FIGURE 38.1 Eye of infant with retrolental fibrovascular membrane from stage 5 ROP. Total retinal detachment, determined by ultrasonogram, exists posterior to the white retrolental membrane.
However, ROP continues to cause significant visual morbidity with increased survival of young gestational age and very low birth weight premature infants and with emerging neonatal care throughout the world. We now recognize that multiple factors are involved in the development of ROP, and the role of supplemental oxygen appears more complex. In addition, the recognition of earlier stages of ROP has both facilitated investigation into the causes of ROP and led to earlier treatment to reduce the risk of retinal detachment and blindness (Tables 38.1 to 38.3).
TABLE 38.1
Major clinical trials in retinopathy of prematurity


a240 had bilateral disease and were treated with cryotherapy (cryo) in one eye and observed in the fellow eye. In 51 asymmetric eyes, treatment was randomly assigned to either cryo or observation.
bFor the 1-year and 3½-year studies, Teller acuity vision was determined. For the 5½-, 10-, and 15-year outcome studies, testing using the ETDRS visual acuity chart was also performed.
cOf the 401 infants randomized in the high-risk prethreshold group, 317 had bilateral disease and were treated with either early ablative therapy within 48 hours of the first diagnosis of high-risk prethreshold or conventionally. In the remaining 88 asymmetric cases, the eye was randomized to either early treatment or CM.
CRYO-ROP, Cryotherapy for Retinopathy of Prematurity; Light-ROP, Light Reduction on Retinopathy of Prematurity; BW, birth weight; ROP, retinopathy of prematurity; MO, month; GA, gestational age; STOP-ROP, Supplemental Therapeutic Oxygen for Prethreshold Retinopathy of Prematurity; NICU, neonatal intensive care unit; RA, room air; Con, conventional oxygen; S, supplemental oxygen; PMA, postmenstrual age; HOPE-ROP, High-Oxygen Percentage Retinopathy of Prematurity; ETROP, Early Treatment for Retinopathy of Prematurity;BEAT-ROP, Bevacizumab Eliminates the Angiogenic Threat of Retinopathy of Prematurity; CM, conventional management; BCVA, best-corrected visual acuity; ETDRS, Early Treatment Diabetic Retinopathy Study visual acuity; PUFAs, polyunsaturated fatty acids.
TABLE 38.2
Later studies performed on original CRYO-ROP cohort

RD, retinal detachment; ROP, retinopathy of prematurity; CRYO-ROP, Cryotherapy for Retinopathy of Prematurity; Light-ROP, Light Reduction on Retinopathy of Prematurity; ETROP, Early Treatment for Retinopathy of Prematurity; OU, both eyes; PMA, postmenstrual age (gestational age plus chronological age in weeks).
TABLE 38.3
Later studies performed on ETROP-ROP cohort

ETROP, Early Treatment for Retinopathy of Prematurity; ROP, retinopathy of prematurity; ET, early treatment; CM, conventional management; OU, both eyes; RD, retinal detachment.
International Classification of Retinopathy of Prematurity Characterizes Progression of Retinopathy of Prematurity
The International Classification of Retinopathy of Prematurity was developed to describe the levels of severity of ROP based on several parameters: zone, stage, extent of stage, and presence of plus disease (47). This classification was updated in 2005, to reflect changes in the understanding of the disease since the initial publication in 1984 (48). The zone of ROP refers to one of three areas that best describes the retinal area that has apparent intraretinal vascularization (Fig. 38.2). Normally, in development, vascularization of the retina starts posteriorly with early vascularization around the optic nerve disputed to be a result of angioblast growth or vasculogenesis. From these early vessels, further proliferation and migration extends vascularization toward the ora serrata (see Chapters 1 and 5).
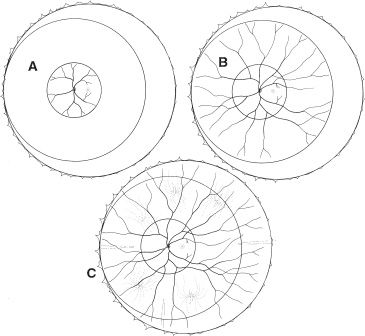
FIGURE 38.2 Circular zones I (A), II (B), and III (C) drawn onto schematic of fundus. Zone 1 is the least mature and encompasses the smallest area of retina with developed retinal vasculature.
Zone I is the retinal area encompassed by a circle centered around the optic nerve with a radius equivalent to two times the distance from the optic nerve to the fovea. Zone II is the area outside zone I but within a circular area centered on the optic nerve with a radius equivalent to the distance from the optic nerve to the nasal horizontal ora serrata. Zone III is the remaining temporal crescent. Where there is no retinal vascularization, an avascular area is clinically apparent peripheral to vascularized retina. The stage of ROP defines the clinical appearance of the retina at the junction of vascularized retina and the avascular area. There are five stages. In stage 1, a line is apparent (Fig. 38.3). In stage 2, there is a ridge having obvious volume (Fig. 38.4). In stage 3 ROP, there is neovascularization growing onto the vitreous at the ridge (Fig. 38.5). Stage 4 ROP is partial retinal detachment, 4A without macular involvement and 4B with macular involvement (Fig. 38.6). Stage 5 ROP is a total retinal detachment and is described as closed where the retina is adherent to itself or open where it is not. In some cases of stage 5 ROP, there is a peripheral area of attached avascular retina causing a peripheral retinal trough. The extent of ROP refers to the number of clock hours of the highest stage. Plus disease refers to dilation and tortuosity of the retinal arterioles and veins in the posterior pole (Fig. 38.7) and is based on a standard photograph published in the multicenter trial of CRYO-ROP (8). In later clinical trials, the definition of plus disease was modified so that the diagnosis of plus disease could be made if sufficient vascular dilation and tortuosity were present in at least two quadrants (48). Pre–plus disease was defined as abnormal dilation and tortuosity of the posterior pole vessels, which is insufficient for the diagnosis of plus disease based on the standard photograph but is more so than normal (48). Aggressive posterior ROP (AP-ROP) was defined as aggressive posterior retinopathy observed commonly in zone I or posterior zone II, with dilation and tortuosity of both arteries and veins in all four quadrants that is out of proportion to the peripheral retinopathy. AP-ROP does not usually progress through the classic stages, extends circumferentially, and can be accompanied by a circumferential vessel. Though there may only be a flat neovascularization at the junction of vascularized and nonvascularized retina, this aggressive form of ROP usually progressed to stage 5 if it is not promptly treated (48).
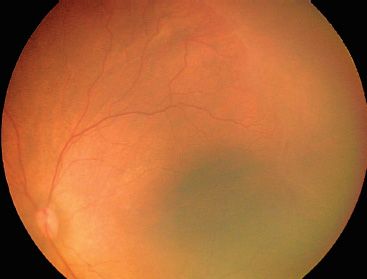
FIGURE 38.3 Stage 1 ROP seen as a discontinuous white, tortuous line between vascularized (posterior) and avascular (peripheral) retina temporally.
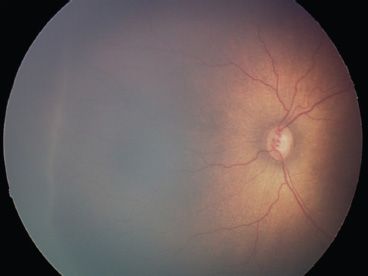
FIGURE 38.4 Stage 2 ROP with a prominent ridge at the junction of the vascular and avascular retina.
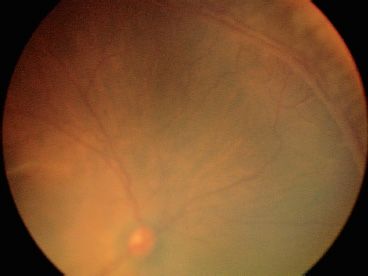
FIGURE 38.5 Image of eye treated with laser to avascular retina (upper right of field). Note stage 3 neovascularization extending into vitreous. Some blood is seen within the neovascular tissue.
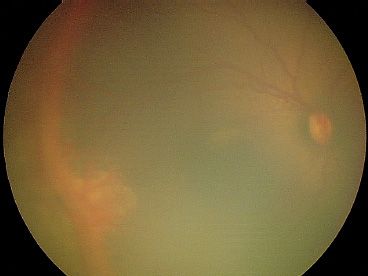
FIGURE 38.6 Early stage 4A ROP with exudation posterior to the ridge. The elevated retina extending superiorly and inferiorly from area of exudation at 9 o’clock appears out of focus.
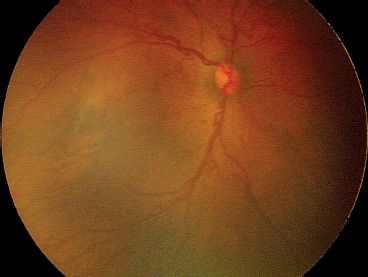
FIGURE 38.7 Moderate plus disease present posteriorly as dilated, tortuous vessels around the optic nerve.
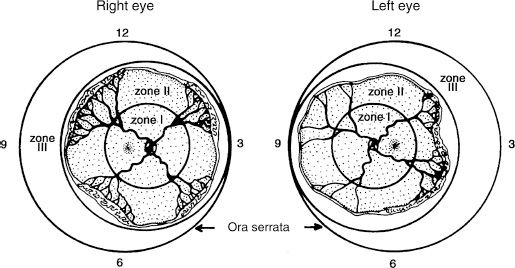
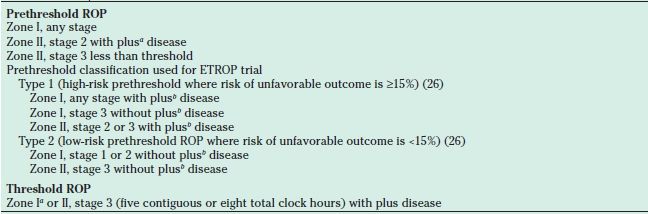
FIGURE 38.8 Artist rendition of threshold disease defined in the multicenter trial of CRYO-ROP study as five contiguous or eight total clock hours of stage 3+ in zone I or II.
Age of Onset
Most infants that develop some form of ROP do so at about 32 weeks’ postmenstrual age (PMA). PMA equals gestational age plus age after birth in weeks (36,49). Threshold disease, at which the risk of a poor outcome (Table 38.4) approached 50% (see later), peaks at approximately 37 weeks’ PMA (16,22) and high-risk prethreshold ROP (see “Prethreshold and Threshold Retinopathy of Prematurity”), where risk of a poor outcome is ≥15%, at approximately 35 weeks (26). Eyes that rapidly progress to prethreshold ROP have a greater risk of developing threshold ROP (16,20) and will develop threshold disease at an earlier PMA. When infants achieve 45 weeks’ PMA without developing prethreshold ROP, they have a low risk of developing threshold ROP or of having a poor outcome (36).
TABLE 38.4
Unfavorable retinal outcomes

ROP, retinopathy of prematurity.
Course of Retinopathy of Prematurity
Most ROP regresses; however, the disease typically progresses before regressing and this progression can be rapid—in the ETROP study, in infants born weighing <1,251 g, 22% of infants with type 2 prethreshold ROP progressed to type 1 prethreshold and half of these in <7 days (40). In about 6% of infants born weighing <1,251 g, threshold ROP develops (9). Without treatment, this results in an unfavorable outcome in about 52% of eyes at 15-year follow-up (10). In the CRYO-ROP trial, approximately 30% of eyes experienced an unfavorable anatomic outcome even after treatment for threshold ROP (10). Unfavorable visual outcomes were 45% for treated eyes and 64% for untreated eyes, respectively (10). However, based on outcome data from the ETROP trial, treatment at a less severe level of ROP, that is, for type 1 prethreshold ROP (type 1 ROP), reduces poor visual and functional outcomes compared to waiting until threshold ROP develops (50).
Prethreshold and Threshold Retinopathy of Prematurity
Based on the CRYO-ROP study, threshold ROP is defined as the level of severity of ROP at which the risk of an unfavorable anatomic outcome approached 50% (8,9) was diagnosed by the presence of five contiguous or eight total clock hours of stage 3 ROP in zone I or II with plus disease (Fig. 38.8). Because a high frequency (87%) of zone I eyes had an unfavorable outcome in the CRYO-ROP study, later studies such as Supplemental Therapeutic Oxygen to Prevent Prethreshold Retinopathy of Prematurity (STOP-ROP) (22) and High-Oxygen-Percentage Retinopathy of Prematurity (HOPE-ROP) (23) defined threshold ROP less stringently for zone I eyes (Table 38.5). Prethreshold ROP defined eyes at high risk of developing threshold ROP and that should be monitored closely. In the ETROP trial, prethreshold ROP was further subdivided based on the risk of an unfavorable outcome, as assessed by a risk analysis program based on natural history data from the CRYO-ROP study, RM-ROP2. Eyes with high risk were defined as having ≥15% risk, whereas eyes at low risk had <15% risk of an unfavorable outcome. An unfavorable outcome was primarily based on one of four categories of visual function as tested with Teller Acuity Cards (26). Secondary outcomes were based on structure where poor outcomes were a retinal fold or detachment involving the macula, or a retrolental opacity blocking the visual axis (Table 38.4). Type 1 ROP was defined as zone I, any stage ROP with plus disease; zone I, stage 3 ROP without plus disease; and zone II, stage 2 or 3 with at least two quadrants of plus disease based on the standard photograph from the CRYO-ROP study (see Table 38.5) (50). Treatment of the peripheral retina with laser for type 1 ROP was found to decrease unfavorable structural outcomes from 15.6% to 9.0% at 9 months (26,50) and from 15.2% to 8.9% at 6-year follow-up (38,39). Though a statistically significant benefit in Teller grated acuity was seen at 9 months in the early treatment group compared to CM, the difference was not statistically significant at 6 years (39). However, type 1 eyes receiving early treatment had a significantly lower rate of unfavorable outcomes when compared to conventional treatment (39). Based on the ETROP results, treatment for type 2 ROP (defined as zone I, stages 1 and 2 without plus disease, or zone II, stage 3 without plus disease) was recommended only if it progressed to type 1 ROP or threshold (50).
TABLE 38.5
Definitions of prethreshold and threshold ROP
aThe definition of threshold ROP was different for the CRYO-ROP and STOP-ROP studies: (i) For STOP-ROP, plus disease was defined as two quadrants of posterior pole dilation/tortuosity, whereas in CRYO-ROP it was defined as about four quadrants of posterior pole dilation/tortuosity, and (ii) for STOP-ROP, a less stringent definition of threshold disease was given for zone I eyes that included any ROP with plus disease or any stage 3 with or without plus disease.
bBased on standard photograph used in CRYO-ROP studies; requires two quadrants of plus disease (similar to criteria used in STOP-ROP study).
ROP, retinopathy of prematurity; ETROP, Early Treatment for Retinopathy of Prematurity.
These definitions provide guidelines for managing infants with acute ROP.
Diagnosis
An accurate diagnosis of ROP is essential to detect eyes that require urgent treatment for type 1 ROP and, in eyes that do not meet the criteria for treatment, to determine when the follow-up examination should be performed. To obtain an accurate diagnosis, a complete retinal examination must be performed in a well-dilated infant using an indirect ophthalmoscope and, often, scleral depression. Once the infant’s eyes are fully dilated and the infant is swaddled, a drop of topical anesthetic is instilled into one eye and a lid speculum is inserted. Initially no scleral depression is performed so as to reduce possible blanching of plus disease. The retinal vessels are assessed for vascular dilation and tortuosity in each of the four quadrants, first, in the vascular arcades around the optic nerve (Fig. 38.7) and, second, in the periphery (Fig. 38.9). Scleral depression can be useful to determine the zone of intraretinal vascularization. Determination of zone I is made either by estimating the circle surrounding the optic nerve having a radius of twice the distance from the optic nerve to the fovea or by viewing the image of the fundus through a 28-diopter lens with nasal edge of the optic nerve at one edge of the field of view; the limit of zone I is at the temporal field of view (48). If the retinal vasculature does not extend beyond the image size of the 28-diopter lens, then vessels are likely within zone I. Zone II is retinal vascularization beyond zone I but not within one disc diameter of the nasal ora serrata, for example, 9 o’clock OS or 3 o’clock OD (48). Zone III is the remaining avascular temporal area of retina (Fig. 38.2).
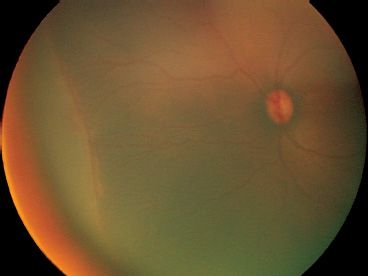
FIGURE 38.9 Peripheral plus disease noted by dilated and tortuous retinal arterioles and veins in the peripheral retina extending into the ridge.
There can be difficulties examining the lightly pigmented, blonde fundus, or deeply pigmented fundus. In the blonde fundus (Fig. 38.10), the choroidal vessels are apparent, and the examiner may mistake these for retinal vessels. In the deeply pigmented fundus (Fig. 38.11), it can be difficult to visualize the retinal vessels against the dark, pigmented background. In both of these cases, the 20-diopter lens can improve diagnostic ability.
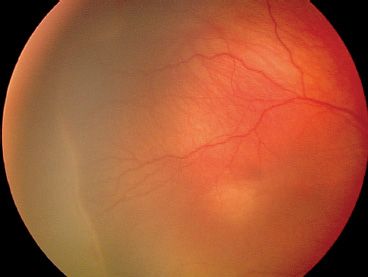
FIGURE 38.10 Blonde fundus permits visualization of deeper choroidal vessels that can be mistaken for retinal blood vessels.
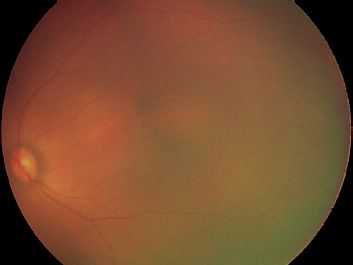
FIGURE 38.11 Deeply pigmented fundus reduces the contrast between melanin of retinal pigment epithelium and choroid and hemoglobin of retinal vessels, making it difficult to visualize retinal vessels.
Classically, at the junction of vascularized and avascular retina, the examiner determines the stage of ROP and the clock-hour extent of each stage. The clock-hour extent of the highest stage, the zone, and the presence or absence of plus disease are recorded. The clock-hour extent may be less important in determining eyes at risk of an unfavorable outcome that should have laser treatment (11,26) but still is important in determining eyes at risk of progressive stage 4 ROP after laser treatment (51).
The same procedure is performed in the fellow eye.
Differential Diagnosis
The differential diagnosis of ROP includes familial exudative vitreoretinopathy, Norrie disease, incontinentia pigmenti, congenital retinal fold, Toxocara canis infection, and causes of leukocoria (Table 38.6). A white pupil, referred to as leukocoria, can occur from media opacities at any level from the cornea to the retina. The history of prematurity and low birth weight are the most helpful historical data in the differential diagnosis of ROP.
TABLE 38.6
Differential diagnosis of leukocoria
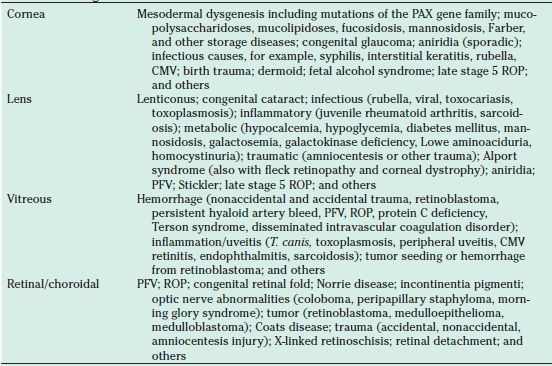
CMV, cytomegalovirus; PFV, persistent fetal vasculature; ROP, retinopathy of prematurity
Clinical Symptoms and Signs
There are no symptoms of acute ROP, nor can a specific visual behavior in the preterm newborn herald a concern for ROP. Therefore, effective screening is essential for diagnosis.
Screening and Management
The screening protocol at each neonatal intensive care unit (NICU) should be based on published recommendations and preferences of the screening ophthalmologists, neonatologists, and NICU nurses. All at-risk infants should be identified and receive adequate dilated retinal evaluations at appropriate times. The Joint Statement of the American Academy of Pediatrics, the American Association for Pediatric Ophthalmology and Strabismus, and the American Academy of Ophthalmology (published in 2013) provided the following guidelines for infants who should be screened for ROP: (i) all infants born weighing ≤1,500 g and/or ≤30 weeks’ gestational age and (ii) infants born weighing >1,500 g or more than 30 weeks but who experience an unstable clinical course at the discretion of the neonatologist or pediatrician. The first examination should be performed prior to hospital discharge, at 4 weeks after birth, or at 31 weeks’ PMA depending on the gestational age (49).
Examinations should be repeated periodically, for example, every 2 weeks if there is no ROP, weekly with any ROP, and more frequently with prethreshold ROP. If intraretinal vascularization proceeds toward the ora serrata, examinations are performed less frequently. Signs indicating that the risk of visual loss from ROP is minimal include PMA of 45 weeks, intraretinal vascularization into zone III without previous zone II ROP, and complete intraretinal vascularization determined on two consecutive examinations (49). When any of these signs is present, screening can be discontinued and the focus of care changed to that of visual rehabilitation.
If type 1 disease develops, treatment should be performed within 48 hours (52) based on the ETROP protocol. Treated eyes are followed closely, often weekly or more frequently, for signs of regression, need for further laser, or possible vitreoretinal surgery if progressive stage 4 ROP develops.
The Bevacizumab Eliminates the Angiogenic Threat of Retinopathy of Prematurity (BEAT-ROP) study demonstrated a reduction in the rate of recurrence of neovascularization in infants with stage 3 zone 1 ROP treated with intravitreal bevacizumab (0.625 mg in 0.025 mL) followed to 54 weeks’ PMA but not for zone II disease when compared to laser therapy (28). Interestingly, the interval for disease recurrence was 19.2 ± 8.8 weeks with bevacizumab compared to 6.4 ± 6.7 weeks with laser for zone 1 disease, introducing the need for longer-term follow-up to assure no disease recurrence. In addition, this study was not sufficiently powered to evaluate the systemic safety of bevacizumab. Though this is a promising new avenue of treatment, a multicenter randomized clinical with long-term follow-up is necessary to further investigate the role of bevacizumab in ROP.
Prevention
ROP is a disease of premature infants. Therefore, reducing the number of premature births, such as through good prenatal care (53), reduction in teenage pregnancies (54), and avoidance of illegal drug use (55), would reduce the number of infants at risk for ROP.
To reduce the incidence of ROP, in the 1960s, NICUs began to monitor oxygen delivery to the preterm newborn, particularly in the perinatal period so as to avoid high oxygen levels, such as those used when RLF was first recognized (6). NICUs now have the technology to maintain oxygen within predetermined “safe” limits, defined by the Fetus and Newborn Committee of the American Academy of Pediatrics and American College of Obstetricians and Gynecologists (56). However, several studies provide evidence that not only the concentration but also the variability of oxygen plays a role in the development and severity of ROP (57,58) (see “Effects of Environmental Factors,” and “Oxygen”).
To prevent blindness from ROP in the preterm infant, screening is important to detect type 1 ROP. Beginning with the CRYO-ROP study (9), it has been shown that laser treatment or cryotherapy (Table 38.7) delivered to the avascular zones of eyes with threshold ROP within 72 hours reduced unfavorable outcomes and vision loss (9,12,22). The ETROP trial refined the definition of eyes at risk of threshold ROP and an unwanted outcome: all zone I eyes with any ROP and plus disease, zone I eyes with stage 3 ROP without plus disease, and zone II eyes with stages 2 or 3 ROP and plus disease (Table 38.1) (2,26,59). If treatment is adequate but ROP progresses to stage 4 ROP with certain retinal features (11) or in eyes with stage 5 ROP, vitreoretinal surgery is considered. Finally, visual rehabilitation is important to treat amblyopia and myopia that occur more commonly in prematurely born children and even more often in those who had ROP, compared to infants born full term (Tables 38.2 and 38.3) (31,60). Segmentation of encircling elements used to treat stage 4 ROP or rhegmatogenous retinal detachment reduces optical irregularities, permits growth of the globe, and may improve visual outcomes (61,62).
TABLE 38.7
Laser compared to cryotherapy for treatment of threshold ROP
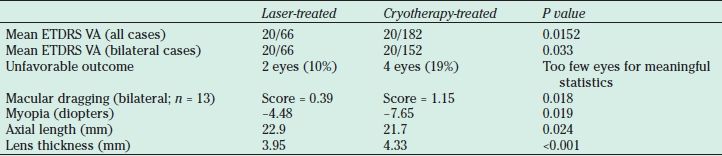
Laser-treated eyes 5.2 times more likely to have 20/50 visual acuity (VA). Linear regression analysis revealed poor correlation (r = 0.36, P = 0.14, n = 18) in best-corrected visual acuity (BCVA) between eyes of the same infant with bilateral disease and had one eye treated with laser and the fellow eye with cryotherapy. For laser-treated eyes that lacked retinal dragging, BCVA was predicted to be 20/32, but for cryotherapy-treated eyes (58), BCVA was predicted to be 20/50.
The thickness of the lens was most strongly correlated to refractive outcomes in both laser-treated (r = 0.885, P < 0.001) and cryotherapy-treated eyes (r = 0.591, P = 0.026) (63).
ETDRS, Early Treatment Diabetic Retinopathy Study visual acuity testing standards; VA, visual acuity.
Vision Rehabilitation
Vision rehabilitation is important in all premature infants with or without ROP. Premature infants are more likely to require strabismus surgery, to be treated for amblyopia, and to be myopic than full-term infants (Tables 38.2 and 38.3). These conditions are even more common in premature infants who had more severe levels of ROP and threshold disease than those with milder ROP (31,45,59,64). Infants with macular heterotopia may have potential for visual acuity (65); therefore, correction of refractive errors and treatment of amblyopia are indicated. The ETROP also showed that patients without obvious macular heterotopia had moderate visual loss and causes for this are still being studied (66). For the aphakic infant who had retinal detachment repair, visual rehabilitation with special adjustments for low vision is recommended. Even when grating acuity cannot be measured, low vision can be stratified into levels characterized by the presence of light perception in different fields of gaze (66) and may be important to infant and child development. Treatment of aphakic amblyopia and aphakia is controversial, particularly in the infant after successful retinal reattachment of stage 5 ROP. Macular vision is not likely to develop, but aphakic spectacles are believed by vision teachers to improve the peripheral vision of the child (Perkins School of the Blind, Watertown, MA). In the child with macular acuity in one eye and aphakic amblyopia in the fellow eye, spectacle wear also provides protection against ocular trauma. We recommend that infants be managed by a pediatric ophthalmologist for visual rehabilitation starting at an earlier age than that recommended for full-term infants and that children be enrolled in an early intervention program that enhances the use of all of the senses.
Role of Other Physicians and Health Care Providers
A team approach and effective communication are important in the care of the premature infant. During the time that the infant is in the NICU, the core caregivers include the neonatologist, the screening ophthalmologist, and the nursing staff. Often a nurse manager or ophthalmic technician can facilitate communication between the ophthalmologist and the NICU to assure that eye drops are given at the appropriate time, consultation reports are available for the examining ophthalmologist to fill out, and the individual infant’s nurse is available to assist and monitor for apnea and bradycardia during the examination. We prefer to have a nurse, fellow physician, or trained technician assist by securely holding the swaddled infant during the examination. It is important to provide a written consultation for all infants and to communicate with the neonatologist, especially about infants with progressing ROP. Ongoing communication with the parents helps to avoid any possible surprise if treatment is urgently needed.
Once the infant is discharged from the NICU, follow-up examinations for the retina and later for visual rehabilitation are important. Often, a written information booklet is given to parents or guardians with emphasis placed on the importance of the timing of appointments. A system is required to reschedule infants as soon as possible if appointments are missed and requires training of the office staff to assure infants needing urgent appointments are examined. When repeated appointments are missed, we send a certified letter to the parents or guardians. Once the risk of ROP is reduced, follow-up with a pediatric ophthalmologist and in an early intervention program is important for visual rehabilitation.
Ethical Considerations
Ethical questions arise when working in neonatal intensive care. Each question is taken on an individual basis, accounting for the infant’s general condition and fellow eye, and is addressed by a team that may include the neonatologist, parents, and ophthalmologists. For example, it is universally agreed that stage 5 ROP should be prevented, and efforts are made to accomplish this. Careful monitoring of eyes after treatment for ROP is essential to diagnose progressive stage 4 ROP that might be repaired with a lens-sparing vitrectomy (67,68). However, cases of stage 5 ROP can occur when an infant is too sick to undergo treatment, when treatment does not prevent retinal detachment, or when infants are lost to follow-up despite efforts to prevent this. The question may arise as to whether heroic surgery should be performed to reattach the retina in an eye with stage 5 ROP in a preterm infant with multiple medical conditions. Evidence exists that low vision can be stratified in infants with repaired retinal detachments from stage 5 ROP (69) and can be useful in children when at a level believed to have little value in adults (70). Still, long-term benefits of low vision on child development and quality of life are difficult to study and remain largely unknown.
Worldwide Impact on Child Blindness, Including Public Health Issues
ROP is a growing cause of blindness, especially in middle-income developing countries where neonatal care is now emerging.
Risk Factors
Severe Retinopathy of Prematurity
The greatest single risk factor for developing ROP is being born prematurely (71–84). However, we now know much more about the risks of progression of the disease. The CRYO-ROP study provided data from the natural history cohort (Table 38.1) on ocular and systemic characteristics associated with increased risk of ROP, progression to threshold ROP, and development of an unfavorable macular outcome (Table 38.4) (16). An important ocular risk factor was zone I. If, at 32 weeks’ PMA, incomplete vascularization in zone I was present, 32.8% of eyes developed threshold ROP, compared to 9.3% with incomplete vascularization in zone II. Eyes with zone I ROP also were at high risk of developing threshold ROP. Other ocular risk factors for developing threshold ROP included plus disease, stage 3 ROP (16), ≥6 clock-hour stage 3 (18), and iris vessel dilation (19). Nonocular risk factors associated with the development of threshold ROP included young gestational age, multiple births, out-of-nursery birth, low birth weight (16), and Caucasian race (Table 38.1) (16,17). Once prethreshold ROP developed, the risk of developing threshold ROP was about the same given these factors. For each 100-g increase in birth weight, there was a 27% decrease in the percentage of infants who developed threshold ROP. Black race was associated with a 65% lower chance of developing threshold ROP compared to Caucasian race if any ROP was present and a 51% decreased chance of developing threshold if prethreshold ROP developed. Once threshold ROP developed, the risk of an unfavorable anatomic outcome (Table 38.4) was about the same in blacks as for Caucasians (16). Factors associated with increased risk of prethreshold progressing to threshold ROP included lower PMA at the diagnosis of any ROP, ROP in zone I at the first screening examination, rapid progression of ROP to prethreshold characteristics, plus disease at the first prethreshold examination, and white race (16,20).
In a recent study performed in the United Kingdom, the odds of severe ROP requiring treatment was higher in Asian infants (odds ratio 2.52) and black infants (odds ratio 2.51). This additional risk from ethnicity was present even after adjusting for birth weight and gestational age (74). There have been few other studies of race and risk of ROP in premature infants. These results were consistent with an earlier study also from the United Kingdom demonstrating that Asians had a greater risk of developing ROP than Caucasians and that this perceived risk was related to an increased survival of Asians (74). Another small study reported that native Alaskans were more susceptible to ROP compared to nonnative Alaskans (85). Although race cannot be addressed using animal models, different strains of rats were shown to develop different degrees of severity of retinopathy under the same conditions (86).
Postnatal clinical factors may also influence the risk of developing ROP in complex ways. The weight, insulin-like growth factor [IGF], neonatal ROP (WINROP) algorithm was developed to detect early indications of ROP on the basis of postnatal weight gain, IFG-I, and IGF-binding protein in the serum (87). Subsequently, postnatal weight gain alone was shown to detect a slowdown in growth that correlated with an increased risk of ROP requiring treatment in infants born at gestational ages of <32 weeks (88). The WINROP algorithm detected 100% of infants who developed ROP requiring treatment and
correctly predicted the majority of those that did not, in a study of 353 infants (88).
Recent research suggests that in addition to environmental factors, genetics may play a role in the development of severe ROP. However, none of the genetic factors identified thus far account for a significant portion of the patient population, and additional study in this area may bring exciting changes to our understanding of ROP (89).
Unfavorable Macular Outcome
In the initial natural history report, ocular characteristics associated with an unfavorable macular outcome included zone I ROP, plus disease, and stage 3 ROP (Table 38.1). Eyes with zone I ROP had an odds risk of 8.24 toward developing an unfavorable outcome compared to zone II ROP (16). For each clock hour of stage 3 ROP greater than 5 clock hours, there was a 26% increased risk of an unfavorable macular outcome (16). Stage 3 ROP with plus disease in zone II was associated with a 62% risk of an unfavorable result compared to stage 3 ROP without plus disease in zone II in which the risk was 3%. In the 5.5-year report from the natural history cohort of CRYO-ROP (18), an unfavorable anatomic outcome occurred in 62.5% of zone I eyes compared to 44.2% of zone II eyes. All unfavorable anatomic outcomes in this cohort from CRYO-ROP occurred in eyes with 6 or more clock hours of stage 3 ROP with plus disease in zone I or II (18).
In the ETROP study, at 6-year follow-up, the difference in unfavorable outcomes for all high-risk pre-threshold eyes was 8.9% for early treated eyes versus 15.2% for conventionally managed eyes (P < 0.001). The greatest benefit of early treatment was seen with zone I, stage 3 disease, with or without plus (unfavorable outcomes were doubled for these eyes in the conventionally treated group) and zone II, stage 3 disease with plus (38).
Predicting Progressive Stage 4 Retinopathy of Prematurity Requiring Surgery
Few studies have provided information regarding risk factors associated with progressive stage 4 ROP requiring surgical intervention. Clinical trials have not been funded to pursue this question. To address this question, we studied features of 72 eyes of 36 infants who had been treated with laser for threshold ROP. Features were abstracted from examinations made 1 week prior to the development of stage 4 ROP that was found to progress over time or 2 weeks after laser in eyes that had regression of threshold ROP. Eyes were stratified into two groups based on whether progressive stage 4 ROP or regressed stage 3 ROP occurred. A generalized estimating equation model was used to account for within-subject variability and determine predictive features of progressive stage 4 ROP. Features assessed included clock hours of ridge elevation, quadrants of plus disease, quadrants of neovascularization, and vitreous state. Ridge elevation was defined as white thickened tissue at the junction of vascularized and avascular retina. Absence of ridge meant that no or only a line denoted the junction. Plus disease was defined as dilated and tortuous retinal arterioles and veins. The presence of only dilated veins in a quadrant was counted as a half quadrant. The vitreous state was defined as hazy, with hemorrhage, or vitreous condensation permitting visualization of the regressed primary vitreous structures. The following features predicted progressive stage 4 ROP: ≥6 clock hours of ridge elevation (P = 0.0248), ≥2 quadrants of plus disease (P = 0.0490), and vitreous haze (P = 0.0039) (51). Neovascularization did not predict progressive stage 4 ROP; however, in a separate analysis, it was associated with a poor surgical outcome (90).
Effects of Environmental Factors and Nonocular Treatments on Retinopathy of Prematurity
Oxygen
The relationship of oxygen to the development of ROP is complex and incompletely understood. Metabolic and oxygen needs increase during the development of the retinal vasculature and the maturation of the neural retina and photoreceptors (91). Early retinal vessels are also sensitive to fluctuations in outside oxygen delivery. A number of factors also affect oxygen delivery to the retinal vasculature and neural retina in the premature infant, including poor blood oxygenation secondary to immature lungs and respiratory disease, anemia of prematurity, and changes in the ratio of fetal to adult hemoglobin affecting oxygen affinity to hemoglobin.
During the 1950s when ROP was initially described, high, unregulated oxygen at the time of birth was found to be a significant factor that led to severe ROP (92). Efforts then to reduce oxygen concentration led to reduced blindness but also increased pulmonary and cerebral morbidity. Although today’s NICUs provide oxygen in a controlled fashion at lower inspired O2 than when ROP was first recognized, ROP continues to cause blindness. However, high oxygen, proposed as an important risk factor for ROP in the past (7,92,93), now is no longer considered an independent risk factor for ROP (94) because oxygen levels are lower and better controlled. Research is ongoing regarding the interplay of hyperoxemia, hypercapnia, and acidemia in the development and severity of ROP (95).
Because of ethical considerations—inability to obtain retinal tissue and to adequately experiment in the human infant—animal models have been created by varying oxygen concentrations to induce retinal avascularity followed by intravitreous neovascularization. Early models of oxygen-induced retinopathy (OIR) and some of the current models exposed animals to constant high oxygen followed by room air to create avascular areas of retina first and, to a variable degree, subsequent intravitreous neovascularization (96–98). These models showed that high concentrations of oxygen led first to large areas of avascular retina, with subsequent development of intravitreous neovascularization, when animals were placed into room air (96). These models were useful in understanding the events of acute ROP in the 1950s but do not describe the oxygen delivery currently used in neonatal care.
STOP-ROP was a large multicenter trial, designed to test supplemental oxygen as a strategy to prevent threshold ROP (Table 38.8), did not show additional progression of prethreshold ROP at high pulse oximetry values of 96% to 99%, but also did not decrease the need for peripheral retinal ablative procedures (22). In a post hoc analysis, the study showed reduced progression to threshold ROP in a subgroup of infants treated with supplemental oxygen (to achieve SaO2 of 96% to 99%) who had prethreshold ROP without plus disease.
TABLE 38.8
Results from STOP-ROP study
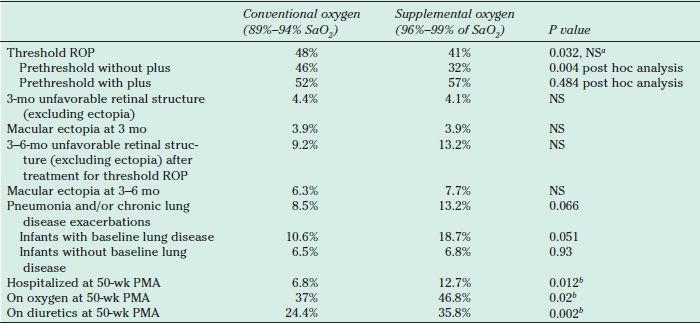
aThe target alpha value was 0.025, deliberately conservative to allow for preplanned sequential testing. To balance the benefits and risks of supplemental oxygen, a number-needed-to-treat analysis was performed. This analysis compares the number of infants with prethreshold ROP who would have to be treated with supplemental oxygen to spare an infant from retinal surgery to the number of infants treated with supplemental oxygen to be expected to cause one episode of pulmonary exacerbation. The number requiring supplemental oxygen to prevent an adverse retinal event was 13.2 and to cause a pulmonary exacerbation was 13.7. Pulmonary events were not lethal, although they did prolong hospitalizations (22,99).
bThe proportion of infants who experienced any one or more of these adverse pulmonary events by 3 months’ corrected age was 57% (supplemental) vs. 46% (conventional), P = 0.005, and was unaffected after adjusting for baseline covariates of race, ROP severity, gestational age, and pulmonary status.
STOP-ROP, Supplemental Therapeutic Oxygen for Prethreshold Retinopathy of Prematurity; ROP, Retinopathy of Prematurity. NS, not significant; PMA, postmenstrual age (gestational age plus chronologic age in weeks).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


