ways, including simple intraoral or extraoral incision and drainage for superficial abscesses, a more extensive external cervical approach with drain placement for more complicated infections, and minimally invasive techniques such as image-guided needle aspiration and indwelling catheter placement.
Complications
Complications include mediastinitis, aspiration pneumonia, lung abscess, empyema, Lemierre syndrome (suppurative thrombophlebitis of the internal jugular vein), carotid artery aneurysm or rupture, osteomyelitis involving the mandible or cervical vertebral bodies, meningitis, intracranial abscess, and disseminated intravascular coagulation.
Outcome and Follow-Up
The initiating etiology, if recognized (an infected tooth, a tonsillar abscess), and predisposing systemic conditions (diabetes mellitus) should be addressed.
Infection Site | Surgical Approaches for Drainage |
|---|---|
Peritonsillar | Intraoral needle aspiration or incision and drainage |
Submandibular space | Supramylohyoid – intraoral drainage Inframylohyoid – extraoral surgical drainage |
Parapharyngeal space | External cervical approach along the anterior border of the sternocleidomastoid muscle CT-guided transoral drainage |
Masticator space | External incision along inferior border of mandible Intraoral approach via retromolar trigone |
Parotid space | External parotidectomy incision |
Retropharyngeal space | Protected airway Transoral |
Prevertebral space | External cervical approach |
Carotid space | External cervical approach |
ICD-9 Codes
478.22 | Parapharyngeal abscess |
478.24 | Retropharyngeal abscess |
475 | Peritonsillar abscess |
| Abscess of tonsil |
| Peritonsillar cellulitis |
| Quinsy |
Further Reading
Har-El G, Aroesty JH, Shaha A, Lucente FE. Changing trends in deep neck abscess: a retrospective study of 110 patients. Oral Surg Oral Med Oral Pathol 1994;77(5):446–450
Larawin V, Naipao J, Dubey SP. Head and neck space infections. Otolaryngol Head Neck Surg 2006;135(6):889–893
Vieira F, Allen SM, Stocks RMS, Thompson JW. Deep neck infection. Otolaryngol Clin North Am 2008;41(3):459–483
Weed HG, Forest LA. Deep Neck infection. In: Cummings CW, Flint PW, Harker LA, et al, eds. Otolaryngology: Head and Neck Surgery. Vol. 3, 4th ed. Philadelphia, PA: Elsevier Mosby; 2005:2515–2524
5.1.1.4 Neck Trauma
Key Features
• Neck trauma can be either penetrating or blunt.
• There is potential injury to the larynx, trachea, esophagus, major vessels, and nerves.
• Airway management is always the priority.
• Cervical spine injury must be excluded.
Factors in the mechanism of neck trauma determine the location of injury, the injury characteristics, the tissues and organs involved, and the extent of damage to the tissues and organs.
Epidemiology
Neck trauma accounts for 5 to 10% of all serious traumatic injuries. Blunt trauma to the neck typically results from motor vehicle crashes, but it also occurs with sports-related injuries, clothesline injury, strangulation, or blows from the fists or feet. Blunt trauma has become much less common since routine seatbelt use has been established.
For penetrating trauma, 95% of wounds result from guns and knives, with the remainder resulting from motor vehicle accidents, household injuries, industrial accidents, and sporting events. The male to female ratio of penetrating neck trauma is 5:1.
Clinical
Critical organs and structures are at risk from neck trauma; clinical manifestations may vary greatly. The presence or absence of signs and symptoms can be misleading, serving as a poor predictor of underlying damage.
Signs
Signs of airway injury:
• Subcutaneous emphysema – tracheal, esophageal, or pulmonary injury
• Air bubbling through the wound
• Stridor or respiratory distress – laryngeal and/or esophageal injury
• Cyanosis
Signs of vascular injury:
• Hematoma (expanding) – vascular injury
• Active external hemorrhage from the wound site – arterial vascular injury
• Bruit/thrill – arteriovenous fistula
• Pulselessness/pulse deficit
• Distal ischemia (neurologic deficit in this case)
Signs of pharyngoesophageal injury:
• Hematemesis, inability to tolerate secretions
• Neck crepitus
• Development of mediastinitis
Symptoms
• Clinical manifestations may vary greatly depending on involved organs and systems.
• Dysphagia – tracheal and/or esophageal injury
• Hoarseness – tracheal and/or esophageal injury
• Oronasopharyngeal bleeding – vascular, tracheal, or esophageal injury
• Neurologic deficit – vascular and/or spinal cord injury
• Hypotension – nonspecific; may be related to the neck injury or may indicate trauma elsewhere
Differential Diagnosis
Considerations with neck trauma include cervical spine injury, laryngotracheal injury, vascular injury, and pharyngoesophageal injury.
Evaluation
History
History, if available, can provide important details regarding the mechanism of injury.
Physical Exam
Advanced Trauma Life Support (ATLS) protocols are followed. Exam begins with ABCs (airway, breathing, circulation), followed by secondary assessment once a patient has a safe airway and is hemodynamically stable. All patients with neck trauma should be assumed to have a cervical spine injury until this has been ruled out.
With blunt trauma, injury to the larynx or trachea is the most common serious finding and often presents with subcutaneous air, hoarseness, or odynophagia. In a stable patient, flexible fiberoptic laryngoscopy can reveal evidence of injury such as blood, motion impairment, or edema.
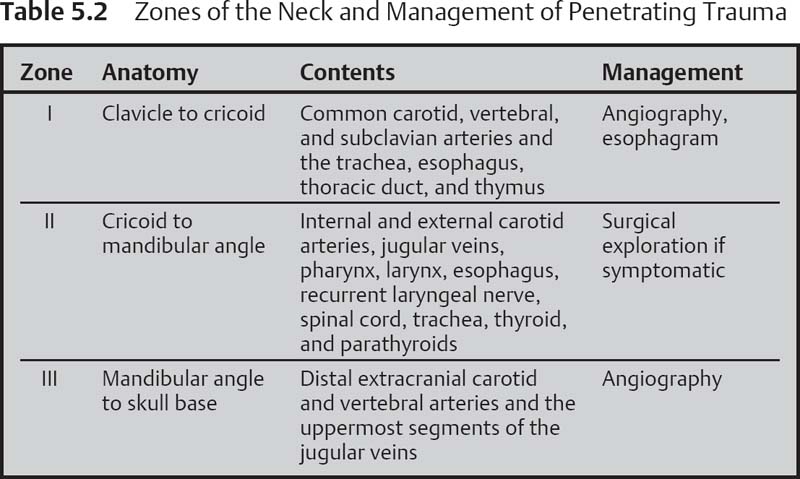
With penetrating trauma, determine which vertical zones of the neck are involved (Table 5.2). Zone I extends from the clavicle to the cricoid cartilage; zone II extends from the cricoid to the mandibular angle; zone III extends from the mandibular angle to the skull base.
Imaging
Controversy exists regarding aspects of trauma management, with trends away from surgical exploration in stable patients and expanding use of imaging with observation. There is an inherent delay with any imaging study; transport to the operating room should not be delayed by an imaging study when the patient’s condition warrants emergent surgery.
Penetrating zone I and III injuries should undergo angiographic evaluation of the carotids. Zone I injuries should also undergo esophageal studies (i.e., Gastrografin swallow study).
Cervical spine radiography is routine. Review for emphysema, fractures, displacement of the trachea, and the presence of a foreign body.
CT scans prove most useful when bony or soft tissue damage is a consideration. MRI or MR angiography is used for evaluation of the patient exhibiting neurologic impairment with minimal or absent abnormalities of the cervical spine on plain radiograph.
CT angiography offers advantages over angiography. It is readily accessible, can be rapidly performed, and causes fewer complications than angiography. Additionally, some experts assert that subtle disruptions of the vessel wall may be detected on CT angiography. Artifacts secondary to metal can obscure vascular detail and can limit CT angiography.
Labs
As necessary, a trauma panel, CBC, electrolytes, other warranted blood chemistry levels, and blood type and cross-matching should be obtained.
Treatment Options
Establishment of the airway is the first priority. If there is airway compromise, a surgical airway, rather than endotracheal intubation, is usually preferred. Either a cricothyroidotomy or tracheotomy is performed if there is respiratory distress. Bleeding is initially managed with direct pressure and establishment of large-bore IV access to permit fluid resuscitation.
• Endoscopy
 Laryngoscopy, bronchoscopy, pharyngoscopy, and esophagoscopy may be useful in the assessment of the aerodigestive tract. Rigid endoscopes are superior to flexible scopes.
Laryngoscopy, bronchoscopy, pharyngoscopy, and esophagoscopy may be useful in the assessment of the aerodigestive tract. Rigid endoscopes are superior to flexible scopes.
• Angiography
 Angiography routinely is used to evaluate stable patients sustaining penetrating wounds to zones I and III that pierce the platysma.
Angiography routinely is used to evaluate stable patients sustaining penetrating wounds to zones I and III that pierce the platysma.
 A four-vessel study is a prerequisite.
A four-vessel study is a prerequisite.
 Drawbacks include cost and the inherent danger of any vascular, particularly arterial, invasive procedure.
Drawbacks include cost and the inherent danger of any vascular, particularly arterial, invasive procedure.
The unstable patient (hemodynamic instability, severe hemorrhage, expanding hematoma) is taken to the operating room. The stable patient is categorized as symptomatic or asymptomatic. Signs or symptoms of injury to the airway, esophagus, vessels or nerves (i.e., hemoptysis, hoarseness, dysphagia, crepitus) dictate further workup/interventions. If the penetrating injury is in zone II, neck exploration is generally done; if the injury is zone I, angiography and esophageal studies are performed. If the injury is in zone III, angiography should be performed. Asymptomatic patients are also typically imaged as above if the injury is zone I or III and observed if the injury is zone II.
In general, vascular injuries are managed either with embolization or surgical control. Surgery involves exploration and management of injuries of the carotid sheath, esophagus, and laryngotracheal complex (Table 5.3).
There is no role for probing or local exploration of the neck in the trauma bay or emergency room because this may dislodge a clot and initiate uncontrollable hemorrhage.
Carotid artery injuries |
Vertebral artery injuries |
Jugular vein injury |
Laryngotracheal injuries |
Esophageal injuries |
Nerve injuries |
Thoracic duct injuries |
Thyroid injuries |
Outcome and Follow-Up
Standard postoperative management for neck surgery is followed. Overall mortality is 1 to 2%.
ICD-9 Codes
959.09 | Injury of face and neck |
900.9 | Injury to unspecified blood vessel of head and neck |
957.0 | Injury to superficial nerves of head and neck |
Further Reading
Biffl WL, Moore EE, Rehse DH, Offner PJ, Franciose RJ, Burch JM. Selective management of penetrating neck trauma based on cervical level of injury. Am J Surg 1997;174(6):678–682
Scott BG. Approach to penetrating injuries of the neck. In: Stewart MG, ed. Head, Face, and Neck Trauma: Comprehensive Management. Stuttgart/New York: Thieme; 2005:202–206
5.1.2 Approach to Neck Masses
Key Features
• A neck mass may be inflammatory, congenital, or neoplastic.
• They may be anterior (midline), lateral, posterolateral, or supraclavicular.
• The patient’s history should include age, duration, progression, pain, infection, smoking, prior cancer, exposure to tuberculosis, and animal exposure.
Neck masses are relatively common and may present at any age group. The differential diagnosis is broad, and both benign and malignant processes should be considered. A systematic approach is crucial to developing a rapid diagnosis and treatment plan.
Age is an important factor in evaluating a neck mass. The age groups include pediatric, young adult, and older adult (40 years). Each age group exhibits a certain relative frequency of disease occurrences, which can guide the diagnostician to further differential considerations. In general, neck masses in children are more commonly inflammatory or infectious. This is also true in young adults, although lymphoma is a consideration. In older adults, a neck mass should be considered neoplastic until proven otherwise. The location of congenital neck masses is important, as such lesions are often characterized by their location. The location of malignant neck masses particularly if metastatic may help identify the primary tumor (Fig. 5.5).
Clinical
Signs and Symptoms
Depending on the cause, the neck mass may be painless (early neoplasm or congenital mass) or painful (infection or trauma). Depending on the etiology, associated symptoms may be those of an upper respiratory infection, toothache (infectious or inflammatory mass) or dysphagia, odynophagia, hoarseness, otalgia, hemoptysis, weight loss, night sweats, and fever (neoplasm).
Differential Diagnosis
See Table 5.4.
Evaluation
History
A thorough review of the developmental time course of the mass, associated symptoms, personal habits prior to the trauma or infection, irradiation, or surgery is important. Ask about smoking, tobacco chewing, alcohol use, fever, pain, weight loss, night sweats, exposure to tuberculosis, animals, pets, and occupational/sexual history.
Physical Exam
All mucosal surfaces of the nasopharynx, oropharynx, larynx, and nasal cavity should be visualized by direct examination or by indirect mirror or fiberoptic visualization. State of dentition should be noted. The oropharyngeal surfaces should be palpated. Regarding the neck mass, emphasis on location, tenderness, mobility, and consistency of the neck mass can often place the mass within a general etiologic grouping.
Imaging
CT is important and can better elucidate the nature of a neck mass and its association with surrounding structures. Criteria such as heterogeneity of the center of the mass, blurred borders, and a round shape are suggestive of malignancy. CT may also be able to identify additional neck masses not clinically palpable and/or the primary tumor. Contrast should be used except in the suspected thyroid lesion as it may interfere with radioactive-iodine imaging studies or therapy.
MRI is comparable to CT, but it is more expensive and has longer imaging time. With contrast, it is especially good for vascular and neural delineation.
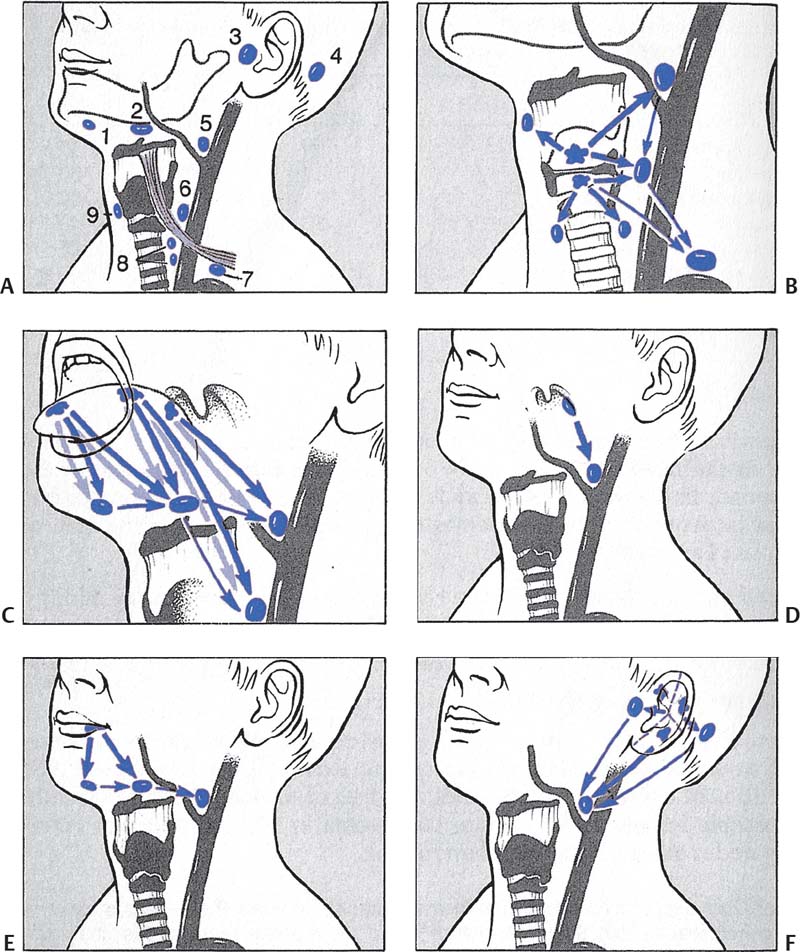
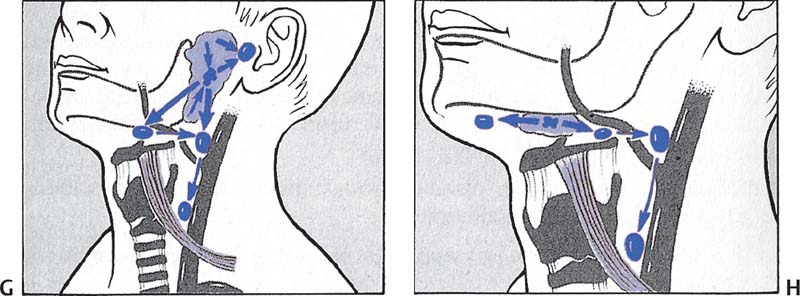
Fig. 5.5 Head and neck masses. (A) Typical sites of regional lymph node metastases: 1. submental lymph nodes; 2. submandibular lymph nodes; 3. parotid and preauricular lymph nodes; 4. retroauricular lymph nodes; 5. lymph nodes of the jugulofacial venous angle; 6. deep cervical lymph nodes; 7. lymph nodes in the juguloclavicular venous angle: lower deep cervical lymph nodes and supraclavicular lymph nodes; 8. pre- and peritracheal lymph nodes; 9. prelaryngeal lymph nodes. (B) Laryngeal carcinoma. (C) Carcinoma of different parts of the tongue. Note the tendency to contralateral metastases. (D) Tonsillar carcinoma. (E) Lower lip carcinoma. (F) Carcinoma of the external ear. Note the segmental lymphatic efferent from the auricle. (G) Parotid carcinoma. Note the intraglandular lymph node metastases. (H) Submandibular gland carcinoma. (From Becker W, Naumann HH, Pfaltz CR. Ear, Nose and Throat Diseases: A Pocket Reference. 2nd ed. Stuttgart/New York: Thieme; 1994:516.)
Angiography is recommended if primary vascular neck mass is suspected and synchronous embolization is being considered.
Ultrasound is helpful in differentiating solid from cystic masses and congenital cysts from solid lymph nodes and glandular tumors.
Labs
A routine complete blood count test may reveal an infectious process. More specialized laboratory tests may become necessary as the investigation proceeds.
Other Tests
• Fine-needle aspiration biopsy (FNAB): Currently, FNAB is the standard of diagnosis for neck masses and is indicated in any neck mass that is not an abscess and persists despite appropriate antibiotic therapy. FNAB separates inflammatory and reactive processes that usually do not require surgery from neoplastic lesions, either benign or malignant.
• Panendoscopy: If careful examination in the office does not identify the etiology of the neck mass and a tumor is suspected, the upper aerodigestive tract should be examined under anesthesia. Biopsies should be performed on any suspicious mucosal lesions.
Treatment Options
Medical
A tender, mobile mass or one highly suggestive of inflammatory or infectious etiology may warrant a short clinical trial of antibiotics and observation with close follow-up. Use steroids judiciously; steroids may shrink a neck mass caused by lymphoma lulling the physician and patient into a false sense that the condition is improving.
Surgical
• Open excisional biopsies should be avoided in cases in which a nonlymphoma malignancy (epidermoid, melanoma) is suspected. If FNAB results are negative or equivocal but suspicion for malignancy persists, an open excisional biopsy of the cervical lymph node may be performed. The patient and surgeon should be prepared to proceed immediately with a complete neck dissection depending on the results of frozen sections.
• Open excisional biopsies may be performed for lymphomas or granulomatous disease.
• Inflamed congenital masses are typically treated with antibiotics and then surgically removed after inflammation has subsided.
• Infectious processes causing neck masses are treated medically. Surgery in the form of incision and drainage is used in cases that do not respond to appropriate medical therapy.
ICD-9 Code
784.2 | Swelling mass or lump in head and neck |
Further Reading
Armstrong WB, Giglio MF. Is this lump in the neck anything to worry about? Postgrad Med 1998;104(3):63–64
McGuirt WF. The neck mass. Med Clin North Am 1999;83(1):219–234
Mendenhall WM, Mancuso AA, Amdur RJ, Stringer SP, Villaret DB, Cassisi NJ. Squamous cell carcinoma metastatic to the neck from an unknown head and neck primary site. Am J Otolaryngol 2001;22(4):261–267
5.2 Head and Neck Cancer
Key Features
• Head and neck cancer is a heterogeneous group of tumors, consisting predominantly of squamous cell carcinoma (SCC) of the upper aerodigestive tract.
• This type of cancer is linked to tobacco smoking and alcohol.
• There is a propensity for second primary tumors (between 4 and 7% per year), especially if still smoking.
• Multimodality treatment includes surgery, radiation, and chemo-therapy.
• Adult with persistent neck mass is SCC until proved otherwise.
Head and neck cancer, predominantly SCC, can affect the oral cavity, pharynx, larynx, hypopharynx, cervical esophagus, nose, and paranasal sinuses. The goal of treatment is cure or palliation with preservation of function. Specific sites and subsites of head and neck cancer are discussed in subsequent chapters.
Epidemiology
Head and neck SCC accounts for ~5% of cancers in the United States. This corresponds to an estimated 17 per 100,000 Americans with newly diagnosed SCC of the head and neck per year. These cancers are more common in men, and typically occur in patients over age 50. The etiology includes tobacco use (smoking and smokeless) and alcohol consumption. Eighty-five percent of head and neck SCC are linked to tobacco use. The synergistic effect of alcohol and smoking increases the risk of disease many more times than the simple additive risk of either risk factor alone. In SCC, mutations in the p53 gene correlate with drinking and smoking habits. Some 15% of patients have a viral etiology. Epstein-Barr virus (EBV) has been implicated in the development of nasopharyngeal carcinoma. Human papillomavirus (HPV) infection is another factor implicated in the carcinogenesis of upper aerodigestive tract tumors. In particular, HPV-16 can be isolated in up to 72% of oropharyngeal cancers. The recent increase in cancers of the tongue and tonsils in developed countries, particularly in younger patients, has been linked to HPV infection.
During the past 20 years, the overall incidence of head and neck SCC has been declining in the United States, a decline that is attributed to a decrease in the prevalence of smoking. In other parts of the world, head and neck SCC is attributed to habitual and cultural habits such as chewing of paan (betel leaf with areca nut), smoking khat (Catha edulis), and drinking yerba maté.
Clinical
Signs
Signs may include hoarseness, muffled speech, trismus, and recurrent epistaxis. Many patients present with a neck mass as chief complaint, representing metastatic nodal disease from an occult primary tumor in the upper aerodigestive tract (Table 5.5).
Symptoms
Symptoms of head and neck SCC are variable and depend on the site and stage of the primary tumor (see Staging of Head and Neck Cancer, below). Early symptoms may be vague and mimic benign disease and are therefore only discovered at advanced stages of disease. Symptoms may include dysphagia, odynophagia, a globus sensation, changes in voice (this includes both hoarseness and velopharyngeal insufficiency), referred otalgia, CN hypesthesia, nasal obstruction, epiphora, and hyposmia.
Differential Diagnosis
• Upper respiratory infections such as pharyngitis, laryngitis, deep neck infections or abscesses
• Congenital masses and cysts
• Upper airway manifestations of rheumatologic and autoimmune diseases
• Hematologic malignancies (lymphoma)
• Tuberculosis
• Fungal infections
Tumor Location | Incidence of Cervical Lymph-Node Metastases at Diagnosis (%) |
|---|---|
Oral cavity | 30–65 |
Oropharynx | 39–83 |
Nasopharynx | 60–90 |
Hypopharynx | 52–72 |
Supraglottis | 35–54 |
Glottis | 7–9 |
Nasal cavity and paranasal sinuses | 10–20 |
Salivary glands | 25–50 |
Thyroid gland | 18–84* |
Source: Probst R, Grevers G, Iro H. Basic Otorhinolaryngology: A Step-by-Step Learning Guide. Stuttgart/New York: Thieme; 2009:333.
*Depends on age and histologic subtype.
Evaluation
History
History should include questions about risk factors, breathy voice or prolonged hoarseness, dysphagia, hemoptysis, otalgia, and unintentional weight loss.
Physical Exam
Physical exam should include careful inspection of the oral and oropharyngeal mucosa for lesions, and palpation of the tonsillar region and tongue base for firm nodules or masses.
An indirect mirror or flexible fiberoptic laryngoscopy should be performed. During this examination, the patient should be asked to perform several maneuvers such as tongue protrusion, puffing out the cheeks, lightly coughing and speaking to better visualize and access the larynx and the hypopharynx. It is important that laryngeal motility be assessed, as this is critical in tumor staging. The neck should be examined in a systematic fashion. Any palpable lymph nodes should be assessed with regard to size, location, and mobility.
Imaging
A contrast-enhanced CT scan or MRI scan of head and neck should be obtained to assess local and regional extent of disease and involvement of adjacent structures, such as the great vessels of the neck and the prevertebral fascia. Specific characteristics of regional lymphadenopathy, if present, should be noted, such as extracapsular spread, central necrosis, and size of involved lymph nodes.
A metastatic workup may consist of either a chest radiograph with liver function tests, CT scan of the chest and abdomen, or alternatively, a fluorodeoxyglucose-positron emission tomography (FDG-PET) scan.
Labs
Blood count, electrolyte, and liver function tests should be performed to assess nutritional status.
Other Tests
FNAB is a highly accurate technique for the investigation of cervical lymph node metastases in head and neck SCC and is the first-line test in a patient with lymphadenopathy. Sensitivity of this test is improved when performed with ultrasound guidance. It is best to avoid open biopsy of a neck mass, as tumor spillage and violation of fascial planes is problematic.
Patients in whom a suspicion of head and neck SCC exists should undergo biopsy of suspicious primary site lesions. Because of the propensity for second primary tumors that accompany head and neck SCC, these patients should undergo a panendoscopy (i.e., triple endoscopy), laryngoscopy, esophagoscopy, and bronchoscopy together, to search for synchronous lesions. This may also lend insight as to the extent of the primary lesion, particularly important in the smoking patient. This is a point of controversy, and PET scanning may play a greater role in the future. Mention should be made that in the current literature, the sensitivity of PET scanning is not adequate to replace panendoscopy: for instance, the larynx has baseline activity on PET due to patient breathing and/or talking during the scan.
Pathology
Ninety percent of head and neck cancers are SCCs.
Treatment Options
Treatment of head and neck SCC consists of surgery, radiotherapy, chemotherapy, or a combination of these. Surgical resection remains the gold standard for treatment of head and neck cancer. Surgery may address the primary tumor as well as cervical metastasis.
Radiation therapy for SCC of the head and neck involves the delivery of high-energy ionizing radiation to targeted tissues. Radiation doses can be delivered by different methods, including fractionation, hyperfractionation, accelerated fractionation, and intensity-modulated radiotherapy (IMRT).
Chemotherapy for SCC of the head and neck involves the systemic administration of cytotoxic drugs that target rapidly dividing cells. Individual chemotherapeutic agents effective in the therapy of head and neck cancer include cisplatin, methotrexate, 5-fluorouracil, taxanes, ifosfamide, and bleomycin.
Of the molecularly targeted agents, cetuximab is an IgG1 chimeric antibody directed against the epidermal growth factor receptor (EGFR).
Other therapy methods for head and neck cancer include photosensitizers and interstitial laser therapy, photodynamic therapy, immune therapy, gene therapy, and targeted therapy against EGFR and HPV.
Cancer of Unknown Primary
Patients with head and neck cancer typically present with a painless neck mass. In 2 to 8% of these patients, the tumor origin is not known. This is known as cancer of unknown primary (CUP), carcinoma of unknown primary origin, or occult primary malignancy. A primary tumor is considered unknown only after a thorough investigation (including physical exam, imaging, and biopsies) has been completed.
Evaluation of possible CUP begins with a thorough history and physical examination, including flexible fiberoptic laryngoscopy. FNAB of the neck mass provides a histologic diagnosis, which may assist with finding the primary tumor, but 90% are attributable to SCC.
The next step is to perform panendoscopy with biopsies whether or not the primary site was located on imaging. As noted earlier, panendoscopy typically included bronchoscopy, esophagoscopy, and direct laryngoscopy. If no obvious tumor is visualized, tonsillectomy and guided biopsies are performed. Some advocate a unilateral tonsillectomy limited to the side of the neck mass, but others advocate bilateral tonsillectomies in this circumstance. The most common sites of CUP include the palatine tonsil and base of tongue, followed by the nasopharynx and pyriform sinus. Each of these sites should at least be inspected with consideration of guided biopsies.
Treatment of CUP is controversial. Surgical excision in the form of a neck dissection followed by radiation therapy allows for a lower total dose of radiation. Primary radiation therapy provides treatment to both the upper aerodigestive tract and its locoregional metastasis but forces the radiation oncologist to treat a wider field as the primary site is unknown.
Outcome and Follow-Up
The treatment of head and neck SCC, whether surgical or chemoradiation, often leaves the patient with significant speech and swallowing deficits. Therefore, is it paramount for a multidisciplinary team to treat head and neck SCC. This should include the surgical oncologist, medical and radiation oncologist, oral surgeon, prosthodontist, speech language and swallowing pathologist, nurse, and social worker.
Patients with regional neck disease prior to treatment should have a CT scan or an integrated FDG-PET/CT. There is controversy as to the need for a planned neck dissection following radiotherapy in patients with high-risk disease. Most recurrences of SCC occur within 3 years of the initial treatment. Patients should be seen at 1- to 2-month intervals for 2 years after treatment. Thereafter, they should be seen at 3-month intervals for the next year. There is no evidence that routine follow-up beyond 3 years improves prognosis, although many clinicians support yearly follow-up.
Patients should be told of the risk of a second primary tumor and encouraged to report any new symptoms. The risk of a second primary carcinoma is highest in those who continue to smoke. Patients should be counseled for tobacco cessation.
ICD-9 Code
199.1 | Other malignant neoplasm of unspecified site |
[See subsequent chapters for appropriate codes for cancers of specific sites.]
Staging of Head and Neck Cancer
• The regional lymph node metastases and their effect on stage grouping are fairly consistent throughout all anatomic sites of head and neck cancer.
NX – Regional lymph nodes cannot be assessed.
N0 – No evidence of regional lymph node metastasis
N1 – Metastasis to a single ipsilateral lymph node measuring ≤3 cm in greatest diameter
N2 – Further divided into three categories:
N2a – Single ipsilateral lymph node between 3 and 6 cm
N2b – Multiple ipsilateral lymph nodes <6 cm
N2c – Bilateral or contralateral lymph nodes <6 cm in greatest dimension
N3 – Lymph node >6 cm
• Distant metastatic disease is divided into two categories:
M0 – Absence of distant disease
M1 – Presence of distant metastatic disease
• The T stage of a tumor indicates the extent of the primary tumor and varies by anatomic subsite. This can be measured by size, as in the oral cavity, oropharynx, and salivary glands; by involvement of varying sub-sites, as in the nasopharynx, hypopharynx, and larynx; or by extent of invasion and destruction, as in the maxillary sinus. Across all anatomic sites of the head and neck, the following classifications apply:
Stage I disease – Includes only T1 N0 M0 tumors
Stage II disease – Includes T2 N0 M0 tumors
Stage III disease – Includes T3 N0 M0 and T1–3 disease, which is N1 M0
Stage IV disease – Includes T4 tumors with or without nodal disease, as well as any tumor with N2 or N3 disease or evidence of distant metastatic disease
Further Reading
American Joint Committee on Cancer. AJCC Cancer Staging Handbook. 7th ed. New York: Springer-Verlag; 2010
Cianchetti M, Mancuso AA, Amdur RJ, et al. Diagnostic evaluation of squamous cell carcinoma metastatic to cervical lymph nodes from an unknown head and neck primary site. Laryngoscope 2009;119(12):2348–2354
Genden EM, Varvares MA, eds. Head and Neck Cancer: An Evidence-Based Team Approach. Stuttgart/New York: Thieme; 2008
Miller FR, Hussey D, Beeram M, Eng T, McGuff HS, Otto RA. Positron emission tomography in the management of unknown primary head and neck carcinoma. Arch Otolaryngol Head Neck Surg 2005;131(7):626–629
Miller FR, Karnad AB, Eng T, Hussey DH, Stan McGuff H, Otto RA. Management of the unknown primary carcinoma: long-term follow-up on a negative PET scan and negative panendoscopy. Head Neck 2008;30(1):28–34
5.2.1 Chemotherapy for Head and Neck Cancer
Key Features
• Concurrent chemotherapy with definitive radiotherapy is a safe and effective means to treat locally advanced SCC of the head and neck.
• Concurrent chemotherapy with postoperative radiotherapy (i.e., chemoradiotherapy) improves survival in select high-risk patients.
• Palliative chemotherapy can reduce symptoms and modestly extend survival in an incurable setting.
• Newer biologic and cytotoxic agents continue to cause the treatment of head and neck cancer to evolve.
The role of chemotherapy in head and neck cancer is expanding and its utility varies with the stage of the disease. For patients with metastatic or incurable locoregional disease, chemotherapy is palliative. For patients with potentially curable locoregional head and neck cancer, chemotherapy is an integral component of the multimodality approach.
Chemotherapy in the definitive treatment of head and neck cancer is an adjuvant therapy. Strictly defined, an adjuvant therapy is an addition to the potentially curable modality (primary surgery or definitive radiation) that improves outcomes. Broadly speaking, adjuvant therapies can be preoperative (or preradiotherapy), concurrent with radiation, or postoperative (or postradiotherapy). Most early adjuvant chemotherapy trials in cancer were postoperative in nature, so “adjuvant therapy” has also been used to describe only postoperative (or postradiotherapy) chemotherapy. This has given rise to the term “neoadjuvant” chemotherapy to describe preoperative (or preradiotherapy) chemotherapy. Induction chemotherapy and neoadjuvant chemotherapy are synonymous.
Neoadjuvant Chemotherapy
Advantages of neoadjuvant chemotherapy (i.e., induction chemotherapy) include an intact vascular bed for better drug delivery, reduced tumor bulk to improve the ease of resection, and early eradication of regional and distant micrometastases. Disadvantages include delaying surgery in potentially curable patients with chemoresistant disease, relying on clinical staging to make treatment decisions, the morbidity of “overtherapy,” and patient nonadherence after chemotherapy. Neoadjuvant cisplatin and 5-fluorouracil (5-FU) followed by radiotherapy in responders was an organ preservation strategy described in the frequently cited VA Laryngeal Cancer Study Group. Subsequent results have shown this approach to be inferior to concurrent cisplatin with radiation, but newer induction regimens including docetaxel have reintroduced neoadjuvant chemotherapy followed by radiotherapy as a viable option. Neoadjuvant chemo-therapy before surgery has not been found to be helpful in randomized trials.
Concomitant Chemoradiotherapy
The simultaneous use of chemotherapy and radiation continues to be the standard for locally advanced SCC (stages III to IVb). The primary benefit has been in decreasing locoregional failure, which has translated into roughly a 10% overall survival benefit. The effect on decreasing metastatic disease has been inconsistent. It is believed that chemotherapy may have some benefit against radioresistant hypoxic tumor cells. However, the simultaneous use of chemotherapy and radiotherapy has significantly increased grade 3 and 4 toxicity, which can be potentially lethal or cause treatment breaks that decrease radiation’s efficacy. In patients who are receiving surgery and are found to have high-risk features (positive margins, N2 disease, nodal extracapsular extension), postoperative cisplatin with radiation has proven superior to radiation alone. Cisplatin, 5-FU, taxanes, and mitomycin C all act as radiosensitizing agents.
Adjuvant Therapy
The use of postoperative or postradiation chemotherapy has not been found to be helpful in randomized trials, although it is commonly done for three cycles in nasopharyngeal cancer based on the Intergroup Trial showing concurrent cisplatin with radiation followed by three cycles of cisplatin plus 5-FU improved survival over radiation alone. It is controversial whether the cycles given after radiation add any independent benefit.
Types of Chemotherapeutic Agents Used for Head and Neck Cancer
Alkylating Agents
The cytotoxic effects of alkylating agents (e.g., cisplatin) are based on their interaction with DNA. These agents cause substitution reactions, cross-linking reactions, or strand-breaking reactions. These agents alter the information coded in the DNA molecule resulting in inhibition of or inaccurate DNA replication with resultant mutation or cell death.
Antimetabolites
The cytotoxic effect of antimetabolites (e.g., methotrexate) is due to their structural similarity to naturally occurring metabolites involved in nucleic acid synthesis. They inhibit critical enzymes involved in nucleic acid synthesis and become incorporated into the nucleic acid and produce incorrect codes. Both of these mechanisms result in an inhibition of DNA synthesis and ultimate cell death.
Antitumor Antibiotics
Antitumor antibiotics (e.g., mitomycin, bleomycin) are antimicrobial compounds produced by Streptomyces species in culture. They are cytotoxic in that they affect the structure and function of nucleic acids by intercalation between DNA base pairs (doxorubicin), DNA strand fragmentation, or cross-linking of DNA.
Alkaloids
Alkaloids (e.g., vincristine, vinblastine) bind to free tubulin dimers and disrupt the balance between microtubule polymerization and depolymerization, resulting in the net dissolution of microtubules, destruction of the mitotic spindle, and arrest of cells in metaphase.
Taxanes
Taxanes (e.g., paclitaxel, docetaxel) are compounds that disrupt equilibrium between free tubulin and microtubules, causing stabilization of ordinary cytoplasmic microtubules and the formation of abnormal bundles of microtubules.
EGFR Inhibitors
EGFR is a protein found on the surface of some cells and to which epidermal growth factor binds, causing the cells to divide. It is found at abnormally high levels on the surface of many types of cancer cells, so these cells may divide excessively in the presence of epidermal growth factor (EGFR, ErbB1, and HER1). Cetuximab (Erbitux, Bristol-Myers Squibb, New York, NY) specifically targets EGFR and binds to EGFR with higher affinity than its natural ligands. Binding results in the internalization of the antibody receptor complex without activation of the intrinsic tyrosine kinase. Consequently, signal transduction through this cell pathway is blocked, which inhibits tumor growth and leads to apoptosis.
During a recent multinational, randomized study to compare radiotherapy alone with radiotherapy plus cetuximab in patients with locoregionally advanced head and neck cancer, cetuximab was found to improve locoregional control and reduce mortality.
Complications
Each drug or combination of chemotoxic drugs can cause specific side effects, and some can be permanent. These side effects may be so severe that chemotherapy must be ceased. In general, chemotherapy may cause the following side effects: fatigue, nausea, vomiting, hair loss, xerostomia, anorexia, immunocompromise, diarrhea, mucositis, and death.
Further Reading
Adelstein DJ. Induction chemotherapy in head and neck cancer. Hematol Oncol Clin North Am 1999;13(4):689–698
Posner MR, Wirth LJ. Cetuximab and radiotherapy for head and neck cancer. N Engl J Med 2006;354(6):634–636
Cohen EE, Lingen MW, Vokes EE. The expanding role of systemic therapy in head and neck cancer. J Clin Oncol 2004;22(9):1743–1752
Adelstein DJ, Li Y, Adams GL, et al. An intergroup phase III comparison of standard radiation therapy and two schedules of concurrent chemoradiotherapy in patients with unresectable squamous cell head and neck cancer. J Clin Oncol 2003;21(1):92–98
Cooper JS, Pajak TF, Forastiere AA, et al; Radiation Therapy Oncology Group 9501/Intergroup. Postoperative concurrent radiotherapy and chemotherapy for high-risk squamous-cell carcinoma of the head and neck. N Engl J Med 2004;350(19):1937–1944
Mendelsohn J. The epidermal growth factor receptor as a target for cancer therapy. Endocr Relat Cancer 2001;8(1):3–9
Al-Sarraf M, LeBlanc M, Giri PG, et al. Chemoradiotherapy versus radiotherapy in patients with advanced nasopharyngeal cancer: phase III randomized Intergroup study 0099. J Clin Oncol 1998;16(4):1310–1317
5.2.2 Radiotherapy for Head and Neck Cancer
Key Features
• Definitive radiotherapy is a safe and effective means to treat various cancers of the head and neck, either in inoperable patients or as an alternative to surgery for organ preservation.
• Postoperative radiotherapy decreases local failure in select high-risk patients.
• Palliative radiotherapy can reduce local symptoms in an incurable setting.
• Radiation can be improved by sensitizing tumor cells preferentially or by decreasing radiation damage to normal tissues.
Ionizing radiation is a locoregional therapy whereby photons (gamma rays or x-rays), electrons, neutrons, protons, or heavier particles (mesons, particles, carbon ions) cause cells to either undergo death during mitosis or apoptosis, primarily through the creation of DNA double-strand breaks. The therapeutic ratio of radiation depends on the difference in sublethal repair between normal tissues and tumor cells, the use of radioprotectors and/or radiosensitizers, and the use of advanced methods to limit the irradiation of normal tissues.
Fundamental Concepts of Radiation
Radiation dose is defined as the amount of energy (joule) imparted per unit mass (kg). The SI metric unit of dose is the Gray (Gy), defined as 1 J/kg. Historically, the unit used was the rad, which is equivalent to 0.01 Gy, or 1 cGy.
Each radiation treatment is called a fraction because for most situations the total radiation dose is given over multiple sessions. A standard fraction is 1.8 to 2 Gy per fraction, and a standard course is five fractions per week with one fraction given per day. Fractionation is biologically advantageous because of the processes of tumor reoxygenation and reassortment into more radiosensitive parts of the cell cycle. Increasing the number of fractions preferentially spares normal tissues by giving them more time to repair sublethal damage. Increasing the number of fractions cannot be extended indefinitely because of tumor repopulation, which significantly reduces radiation’s efficacy if the total treatment time exceeds 7 weeks.
Various alternative fractionation strategies have been used to try to enhance radiation’s effectiveness. Accelerated radiation delivers treatment faster than standard fractionation (>10 Gy per week). Hyperfractionated radiation is the use of fraction sizes smaller than 1.8 Gy. Hypofractionated radiation is the use of fraction sizes larger than 2.0 Gy. These strategies can be combined, as in accelerated hyperfractionation.
Methods of Radiation Delivery
Radiation is broadly divided into brachytherapy and teletherapy. Brachytherapy is the placement of radioisotopes near or inside the target. In SCC of the head and neck, this is most commonly done by placing catheters in a tumor or operative bed and using an afterloading device to push the source into the catheters for predetermined periods of time to deliver a prescribed dose to the entire target volume. The exposure time ranges from over 2 to 3 days in low-dose rate applications, most commonly with cesium-137, to 10 to 30 minutes in high-dose rate applications, most commonly with iridium-192. With differentiated thyroid cancer, orally administered iodine-131 (131I) preferentially binds to tumor cells, with ablative doses of 100 to 150 mCi delivering 250 to 300 Gy.
Teletherapy, or external beam radiation, is the delivery of radiation by pointing an external source of radiation at the target. The most common source in modern radiotherapy is the linear accelerator, which can generate high energy (4 to 25 MeV) photons and electrons. Gamma knife radiosurgery units use cobalt-60 sources that emit 1.25-MeV photon beams. Intraoperative radiation can be focally delivered to internal structures with a linear accelerator or portable x-ray generator in the operating room. External beam radiation is further subdivided by the technology used.
Conventional radiation planning uses x-ray films to define the target volume. Plans are generally limited to a small number of angles and radiation beams are shaped by fabricating Cerrobend blocks. Three-dimensional (3D) conformal radiation uses CT-based treatment planning systems to improve target identification and evaluate dose distribution more accurately. This increases dose conformality to the target by making it easier to use more fields from virtually any beam angle. IMRT improves dose conformality further by delivering different doses to different sections within the same beam, and optimizes the choice and intensity of beams by using a software algorithm to simultaneously test more plans than a human could do within a reasonable period of time. Image-guided radiotherapy (IGRT), further improves dose conformality by using real-time imaging to confirm the patient is in the appropriate position on the couch before delivering radiation, thereby decreasing set-up error and allowing for tighter margins. Stereotactic radio-surgery, or SRS, is the use of a highly conformal large single fraction of external beam radiotherapy, either using a gamma knife or linear accelerator. A gamma knife uses cobalt-201 sources aimed at the same point in space to produce a small area with a high dose and sharp dose drop-off. A common trait of all modern systems is that increased dose conformality to the target requires a high level of patient set-up consistency, and this is achieved using custom masks or external frames that connect to the patient couch.
Rationale for Definitive (Curative) Radiotherapy
Primary radiotherapy in the treatment of SCC of the nasopharynx, oropharynx, oral cavity, and glottis has long been considered an option even in resectable disease. The primary justification for this is not increased efficacy over surgery but organ and functional preservation without compromising long-term efficacy. This is an option for patients with both early (stage I or II) and advanced (stage III or IV) disease. For patients with advanced disease, definitive radiation with chemotherapy with or without planned neck dissection, with surgery to the primary reserved for salvage, had an equivalent survival compared with surgery followed by radiation in randomized trials of cancers of the larynx, hypopharynx, and other areas of the pharynx. For patients with early-stage lesions of the larynx, no randomized trials of laryngectomy versus other modalities exist, but a large series of mature data exists regarding the long-term efficacy of definitive radiation. The results of these trials cannot be extrapolated to all cases, and it is likely that surgery should be the primary modality in some patient subsets. Tumor control, functional outcome, and quality of life should be considered by a multimodality treatment team before choosing an individual patient’s treatment plan.
Definitive radiation, with or without chemotherapy depending upon the histology, is also used in mucosal melanoma, skin cancer, salivary gland cancer, lymphoma, and plasmacytoma. In select cases, conformal radiation using IMRT, SRS, or brachytherapy can be used in previously irradiated sites to salvage locally recurrent cases.
Rationale for Adjuvant Radiotherapy
Postoperative radiotherapy is used if there is residual disease or a significant risk of occult residual disease. Randomized evidence supports the use of postoperative radiation for SCC that is stage III or IV or that has close or positive margins. The addition of current chemotherapy to adjuvant radiation has proven to be better than radiation alone in large randomized trials. Randomized data for other tissue types does not exist, but postoperative radiation is commonly given in high-risk cases of Merkel cell carcinoma, salivary gland carcinoma, skin cancer, and thyroid cancer. Preoperative radiation is generally reserved for marginally unresectable disease, but is more standard in esthesioneuroblastomas to make the definitive surgery smaller and less morbid.
Rationale for Palliative Radiotherapy
In the noncurative setting, radiotherapy is used to treat areas that are causing local symptoms or at a high risk to cause local symptoms. Common indications in head and neck cancer to treat the primary lesion include uncontrolled bleeding, pain, dysphagia, and a compromised airway. Metastatic disease to the bone, brain, and lung can also be palliated effectively using radiation.
Complications
Radiation side effects can be characterized as acute or late. Acute effects occur during or within the first few weeks after radiotherapy and tend to be transient. Late effects occur months to years after treatment and tend to be permanent. Common acute side effects include dermatitis, mucositis, taste changes, xerostomia, fatigue, facial hair loss, decreased sweating, anorexia, and weight loss. Less common acute effects include cough, hoarseness, nausea, and sialadenitis. Common late effects include xerostomia, trismus, hypothyroidism, soft tissue fibrosis, dysphagia, and taste changes. Less common late effects include soft tissue necrosis, osteoradionecrosis, laryngeal edema, spinal cord myelopathy, carotid stenosis, and second malignancy. Acute effects are generally managed supportively because of their transient nature. Aggressive dental support, stretching exercises, and proper skin care can minimize some late effects. Routine evaluation for hypothyroidism and xerostomia should also be performed, as pharmacologic interventions can improve these conditions.
Improving the Therapeutic Ratio of Radiation
Radiation can be improved by sensitizing tumor cells preferentially or by decreasing radiation damage to normal tissues. Hyperfractionation and accelerated radiation regimes have improved outcomes in stage III or IV SCC compared with standard fractionation, and hypofractionation has improved local control in early-stage glottic lesions. Radiation sensitizers with proven efficacy in randomized trials include concurrent platinum agents, mitomycin C, and cetuximab. Normal tissues can be spared using IMRT, submandibular gland transfer, and amifostine. Future improvements are expected as imaging, radiation delivery, and new agents continue to be further developed.
Further Reading
Chou RH, Wilder RB. Advances in radiotherapy for head and neck cancer. In: Donald PJ, ed. The Difficult Case in Head and Neck Cancer Surgery. Stuttgart/New York: Thieme; 2010:428–444
Isaacson SR, Close LG. Clinical radiation biology and radiotherapy. In: Van de Water TR, Staecker H, eds. Otolaryngology: Basic Science and Clinical Review. Stuttgart/New York: Thieme; 2006:158–163
Sciubba JT, Goldenberg D. Oral complications of radiation therapy. Lancet 2006;7(2): 175–183
5.2.3 Sinonasal Cancer
Key Features
• Sinonasal cancer initially may mimic benign sinus disease.
• Tumors of the paranasal sinuses often present with advanced disease.
• Cure rates are generally ≤50%.
• Most patients die of direct extension into vital areas.
Malignant tumors of the sinonasal tract are extremely rare, accounting for 0.2% of all invasive cancers and 3% of head and neck cancers. Cancers of the maxillary sinus are the most common. Tumors of the ethmoid sinuses are less common (20%), and cancers of the sphenoid and frontal sinuses are rare (<1%). Local extension often makes it difficult to access the sinus of origin.
Epidemiology
Chemical carcinogens such as chromium, nickel, thorium dioxide, and tanning chemicals have been implicated in the development of carcinoma of the paranasal sinuses. Exposure to wood dust has been implicated specifically in adenocarcinoma of the ethmoid. Interestingly, tobacco use was previously thought not to play a role in sinonasal carcinogenesis. However, up to a fivefold increased risk of sinonasal carcinoma has been observed with heavy smoking. Rarely, sinonasal cancers may present as a second primary tumor in tobacco users with other head and neck cancers.
Clinical
Signs and Symptoms
Clinical presentation of sinus malignancies is nonspecific and often mimics benign disease, thus diagnosis is often delayed for months. Key indicators of malignancy are cranial neuropathies, proptosis, and pain of maxillary dentition; trismus, palatal, and alveolar ridge fullness; or frank erosion into the oral cavity. Symptoms include nasal obstruction, discharge, stuffiness, congestion, epistaxis, unilateral tearing, diplopia, exophthalmos, infraorbital nerve hypesthesia, cheek swelling, facial asymmetry, hearing loss, and serous otitis media due to nasopharyngeal extension may occur.
Differential Diagnosis
The differential diagnosis includes benign sinus disease, benign sinus tumors, and metastatic tumors to the sinus.
Evaluation
History
The patient history should include known carcinogen exposure, tobacco usage, and prolonged benign sinus symptoms and signs.
Physical Exam
A complete head and neck examination, including nasal endoscopy, should be performed. The sinonasal, ocular, and neurologic systems should be studied in detail. Evidence of nerve hypesthesia, diplopia, proptosis, and loose dentition should be carefully evaluated. Suspicious lesions should be biopsied.
Imaging
Imaging should include either a contrast enhanced CT scan or MRI. There may be a role for integrated FDG-PET/CT.
Other Tests
A definitive diagnosis requires a biopsy. Special attention should be paid to CN function because malignant paranasal tumors are associated with a high incidence of cranial neuropathies compared with inflammatory or benign sinus disease.
Pathology
SCC is the most frequent type of malignant tumor in the paranasal sinuses (70–80%). Minor salivary gland tumors constitute 10 to 15% of these neoplasms. Some 5% of cases are lymphomas. Other tumors include sinonasal undifferentiated carcinoma (SNUC), chondrosarcoma, osteosarcoma and malignant melanoma, and esthesioneuroblastoma.
Inverted papilloma, a benign tumor with a tendency to recur (see Chapter 3.4), may transform into a malignant SCC of the paranasal sinuses in a small percentage of cases.
Treatment Options
Most stage T1 or T2 maxillary sinus carcinomas are treated by surgery alone, provided adequate resection margins are obtained. This may be en bloc surgical resection or endoscopic sinus surgery, depending on the extent of disease and experience of the surgeon. The specific approach is determined by the location of disease and histology (Fig. 5.6).
T3 and T4 lesions are treated by combination therapy with surgery and radiation. The issue regarding whether radiation is more effective before or after surgery remains controversial. Chemotherapy alone is generally used as a palliative measure.
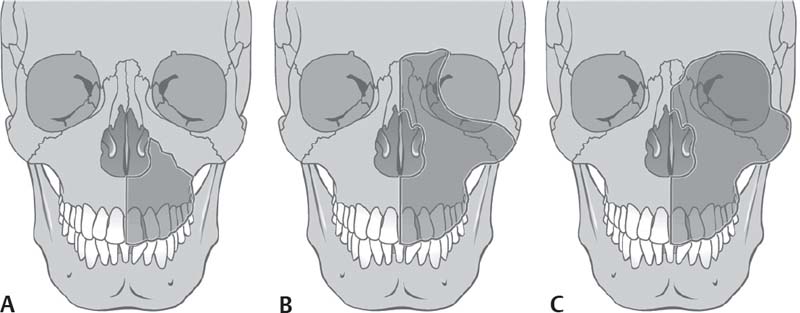
Fig. 5.6 Resection of the upper jaw. (A) Partial resection. (B) Total resection. (C) Total resection with exenteration of the orbit. (From Behrbohm H, Kaschke O, Nawka T, Swift A. Ear Nose and Throat Diseases: With Head and Neck Surgery. 3rd ed. Stuttgart/New York: Thieme; 2009:227.)
Outcome and Follow-Up
Malignancy behind Öhngren’s plane is regarded to carry a much poorer prognosis because of the rapid spread to the orbit and middle cranial fossae (Fig. 5.7). Despite improvements in surgical ablative and reconstructive techniques, radiation delivery modalities, and imaging technologies, disease-free survival at 5 years remains <50%, independent of stage. Five-year disease-free survival for patients with advanced stage cancer drops to 25%.
ICD-9 Codes
160.2 | Malignant neoplasm of maxillary sinus |
160.3 | Malignant neoplasm of ethmoidal sinus |
160.4 | Malignant neoplasm of frontal sinus |
160.5 | Malignant neoplasm of sphenoidal sinus |
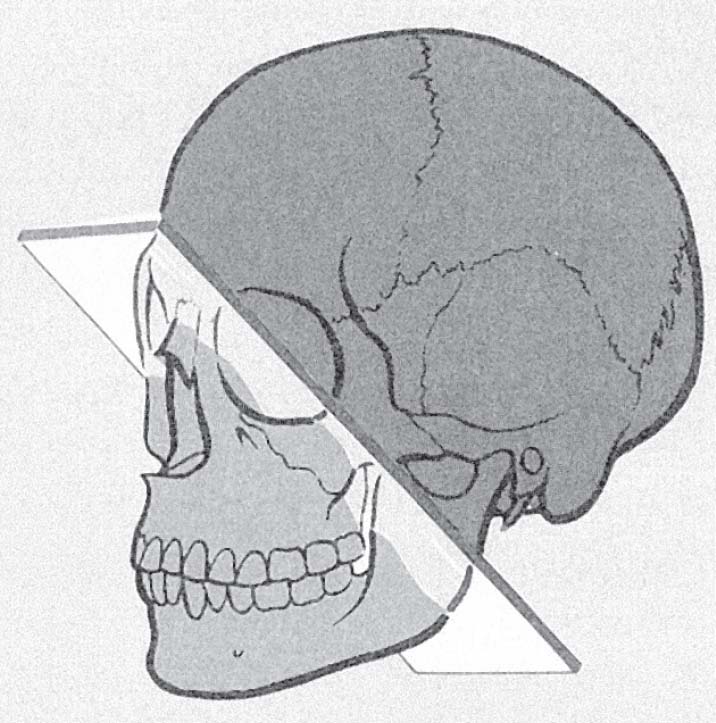
Fig. 5.7 Öhngren’s plane passing through the medial canthus and the mandibular angle. It divides the maxillary sinus into a superoposterior part and an inferoanterior part. Cancer limited to the latter part typically carries a better prognosis. (From Becker W, Naumann HH, Pfaltz CR. Ear, Nose, and Throat Diseases: A Pocket Reference. 2nd ed. Stuttgart/New York: Thieme; 1994:293.)
Staging of Nose and Paranasal Sinus Cancer: For All Carcinomas Excluding Mucosal Malignant Melanoma
Primary Tumor: Maxillary Sinus
TX: Cannot be assessed
T0: No evidence of primary tumor
Tis: Carcinoma in situ
T1: Tumor limited to the maxillary sinus mucosa with no erosion or destruction of bone
T2: Tumor causing bone erosion or destruction including extension into the hard palate and/or middle nasal meatus, except extension to posterior wall of maxillary sinus and pterygoid plates
T3: Tumor invades any of the following: bone of the posterior wall of maxillary sinus, subcutaneous tissues, floor or medial wall of orbit, pterygoid fossa, or ethmoid sinuses
T4a: Tumor invades anterior orbital contents, skin of cheek, pterygoid plates, infratemporal fossa, cribriform plate, or sphenoid or frontal sinuses
T4b: Tumor invades any of the following: orbital apex, dura, brain, middle cranial fossa, cranial nerves other than maxillary division of trigeminal nerve (V2), nasopharynx, or clivus
Primary Tumor: Nasal Cavity and Ethmoid Sinus
TX: Cannot be assessed
T0: No evidence of primary tumor
Tis: Carcinoma in situ
T1: Tumor restricted to any one subsite, with or without bone invasion
T2: Tumor invading two subsites in a single region or extending to involve an adjacent region within the nasoethmoidal complex, with or without bone invasion
T3: Tumor extends to invade the medial wall or floor of the orbit, maxillary sinus, palate, or cribriform plate
T4a: Tumor invades any of the following: anterior orbital contents, skin of nose or cheek, minimal extension to anterior cranial fossa, pterygoid plates, or sphenoid or frontal sinuses
T4b: Tumor invades any of the following: orbital apex, dura, brain, middle cranial fossa, cranial nerves other than maxillary division of trigeminal nerve (V2), nasopharynx, or clivus
Regional Lymph Nodes*
NX: Cannot be assessed
N0: No regional lymph node metastasis
N1: Metastasis in a single ipsilateral lymph node, ≤3 cm in greatest dimension
N2a: Metastasis in a single ipsilateral lymph node, >3 cm but not >6 cm in greatest dimension
N2b: Metastasis in multiple ipsilateral lymph nodes, none >6 cm in greatest dimension
N2c: Metastasis in bilateral or contralateral lymph nodes, none >6 cm in greatest dimension
N3: Metastasis in a lymph node >6 cm in greatest dimension
Distant Metastasis
M0: No distant metastasis
M1: Distant metastasis
Stage Groupings: For All Cancers Except Mucosal Malignant Melanoma
Stage 0
Tis N0 M0
Stage I
T1 N0 M0
Stage II
T2 N0 M0
Stage III
T1 N1 M0
T2 N1 M0
T3 N0,N1 M0
Stage IVA
T1,T2,T3 N2 M0
T4a N0,N1,N2 M0
Stage IVB
T4b Any N M0
Any T N3 M0
Stage IVC
Any T Any N M1
Further Reading
American Joint Committee on Cancer. AJCC Cancer Staging Handbook. 7th ed. New York: Springer-Verlag; 2010
Donald PJ. Cancer of the nose and paranasal sinuses. In: Donald PJ, ed. The Difficult Case in Head and Neck Cancer Surgery. Stuttgart/New York: Thieme; 2010: 212–259
Goldenberg D, Golz A, Fradis M, Mârtu D, Netzer A, Joachims HZ. Malignant tumors of the nose and paranasal sinuses: a retrospective review of 291 cases. Ear Nose Throat J 2001;80(4):272–277
Imola MJ, Schramm VL Jr. Orbital preservation in surgical management of sinonasal malignancy. Laryngoscope 2002;112(8 Pt 1):1357–1365
5.2.4 Nasopharyngeal Cancer
Key Features
• There is a high frequency of nasopharyngeal cancer (NPC) among patients of Chinese ethnicity and descent.
• It is associated with EBV exposure.
• The diagnosis must be excluded in patients with asymptomatic cervical lymphadenopathy and unilateral serous otitis media.
NPC is a distinct type of head and neck cancer that differs from other malignancies of the upper aerodigestive tract with respect to epidemiology, pathology, clinical presentation, and responses to treatment. NPC is an uncommon neoplasm in most parts of the world but is endemic in East Asia. Seventy percent of patients with newly diagnosed NPC present with locally advanced disease. Latent EBV infection seems to be crucial in the pathogenesis of NPC. Studies have established that NPC cells express two distinct EBV latent membrane proteins, LMP-1 and LMP-2. These proteins are attractive targets for adaptive immunotherapy.
Anatomy
The nasopharynx is bounded superiorly by the basiocciput and basisphenoid, posteriorly by the C1 and C2 cervical bodies, anteriorly by the choanae, and inferiorly by the soft palate. The lateral walls are occupied primarily by the eustachian tube orifice. Immediately posterior to the eustachian tube orifice is Rosenmüller’s fossa, where most nasopharyngeal carcinomas originate.
Epidemiology
NPC occurs most often in China where it is the third most common malignancy among men, with an incidence rate of 15 to 50 per 100,000. NPC rates are also high in Vietnamese and Filipino men. There is an intermediate incidence in Inuit Eskimos and in the populations of the Mediterranean basin. Emigration from high- to low-incidence areas reduces the incidence of NPC in first-generation Chinese, but incidence still remains at seven times the rate seen in Caucasians. NPC occurs in younger patients, including children, but the peak incidence is seen in people aged 55 to 64 years. Males are affected three times more often than females.
Clinical
Signs and Symptoms
Early signs and symptoms are subtle and variable, and are often initially ignored by both patient and physician. Cervical lymphadenopathy is the most common sign (50–90% of patients), followed by blood-stained nasal discharge or epistaxis, unilateral serous otitis media, and cranial neuropathies (most often CN VI, followed by CN V). Five to 7% of all patients have systemic metastases at presentation, most often to bone.
Symptoms include unilateral nasal obstruction, unilateral hearing loss and otalgia, diplopia, facial or neck pain, and paresthesia.
Differential Diagnosis
• Minor salivary gland tumors
• Juvenile nasopharyngeal angiofibroma
• Adenoid hypertrophy
• Tornwaldt cysts
• Fibromyxomatous polyps
• Choanal polyps, fibromas
• Papillomas
• Osseous/fibroosseous tumors
• Craniopharyngiomas
• Extracranial meningiomas
• Chordomas
Evaluation
History
History should include questions about epistaxis, nasal obstruction and discharge, hearing loss or clogged ear, headache, diplopia, facial pain, and numbness.
Physical Exam
The physical exam should include a fiberoptic nasopharyngoscopy. The neck should be examined in a systematic fashion. Any lymph nodes should be assessed with regard to size, location, and mobility.
Imaging
Imaging is required for staging and treatment planning for NPC. CT and MRI are recommended for the diagnostic process and evaluation of tumoral extent, bone erosion, and to delineate tumor extension into the parapharyngeal and retropharyngeal spaces, the oropharynx, the orbit, and the intracranial compartment.
A chest x-ray, liver ultrasound, and a bone scan are recommended for all patients with nodal disease. The role of PET in the staging of nasopharyngeal carcinoma has not been well established.
Labs
The quantitative analysis of cell-free EBV DNA in plasma of patients has been studied, and data suggest a possible value of this tool in screening and monitoring treatment. High IgA antibody levels to EBV capsid antigen and early antigen (EA) provide a valuable screening tool for early cases in high-incidence populations.
Other Tests
A dental examination is required before instituting radiotherapy to reduce the development of postradiotherapy complications. Patients should continue with meticulous dental care and fluoride prophylaxis.
Pathology
Classic nasopharyngeal carcinoma has been classified into three types by the World Health Organization (WHO):
Type 1: Keratinizing SCC
Type 2: Nonkeratinizing or poorly differentiated carcinoma
Type 3: Undifferentiated carcinoma (lymphepithelioma)
WHO types II and III exhibit between 82 to 100% positivity with respect to EBV antibody titers. Type 1 may have an association with cigarette and alcohol consumption and accounts for up to 30% of cases in nonendemic areas and <5% in endemic areas. Several genetic markers of the HLA system have been investigated in patients with NPC in China and other parts of Asia. HLA-A2 and HLA-BS-in-2 were associated with an increased incidence whereas HLA-A11 was associated with a decreased risk.
Treatment Options
Medical
Radiotherapy is the cornerstone of the definitive treatment for NPC. This is because NPCs are particularly radiosensitive, and the tumor is in a relatively inaccessible location, making surgical excision difficult and highly morbid. External beam is most commonly delivered by opposed lateral fields to encompass the primary tumor and upper neck. Radiation doses of 70 to 76 Gy in fractions of 1.8 to 2.0 Gy per day to the primary and anatomic structures at risk within the vicinity of the nasopharynx. Because there is a high incidence of subclinical neck disease, radiation doses between 50 and 60 Gy are used to electively treat the neck.
Recent data shows a clear role for concomitant chemoradiotherapy followed by adjuvant chemotherapy, which provides statistically significant improvement in overall survival and disease free survival.
EBV DNA titers seem to be an important index for prognostication. EBV DNA titers correlate with stage, treatment response, relapse, and survival.
Adoptive immunotherapy with EBV-specific CTL awaits further exploration.
Surgical
The role of surgery in NPC is largely confined to the treatment of residual or recurrent disease either in the nasopharynx or in the neck. Neck dissection for postradiation residual or recurrent nodal disease is the most common indication for surgery.
Outcome and Follow-Up
Overall survival with the use of conventional radiotherapy alone in the treatment of NPC is in the range of 50 to 76%. Patients with stage I and II disease have a high rate of cure with radiotherapy alone. Seventy percent of patients with NPC present with locally advanced stage III or IV disease. For these patients, radiotherapy delivered in combination with chemotherapy has become the standard of care. The prognosis for those with distant metastatic spread remains poor.
A first complete evaluation should be performed 2 to 3 months after completion of treatment. The next evaluations should be scheduled for 6 months after this first posttherapeutic workup and on a yearly basis thereafter.
ICD-9 Code
147 | Malignant neoplasm of nasopharynx |
Staging of Nasopharyngeal Cancer
Primary Tumor: Nasopharynx
TX: Cannot be assessed
T0: No evidence of primary tumor
Tis: Carcinoma in situ
T1: Tumor confined to nasopharynx, or tumor extends to oropharynx and/or nasal cavity without parapharyngeal extension*
T2: Tumor with parapharyngeal extension*
T3: Tumor involves bony structures of skull base and/or paranasal sinuses
T4: Tumor with intracranial extension and/or involvement of CNs, hypopharynx, orbit, or with extension to the infratemporal fossa/masticator space
*Parapharyngeal extension denotes posterolateral infiltration of tumor.
Regional Lymph Nodes (N): Nasopharynx†
NX: Cannot be assessed
N0: No regional lymph node metastasis
N1: Unilateral metastasis in lymph node (s), ≤6 cm in greatest dimension, above the supraclavicular fossa
N2: Bilateral metastasis in lymph node (s), ≤6 cm in greatest dimension, above the supraclavicular fossa
N3: Metastasis in a lymph node >6 cm and/or to supraclavicular fossa
N3a: >6 cm in dimension
N3b: Extension to the supraclavicular fossa
Distant Metastasis (M)
MX: Distant metastasis cannot be assessed
M0: No distant metastasis
M1: Distant metastasis
American Joint Committee on Cancer Stage Groupings for Nasopharynx Cancers Except Mucosal Malignant Melanoma
Stage 0
Tis N0 M0
Stage I
T1 N0 M0
Stage II
T2 N0 M0
Stage III
T1 N1 M0
T2 N1 M0
T3 N0 M0
T3 N1 M0
Stage IVA
T1 N2 M0
T2 N2 M0
T3 N2 M0
T4a N0 M0
T4a N1 M0
T4a N2 M0
Any T N3 M0
T4b any N M0
Stage IVC
Any T any N M1
Further Reading
Agulnik M, Siu LL. State-of-the-art management of nasopharyngeal carcinoma: current and future directions. Br J Cancer 2005;92(5):799–806
Al-Sarraf M, LeBlanc M, Giri PG, et al. Chemoradiotherapy versus radiotherapy in patients with advanced nasopharyngeal cancer: phase III randomized Intergroup study 0099. J Clin Oncol 1998;16(4):1310–1317
American Joint Committee on Cancer. AJCC Cancer Staging Handbook. 7th ed. New York: Springer-Verlag; 2010
Wei WI, Sham JST. Carcinoma of the nasopharynx. In: Genden EM, Varvares MA, eds. Head and Neck Cancer: An Evidence-Based Team Approach. Stuttgart/New York: Thieme; 2008:137–151
5.2.5 Oral Cavity Cancer
Key Features
• A nonhealing or bleeding sore in the mouth or on the lip is the most common presentation.
• A persistent white or red patch on oral mucosa needs to be investigated.
• History of tobacco smoking or chewing occurs in most cases.
• Lip cancer may be caused by sun exposure.
The oral cavity extends from the skin–vermilion junctions of the anterior lips to the junction of the hard and soft palates superiorly and to the line of circumvallate papillae posteriorly. Oral cancer should be identified early and screening is useful. It is frequently preceded by an identifiable premalignant lesion. The progression from dysplasia may occur over a period of years.
Epidemiology
Thirty thousand people are diagnosed yearly with oral cancer in the United States, and it will cause 8000 deaths. It constitutes 5% of US cancers, but in India, the incidence is far greater. For all stages combined, the 5-year relative survival rate is 59% and the 10-year survival rate is 44%. It typically occurs in those over the age of 45, and occurs in men twice as often as women. The number of new cases of this disease has been decreasing during the past 20 years. Tobacco smoking and alcohol are the primary risk factors. Smokeless tobacco in the Western world and paan (betel leaf with areca nut) in Asia are also risk factors for oral cancer. Other suspected risk factors include viral infection (HPV), a diet low in fruits and vegetables, vitamin A deficiency, genetic susceptibility, immunosuppressive drugs or immunocompromised conditions. Recently there has been a growing number of young patients with oral cancers, particularly involving the tongue.
Clinical
Signs
Signs may include uncomfortable or poorly fitting dentures, loosening of the teeth, changes in articulation, a mass in the neck, weight loss, and persistent halitosis.
Symptoms
Symptoms depend on site and stage of the primary tumor and its effect on function of that area. They include a nonhealing white or red (leukoplakia, erythroplasia) patch or sore in the mouth (most common symptom), persistent pain in the mouth, and a thickening in the cheek or floor of mouth. More advanced disease may cause a sore throat, difficulty chewing, dysphagia, trismus or tongue tethering, numbness of the tongue or mouth, and pain around the teeth or jaw.
Differential Diagnosis
• Oral and pharyngeal infections such as pharyngitis or stomatitis
• Chancre
• Benign oral or odontogenic lesions
• Denture sores
• Aphthous ulcers or herpetic sores
• Lesion due to cheek biting
• Oral manifestations of systemic diseases
• Necrotizing sialometaplasia
Evaluation
History
Evaluation begins with a detailed history inquiring about tobacco and alcohol usage, oral pain, referred otalgia, dysphagia, articulation changes, and weight loss.
Physical Exam
A physical exam should include a complete head and neck exam. Specific attention should be directed at the site of the lesion. The lesion size should be noted as should its infiltration and spread to adjacent oral cavity or oropharyngeal subsites such as floor of mouth, alveolus, and tongue base. A bimanual examination of the lesion, the surrounding floor of mouth, and the submandibular triangle should be performed.
Careful palpation of the neck may reveal adenopathy. The main routes of lymph node drainage from the oral cavity are into the first echelon nodes (i.e., buccinator, jugulodigastric, submandibular, and submental). Tumor sites close to the midline commonly drain bilaterally.
Imaging
Contrast-enhanced CT of the head and neck is a necessary component of initial evaluation. Tumor size and spread may be evaluated as well as discrete nodal disease, bony destruction, and vascular involvement. MRI may be helpful in the evaluation of oral cancer because it provides a higher contrast between normal tissue and tumor on T2-weighted images, has no beam artifact from dental material, and provides multiplanar imaging.
The combination of PET and CT is a useful diagnostic and staging modality in the evaluation of the patient with head and neck cancer.
A chest CT may be used to rule out pulmonary metastasis. Periapical dental radiographs provide fine detail and may show minimal invasion. Panoramic dental radiographs may show gross bony destruction.
Labs
Routine preoperative laboratory studies are employed.
Other Tests
Patients with suspected oral cancer must undergo a biopsy for pathologic diagnosis. The first test for the evaluation of a neck mass presenting with an oral cavity lesion is FNAB.
The routine use of panendoscopy, which includes bronchoscopy, esophagoscopy, and laryngoscopy, is recommended. It allows for the complete evaluation of the upper aerodigestive tract and helps rule out the presence of a synchronous tumor. The mucous membranes of the upper aerodigestive tract are carefully evaluated, and biopsy samples of any abnormal-looking areas. This is particularly important in the patient who smokes.
Toluidine blue staining and photodynamic agents such as Foscan (Sigma-Aldrich, St. Louis, MO) and 5-aminolevulinic acid (ALA) may be used to enhance detection of oral lesions. Oral brush biopsy (OralCDx; Oral CDx Laboratories, Inc., Suffern, NY) can be used in the screening of oral precancer and cancer.
A dental evaluation should be performed, with attention to dental hygiene, dentition status, and integrity of the mandible.
Pathology
Ninety percent of oral cancers are SCCs, and they may be preceded by various precancerous lesions. Non-SCC oral cancers may also represent minor salivary gland tumors. Other rarer cancers may be of odontogenic apparatus origin, lymphomas, soft tissue sarcomas, and melanomas.
There is no significant correlation between degree of squamous differentiation (i.e., grading of dysplasia) and the biologic behavior of oral cancer (Table 5.6). Vascular and perineural invasion thickness and depth of invasion are all negative prognostic factors.
Verrucous carcinoma (VC) refers to a clinicopathologic concept implying a locally aggressive, clinically exophytic, low-grade, well-differentiated SCC with minimal metastatic potential. It is also known as Ackerman tumor. An HPV infection is thought to facilitate or cause verrucous carcinoma. Associations with verrucous carcinoma have been found in patients who chewed tobacco and betel nut.
Fully developed lesions are white cauliflower-like papillomas with a pebbly surface that may extend and coalesce over large areas of the oral mucosa. Overall, patients with verrucous carcinoma have a favorable prognosis; the course of verrucous carcinoma lesions is characterized by slow, continuous local growth.
Proliferative verrucous leukoplakia (PVL) is a particularly aggressive form of oral leukoplakia that commences with a hyperkeratosis, spreads to become multifocal and verruciform in appearance, and later becomes malignant. It is significant because it has a high recurrence rate and the potential to develop into verrucous carcinoma or SCC in 60 to 70% of the affected patients. PVL is more commonly found in elderly females and is associated with tobacco use or alcohol abuse in 30 to 50% of patients. The etiology of PVL is unknown. An association with HPV infection, particularly strains 16 and 18, has been implicated in some cases. The most common locations are the gingiva or alveolar ridge tongue, and buccal mucosa sites. The gingiva is the most likely site for the malignant transformation of PVL. PVL often begins as a focal lesion spreading laterally over time and can be multifocal. Early in its course it is a flat hyperkeratotic lesion that becomes progressively verrucous and histologically often exhibits varying degrees of epithelial dysplasia.
G1: Well differentiated |
G2: Moderately well differentiated |
G3: Poorly differentiated |
G4: Undifferentiated |
Note : No statistically significant correlation between degree of differentiation and the biologic behavior of the cancer exists; however, vascular invasion is a negative prognostic factor.
Treatment Options
Surgical resection and radiotherapy are the current treatments of choice.
Lip Cancer
Most lip SCCs present on the lower lip (88–95%), 2 to 7% present on the upper lip, and 1% on the oral commissure. Basal cell carcinoma is more common on the upper lip.
Treatment of Lip Cancer
• For T1 and T2 lesions, radiotherapy and surgery produce similar cure rates; the method of treatment is determined by functional and cosmetic factors.
• Advanced lesions of the lip generally require a combination of surgery and radiotherapy.
• Patients with upper lip and oral commissure SCC have a worse overall prognosis.
• The 5-year survival for stage I and II lesions is 90%.
Oral Tongue Cancer
Seventy-five percent of tongue cancers occur on the posterior lateral aspect, 20% on the anterior lateral aspect, and 3 to 5% on the lingual dorsum. At the time of diagnosis, 75% oral tongue cancers are T2 or smaller. Forty percent of patients with oral tongue cancer demonstrate clinical evidence of neck metastasis at presentation.
Treatment of Oral Tongue Lesions
• Early tongue cancer: Wide local excision is often used for T1 lesions that can be resected transorally.
• For larger T1 and T2 lesions, either surgery or radiotherapy are acceptable treatments.
• Deeply infiltrative lesions (>4 mm depth) can be treated with surgery with postoperative radiotherapy and a selective neck dissection.
• Selected patients with T4 tongue cancer can be treated with combined surgery (i.e., total glossectomy, sometimes requiring laryngectomy due to the high risk of postoperative aspiration) and postoperative radio-therapy.
• For T1 and T2 lesions, 20 to 30% of patients harbor metastatic disease in cervical lymph nodes. Thus, therapy (surgery or radiotherapy) aimed at the neck should be considered as part of definitive treatment.
• The 5-year survival is 75% for stage I and II oral tongue cancers and <40% for stage III and IV oral tongue cancers.
Buccal Mucosa Cancer
Carcinomas of the buccal mucosa represent 5 to 10% of oral cancers. The most common area is in the region of the third mandibular molar. Lesions <1 cm in diameter can be treated by surgery alone if the oral commissure is not involved. If involved, radiotherapy should be considered. Premalignant conditions include submucosal fibrosis and lichen planus. The latter has a reported transformation rate of 0.5 to 3%, whereas the former has a malignant transformation rate of 0.5%.
Treatment of Buccal Mucosa Cancers
• Lesions smaller than 1 cm in diameter can be managed by surgery alone if the commissure is not involved. If the commissure is involved, radio-therapy (including brachytherapy) should be considered.
• Advanced lesions of the buccal mucosa can be treated with surgical resection alone, radiotherapy alone, or surgical resection plus postoperative radiation.
• The 5-year survival for buccal mucosa cancer is 75% for stage I, 65% for stage II, 30 to 65% for stage III, and 20 to 50% for stage IV buccal cancer.
Floor of Mouth Cancer
Cancers of the floor of mouth represent 28 to 35% of oral cancers. Thirty-five percent of patients with floor of mouth cancer present with T3 or T4 disease. The most common presentation of cancer of the floor of the mouth is a painless inflamed superficial ulcer with poorly defined margins. Preexistent or coincident leukoplakia can be observed in adjacent tissues in ~20% of cases.
Treatment of Floor of Mouth Cancer
Note that a cancer involving the gingiva adjacent to recent dental extraction is at high risk for bony extension via the tooth socket.
• For T1 lesions either transoral surgery or radiotherapy is an acceptable treatment.
• For small T2 lesions (≤3 cm), surgery is often used if the lesion is attached to the periosteum, whereas radiotherapy is often used if the lesion encroaches on the tongue.
• For large T2 lesions (>3 cm), surgery and radiotherapy are alternative methods of treatment, the choice of which depends primarily on the expected extent of disability from surgery.
• External-beam radiotherapy with or without interstitial radiotherapy should be considered postoperatively for larger lesions.
• For more advanced lesions, surgery should incorporate rim resection plus neck dissection or partial mandibulectomy with neck dissection as appropriate.
• The 5-year survival for floor of mouth cancer is 90% for stage I, 80% for stage II, 65% for stage III, and 30% for stage IV.
Retromolar Trigone Cancer
Retromolar trigone cancers account for ~10% of all oral cancers. These cancers typically present with advanced disease, and 50% of patients have regional metastasis at the time of diagnosis.
Treatment of Retromolar Trigone Cancer
For small lesions without detectable bone invasion, limited resection of the mandible may be performed. Radiotherapy may be used initially, with surgery reserved for radiation failure. Selective neck treatment should be performed—for advanced stages, multimodality therapy with surgery and postoperative radiation is most often used.
Hard Palate Cancer
Cancer of the hard palate accounts for 5% of all oral cavity malignancies. Ten to 25% of patients with head and neck SCC of the hard palate present with regional metastasis. (Only 53% of hard palate cancers are SCC; minor salivary gland malignancies make up the rest.)
Treatment of Cancer of the Hard Palate
For both early and advanced disease, surgery (inferior maxillectomy with surgical obturator) is used for primary therapy. Radiotherapy has a role depending on factors such as close or positive surgical margins, evidence of perineural involvement or the presence of lymph node metastases. The prosthodontist is important in the care of these patients for oral rehabilitation. The 5-year survival for hard palate cancer ranges from 40 to 60%.
Advanced Oral Cavity Cancer
Clinical trials for advanced oral tumors evaluating the use of chemotherapy preoperatively, before radiotherapy, as adjuvant therapy after surgery, or as part of combined modality therapy are appropriate.
Outcome and Follow-Up
Patients should be seen at 2-month intervals for 2 years after treatment. Thereafter they should be seen at 3-month intervals for the next year and then every 6 months for another year. Patients with regional neck disease prior to treatment should have undergo CT scan or integrated FDG-PET/CT 12 weeks after the completion of radiotherapy to assess for residual disease that may necessitate postradiotherapy neck dissection. The risk of a second primary carcinoma is highest in those who continue to smoke, and patients should be strongly urged to quit.
ICD-9 Codes
140–149 | Malignant neoplasm of lip, oral cavity, and pharynx |
140 | Malignant neoplasm of lip |
141 | Malignant neoplasm of tongue |
143 | Malignant neoplasm of gum, includes alveolar (ridge) mucosa gingiva (alveolar) (marginal) interdental papillae |
Malignant neoplasm of floor of mouth | |
145 | Malignant neoplasm of other and unspecified parts of mouth |
149 | Malignant neoplasm of other and ill-defined sites within the lip, oral cavity, and pharynx |
Staging of Oral Cavity Cancer
Primary Tumor
TX: Primary tumor cannot be assessed
T0: No evidence of primary tumor
Tis: Carcinoma in situ
T1: Tumor ≤2 cm in greatest dimension
T2: Tumor >2 cm but not >4 cm in greatest dimension
T3: Tumor >4 cm in greatest dimension
T4a: Moderately advanced local disease.
Lip: Tumor invades through the cortical bone, the inferior alveolar nerve, the floor of the mouth, or the skin of the face, i.e., the chin or nose. Oral cavity: Tumor invades the adjacent structures (e.g., through the cortical bone [mandible, maxilla], into the deep [extrinsic] muscle of the tongue [genioglossus, hyoglossus, palatoglossus, and styloglossus], the maxillary sinus, or the skin of the face).
T4b: Very advanced local disease. Tumor invades the masticator space, pterygoid plates, or skull base, and/or encases the internal carotid artery.
Regional Lymph Nodes*
NX: Regional lymph nodes cannot be assessed
N0: No regional lymph node metastasis
N2a: Metastasis in a single ipsilateral lymph node, >3 cm but not >6 cm in greatest dimension
N2b: Metastasis in multiple ipsilateral lymph nodes, none >6 cm in greatest dimension
N2c: Metastasis in bilateral or contralateral lymph nodes, none >6 cm in greatest dimension
N3: Metastasis in a lymph node >6 cm in greatest dimension
Distant Metastasis
M0: No distant metastasis
M1: Distant metastasis
American Joint Committee on Cancer Stage Groupings for Oral Cavity Cancers
Stage 0
Tis N0 M0
Stage I
T1 N0 M0
Stage II
T2 N0 M0
Stage III
T1 N1 M0
T2 N1 M0
T3 N0 M0
T3 N1 M0
Stage IVA
T1 N2 M0
T2 N2 M0
T3 N2 M0
T4a N0 M0
T4a N1 M0
T4a N2 M0
Stage IVB
Any T N3 M0
T4b any N M0
Stage IVC
Any T any N M1
Further Reading
American Joint Committee on Cancer. AJCC Cancer Staging Handbook. 7th ed. New York: Springer-Verlag; 2010
Donald PJ. Cancer of the oral cavity. In: Donald PJ, ed. The Difficult Case in Head and Neck Cancer Surgery. Stuttgart/New York: Thieme; 2010:1–43
Genden EM, Kao J, Packer SH. Carcinoma of the oral cavity. In: Genden EM, Varvares MA, eds. Head and Neck Cancer: An Evidence-Based Team Approach. Stuttgart/New York: Thieme; 2008:1–23
Goldenberg D, Ardekian L, Rachmiel A, Peled M, Joachims HZ, Laufer D. Carcinoma of the dorsum of the tongue. Head Neck 2000;22(2):190–194
National Cancer Institute. Lip and oral cavity cancer treatment. Available at: http://www.cancer.gov/cancertopics/pdq/treatment/lip-and-oral-cavity/healthprofessional Werning JW, ed. Oral Cancer: Diagnosis, Management, and Rehabilitation. Stuttgart/ New York: Thieme; 2007
5.2.6 Oropharyngeal Cancer
Key Features
• Oropharyngeal cancer includes cancer of the tonsil, tongue base, soft palate, and oropharyngeal wall.
• Oropharyngeal cancer is primarily linked to tobacco and alcohol use.
• HPV is a risk factor for cancer of the tonsil.
• Neck metastasis may be cystic.
The oropharynx is located between the soft palate superiorly and the hyoid bone inferiorly; it communicates with the oral cavity anteriorly, the nasopharynx superiorly and the supraglottic larynx and hypopharynx inferiorly. Oropharynx cancers are typically detected at a more advanced stage than oral cancer. The oropharynx is an important component in swallowing; therefore, treating these tumors is challenging and often requires a multidisciplinary approach and posttreatment rehabilitation.
Epidemiology
In the United States, an estimated 8300 new cases of pharyngeal cancer (including cancers of the oropharynx and hypopharynx) are diagnosed yearly, with an estimated mortality of 2000. It affects men three times more than women. Seventy-five percent of oropharynx cancers occur in the tonsil. Tobacco (including smokeless tobacco) and alcohol abuse represent the most significant risk factors for the development of oropharynx cancer. Viral infection with HPV is a risk factor for SCC of the tonsil and may be a positive prognostic factor.
Other risk factors include Yerba maté, a stimulant beverage commonly consumed in South America, and chewing of paan (betel leaf with areca nut), which is common in parts of Asia.
Clinical
Signs
Signs include changes in articulation, muffled speech, a mass in the neck, unintentional weight loss, hemoptysis, and persistent halitosis.
Symptoms
Symptoms may include pain, dysphagia, globus sensation, referred otalgia, trismus, and fixation of the tongue. Cancers of the base of the tongue and tonsil are typically insidious.
Differential Diagnosis
• Oropharyngeal infections such as pharyngitis or stomatitis
• Chancre
• Benign oropharyngeal or odontogenic lesions
• Aphthous ulcers or herpetic sores
• Oral manifestations of systemic diseases
Evaluation
History
Evaluation begins with a detailed history inquiring about tobacco and alcohol usage, sexual history, oral pain, odynophagia, referred otalgia, dysphagia, hemoptysis, articulation or speech changes, and unintentional weight loss.
Physical Exam
The physical exam should include a complete head and neck exam, with specific attention directed at the site of the lesion. The lesion size should be noted as should its apparent infiltration and spread to adjacent pharyngeal or oral cavity subsites such as oral tongue, hypopharynx, nasopharynx, and vallecula. Palpation of the lesion should be performed if possible in an awake patient. In advanced cases, discerning the primary origin of the lesion, such as the tongue base or tonsil, is not always possible. Fiberoptic laryngoscopy should be performed. Inspection and palpation of the neck often reveals adenopathy.
Imaging
Clinical staging may understage oropharynx tumors, especially the tongue base extension. Both contrast-enhanced CT and MRI are able to assess oropharynx tumor extent as well as regional spread. Neck metastasis from oropharynx cancer may be cystic in morphology; this finding by itself should raise suspicion of a cancer in the tonsil or tongue base.
At minimum, a chest CT should be used to rule out pulmonary metastasis or second primary. Alternatively, an integrated FDG-PET/CT may be used in a combined staging and metastatic workup.
Labs
Standard preoperative laboratories, as indicated, should be obtained.
Other Tests
Patients with suspected cancer of oropharynx must undergo a biopsy and a sample of the lesion taken for pathologic examination. This may be done in an office setting in cases of tonsil cancer and soft palate cancer, but is not usually possible in cases of tongue base lesions. If neck metastases are evident, they should be sampled by FNAB.
Pathology
Histologically, 90% of oropharynx cancers are SCCs. Basaloid SCC is an uncommon but aggressive SCC variant. Other cancers of the oropharynx include minor salivary gland carcinomas, lymphomas, and “lymphoepithelial-like” carcinomas.
Treatment Options
For stage I oropharynx cancer, surgery or radiotherapy may be used depending on the expected functional deficit. Radiation clinical trials evaluating hyperfractionation schedules should be considered.
For stage II oropharynx cancer, surgery or radiation therapies are equally successful in controlling disease. Radiotherapy may be the preferred modality where the functional deficit is expected to be great.
The management of stage III oropharynx cancer is complex and requires a multidisciplinary approach to establish the optimal treatment. A combination of surgery with postoperative radiotherapy and/or chemotherapy is most often used. An alternative is chemoradiation therapy alone based on the patient’s initial nodal involvement.
In stage III tonsil cancer, hyperfractionated radiotherapy yields a higher control rate than standard fractionated radiotherapy.
In advanced unresectable oropharyngeal cancer, radiotherapy or chemo-radiation is used.
Treatments currently under investigation include chemotherapy with radiation clinical trials as well as with radiosensitizers, radiation clinical trials evaluating hyperfractionation schedules and/or brachytherapy, particle-beam radiotherapy, and hyperthermia combined with radiotherapy.
Recently, transoral robotic-assisted resection (TORS) of select oropharyngeal tumors has gained momentum.
Outcome and Follow-Up
The overall 5-year disease-specific survival for patients with all stages of disease is ~50%. Patients with cancer of the oropharynx should have a careful head and neck examination to examine for recurrence monthly for the first posttreatment year, every 2 months for the second year, every 3 months for the third year, and every 6 months to a year thereafter.
ICD-9 Codes
141 | Malignant neoplasia of base of tongue |
145.3 | Malignant neoplasia of soft palate |
146 | Malignant neoplasia of oropharynx/tonsil |
Malignant neoplasia of tonsil fossa | |
146.2 | Malignant neoplasia of tonsillar pillars |
146.3 | Malignant neoplasia of vallecula epiglottica |
146.6 | Malignant neoplasia of lateral wall of oropharynx |
146.7 | Malignant neoplasia of posterior wall of oropharynx |
Staging of Oropharyngeal Cancer
Primary Tumor
TX: Primary tumor cannot be assessed
T0: No evidence of primary tumor
Tis: Carcinoma in situ
T1: Tumor ≤2 cm in greatest dimension
T2: Tumor >2 cm but not >4 cm in greatest dimension
T3: Tumor >4 cm in greatest dimension or extension to the lingual surface of epiglottis
T4a: Moderately advanced local disease. Tumor invades the larynx, the deep/extrinsic muscle of tongue, the medial pterygoid muscles, hard palate, or the mandible.*
T4b: Very advanced local disease. Tumor invades the lateral pterygoid muscle, pterygoid plates, the lateral nasopharynx or skull base, or encases the carotid artery.
Regional Lymph Nodes
NX: Regional lymph nodes cannot be assessed
N0: No regional lymph node metastasis
N1: Metastasis in a single ipsilateral lymph node, ≤3 cm in greatest dimension
N2a: Metastasis in a single ipsilateral lymph node, >3 cm but not >6 cm in greatest dimension
N2b: Metastasis in multiple ipsilateral lymph nodes, none >6 cm in greatest dimension
N2c: Metastasis in bilateral or contralateral lymph nodes, none >6 cm in greatest dimension
N3: Metastasis in a lymph node >6 cm in greatest dimension
Distant Metastasis
MX: Distant metastasis cannot be assessed
M0: No distant metastasis
M1: Distant metastasis
American Joint Committee on Cancer Stage Groupings of Oropharyngeal Cancer
Stage 0
Tis N0 M0
Stage I
T1 N0 M0
Stage II
T2 N0 M0
Stage III
T1 N1 M0
T2 N1 M0
T3 N0 M0
T3 N1 M0
Stage IVA
T1 N2 M0
T2 N2 M0
T3 N2 M0
T4a N0 M0
T4a N1 M0
T4a N2 M0
Stage IVB
Any T N3 M0
T4b any N M0
Stage IVC
Any T any N M1
Further Reading
American Cancer Society. Cancer Facts and Figures 2008. Atlanta, GA: American Cancer Society, 2008
American Joint Committee on Cancer. AJCC Cancer Staging Handbook. 7th ed. New York: Springer-Verlag; 2010
Donald PJ. Cancer of the oropharynx and tongue base. In: Donald PJ, ed. The Difficult Case in Head and Neck Cancer Surgery. Stuttgart/New York: Thieme; 2010: 43–71
Gillison ML, Koch WM, Capone RB, et al. Evidence for a causal association between human papillomavirus and a subset of head and neck cancers. J Natl Cancer Inst 2000;92(9):709–720
Goldenberg D, Golz A, Joachims HZ. The beverage maté: a risk factor for cancer of the head and neck. Head Neck 2003;25(7):595–601
Odell MJ, Walz BJ, Reimers HJ, Varvares MA. Carcinoma of the oropharynx. In: Genden EM, Varvares MA, eds. Head and Neck Cancer: An Evidence-Based Team Approach. Stuttgart/New York: Thieme; 2008:24–43
Weber AL, Romo L, Hashmi S. Malignant tumors of the oral cavity and oropharynx: clinical, pathologic, and radiologic evaluation. Neuroimaging Clin North Am 2003;13(3):443–464
Wong RJ, Lin DT, Schöder H, et al. Diagnostic and prognostic value of [(18) F]fluorodeoxyglucose positron emission tomography for recurrent head and neck squamous cell carcinoma. J Clin Oncol 2002;20(20):4199–4208
5.2.7 Hypopharyngeal Cancer
Key Features
• Hypopharyngeal cancer occurs in the pyriform sinuses, the posterior hypopharyngeal, or postcricoid region.
• Patients present with dysphagia, globus sensation, hoarseness, and referred otalgia.
• It usually presents in advanced stages of disease.
• It has a high propensity for regional metastasis at presentation.
The hypopharynx is the part of the pharynx, which lies behind the larynx and connects the oropharynx and the esophagus. It is subdivided into three subsites: the paired pyriform sinuses, the posterior hypopharyngeal wall, and the postcricoid region. Sixty-five to 85% of cancers of the hypopharynx involve the pyriform sinuses, 10 to 20% involve the posterior pharyngeal wall, and 5 to 15% involve the postcricoid area.
Epidemiology
Cancer of the hypopharynx is uncommon, with ~2500 new cases diagnosed in the United States each year. Cancer of the hypopharynx typically presents in advanced stages, and the incidence of regional metastases and distant metastases are also among the highest of all head and neck cancers. Cancer of the hypopharynx is typically seen in men over 55 years old with a history of tobacco product use and/or alcohol ingestion.
An exception is an increased incidence of postcricoid cancer in women aged 30 to 50 years with Plummer-Vinson syndrome. Asbestos may pose an independent risk for the development of cancer of the hypopharynx.
Clinical
Signs and Symptoms
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


