Purpose
To compare the results of 23- and 20-gauge vitrectomies combined with phacoemulsification and aspiration and intraocular lens implantation (phacoemulsification surgery) 1 year after repair of idiopathic macular holes.
Design
Retrospective, consecutive, comparative case series.
Methods
The medical charts of 100 consecutive eyes were reviewed that had undergone either 23- or 20-gauge vitrectomy combined with phacoemulsification surgery to treat an idiopathic macular hole performed by 1 surgeon. The rate of improvement of the logarithm of the minimal angle of resolution visual acuity (VA) was calculated using the formula: (postoperative value − preoperative value) × 100/(1-year postoperative value − preoperative value).
Results
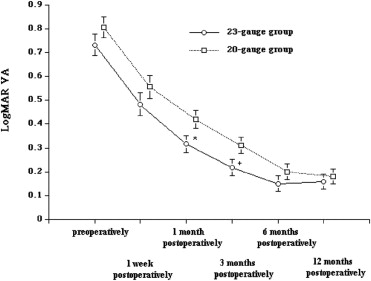
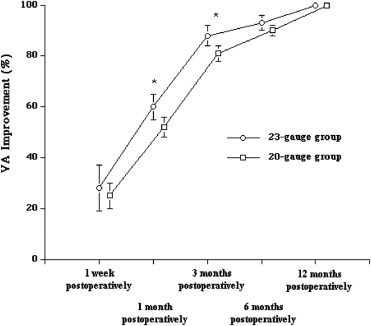
The macular holes closed successfully after the primary vitrectomy in all eyes in both groups. Although the VAs did not differ significantly before surgery or 1 year after surgery between the 2 groups, the VA improvement was significantly greater 1 and 3 months after surgery ( P = .02, for both) in the 23-gauge group compared with the 20-gauge group. The induced corneal astigmatism levels 1 week and 1 and 3 months after surgery were significantly lower ( P = .01, P = .01, and P = .03, respectively) and the surgical time was significantly shorter ( P = .01) in the 23-gauge group than in the 20-gauge group. No apparent complications developed in either group.
Conclusions
Since 23-gauge vitrectomy combined with phacoemulsification surgery is advantageous because the VA improved rapidly after treating the macular holes with an acceptable safety profile, idiopathic macular holes are a good indication for 23-gauge vitrectomy combined with phacoemulsification surgery.
Since Chen initially proposed sutureless vitrectomy in 1996, the procedure has gained in popularity. Fujii and associates, who proposed that modifications in vitrectomy instrumentation could lead to decreased instrumentation size and consequently smaller incisions, introduced 25-gauge vitrectomy with sutureless self-sealing sclerotomies. Eckardt introduced sutureless 23-gauge vitrectomy in 2005. With larger-gauge instruments, there is less instrument flexibility, allowing greater ocular rotation and the ability to perform a more complete peripheral vitrectomy. Because of its well-known intraoperative and postoperative advantages, that is, reduction of surgical time and postoperative inflammation and less pain, small-gauge transconjunctival vitrectomy is gaining wide acceptance. However, compared with the conventional 20-gauge system, less adequate peripheral vitrectomy with the more flexible instruments has been reported. Macular diseases can be good candidates for sutureless transconjunctival small-gauge vitrectomy to capitalize on the advantages and decrease the disadvantages of the surgical system.
Since Kelly and Wendel reported recovery of anatomic structure and visual acuity (VA) after vitreous surgery in eyes with a macular hole, the surgery has been performed widely, and peeling of the internal limiting membrane (ILM) during surgery has been reported to improve the anatomic success rate. A common complication of vitreous surgery in phakic eyes is nuclear sclerotic cataract progression, which frequently leads to cataract extraction within 2 years after the vitrectomy performed during macular hole surgery. Macular holes commonly are found in older patients, and combined cataract extraction and vitrectomy long has been described as a valid treatment for various vitreoretinal disorders. Thus, at our institution, all vitrectomies for idiopathic macular holes in patients 50 years and older are combined with phacoemulsification and aspiration and intraocular lens implantation (phacoemulsification surgery). In the current study, we compared the 1-year results of 23-gauge and conventional 20-gauge vitrectomies combined with phacoemulsification surgery to repair idiopathic macular holes.
Methods
We retrospectively reviewed 50 eyes of 47 consecutive patients with an idiopathic macular hole who underwent 20-gauge vitrectomy combined with phacoemulsification surgery and 50 eyes of 48 consecutive patients with the disorder who underwent 23-gauge vitrectomy combined with phacoemulsification surgery performed by the same surgeon (T.H.), who was experienced with both vitreous and phacoemulsification surgeries ( Table ). Twenty-gauge vitrectomies were performed from April 2004 through June 2006, and 23-gauge vitrectomies were performed from July 2006 through December 2007. All patients were followed up for more than 1 year after surgery. At our institution, all vitrectomies for idiopathic macular holes were converted from conventional 20-gauge surgery to 23-gauge surgery in July 2006. All vitrectomies performed by the same surgeon for treating idiopathic macular holes during these periods were combined with phacoemulsification surgery.
| Parameter | 23 Gauge | 20 Gauge |
|---|---|---|
| No. eyes/patients | 50/48 | 50/47 |
| Age (yrs) | ||
| Mean ± SD | 66 ± 8 | 67 ± 6 |
| Range | 49 to 86 | 52 to 81 |
| Gender | ||
| Men (eyes, %) | 17 patients (17, 34) | 15 patients (15, 30) |
| Women (eyes, %) | 31 patients (33, 66) | 32 patients (35, 70) |
| Macular hole stage, eyes (%) | ||
| 2 | 7 (14) | 9 (18) |
| 3 | 31 (62) | 28 (56) |
| 4 | 12 (24) | 13 (26) |
| LogMAR VA | ||
| Mean preoperative | 0.40 | 0.36 |
| Range | –0.08 to 1.0 | 0.1 to 1.0 |
Surgical Procedure
All patients underwent a surgical procedure combined with phacoemulsification surgery under local anesthesia. One to 2 hours before surgery, 0.1% diclofenac (Wakamoto Pharmaceutical Co. Ltd., Tokyo, Japan), 5.0% phenylephrine (Kowa Pharmaceutical Co. Ltd., Tokyo, Japan), 0.5% tropicamide (Santen Pharmaceutical Co. Ltd., Osaka, Japan), and 1% cyclopentolate (Santen Pharmaceutical Co. Ltd.) eye drops were instilled. Phacoemulsification surgery was performed before vitrectomy in all cases. After creating a superior conjunctival peritomy, a superior 2.5-mm posterior limbal incision was created. Sodium hyaluronate 1% (Healon; Abbott Medical Optics, Inc., Santa Ana, California, USA) was used as a cohesive ophthalmic viscosurgical device during creation of a continuous curvilinear capsulorrhexis. Phacoemulsification was followed by aspiration of the cortical remnants using the 20000 Legacy or Infinity Vision System (Alcon Laboratories, Inc, Fort Worth, Texas, USA). The ophthalmic viscosurgical device was used to expand the capsular bag into which a foldable acrylic intraocular lens was implanted. No sutures were used to close the incisions.
The Accurus Vitrector (Alcon Laboratories) was used for vitrectomy in all cases. Triamcinolone acetonide (40 mg/1 mL; Kenacort-A, Bristol Myers Co. Ltd., Tokyo, Japan) mixed with 4 mL balanced salt solution was left standing for 30 minutes, and the vehicle of the suspension was discarded. The remaining triamcinolone acetonide was mixed with 4 mL balanced salt solution and was used as a triamcinolone acetonide suspension. After core vitrectomy, approximately 0.2 mL of triamcinolone acetonide suspension was injected over the posterior pole. The posterior hyaloid membrane was detached at the optic disc by aspiration using a vitrector in the absence of a posterior vitreous detachment. A subtotal vitrectomy then was performed. Approximately 0.1 mL triamcinolone acetonide suspension was injected over the posterior pole, and excess triamcinolone acetonide was aspirated with a vitrector. Numerous triamcinolone acetonide particles were observed as white specks on the posterior retina. The ILM was incised with a microhook and was peeled more than 2 disc areas around the macular area using ILM forceps. Excess triamcinolone acetonide then was aspirated again. When triamcinolone acetonide particles were found in the macular hole after the ILM was peeled, the triamcinolone acetonide was aspirated with a vitrector over the macular hole. Fluid–air exchange then was performed. Other gases such as sulfur hexafluoride or perfluoropropane were not used in any cases. All patients were instructed to maintain a face-down position for 3 days after surgery. The surgical procedures mentioned above were the same in both the 23- and 20-gauge vitrectomy groups.
Postoperative treatment consisted of 0.5% levofloxacin (Santen Pharmaceutical Co. Ltd.), 0.02% dexamethasone metasulfobenzoate sodium (Santen Pharmaceutical Co. Ltd.), and 0.1% diclofenac sodium eye drops applied topically three times daily for 2 weeks and oral cefcapene pivoxil hydrochloride (Shionogi & Co., Ltd., Osaka, Japan) 75 or 100 mg 3 times daily for 3 days in all cases in both groups.
23-Gauge Vitrectomy
A 23-gauge vitrectomy was performed using the Eckardt 23-gauge vitrectomy system (Dutch Ophthalmic Research Center, Zuidland, The Netherlands), and sclerotomies were performed using a 2-step approach. The conjunctiva was displaced, and angled scleral incisions were made through the pars plana with a 23-gauge angled microvitreoretinal blade in the inferotemporal, supranasal, and supertemporal quadrants. Trocars were inserted through the wounds, and the infusion trocar was placed in the inferotemporal quadrant. During vitrectomy, a vitreous cutter was set to 2500 cycles per minute, and its maximum aspiration vacuum was 600 mm Hg. After the surgical procedures, the trocars were removed and the conjunctiva was repositioned over the sclerotomy sites.
20-Gauge Vitrectomy
After conjunctival peritomy, 20-gauge sclerotomies were performed and an infusion cannula was positioned. During the vitrectomy, a vitreous cutter was set to 2500 cycles per minute and its maximum aspiration vacuum was 200 mm Hg. At the end of surgery, the sclerotomies were closed using an 8.0 Vicryl suture (Johnson & Johnson K.K., Tokyo, Japan) in a mattress fashion, and conjunctival incisions also were closed using an 8.0 Vicryl suture.
The recorded data included patient age, gender, best-corrected VA, keratometry, intraocular pressure (IOP), and the flare value in the anterior chamber. The best-corrected VA, keratometry, and the IOP values were recorded before surgery and at 1 week and 1, 3, 6, and 12 months after surgery, and the flare values in the anterior chamber were measured before surgery and 1 week and 1 month after surgery. Those examinations at the various time points were performed routinely before surgery and after surgery. Best-corrected VA was measured using a Landolt ring chart. The IOP and corneal astigmatism values were measured using pneumatic tonometry and autokeratorefractometry (RKT-7700; Nidek Co., Ltd., Gamagori, Aichi, Japan). The flare values in the anterior chamber were measured using the laser-flare meter (FM-600; Kowa Company, Ltd., Nagoya, Japan). Because the authors knew that the flare value in the anterior chamber improved to the preoperative level within 3 months after surgery at the early phase of this study, the flare values were measured in 15 eyes in the 23-gauge group and in 20 eyes in the 20-gauge group 3, 6, and 12 months after surgery. Furthermore, to compare the flare values in the anterior chamber between vitrectomy combined with and without phacoemulsification surgery, 11 pseudophakic eyes treated with vitrectomy alone for an idiopathic macular hole were examined using the laser-flare meter before surgery and 1 week after surgery. Of the 11 pseudophakic eyes, 5 eyes underwent 23-gauge vitrectomy and 6 eyes underwent 20-gauge vitrectomy.
The surgical times were compared between the groups. In the 23-gauge vitrectomy group, the surgical time included phacoemulsification surgery, insertion of trocars, vitrectomy, removal of the preretinal membrane and ILM, fluid–air exchange, and withdrawal of trocars. In the 20-gauge vitrectomy group, the surgical time included phacoemulsification surgery, creation of the sclerotomies, positioning of the infusion cannula, vitrectomy, removal of the preretinal membrane and ILM, fluid–air exchange, scleral suturing, and conjunctival suturing. The rate of improvement of logarithm of the minimal angle of resolution VA was calculated as follows: (postoperative value − preoperative value) × 100/(1-year postoperative value − preoperative value). The surgically induced corneal astigmatism was calculated by vector analysis described previously.
Statistical Analysis
The difference between the groups was assessed for statistical significance using the Student t test when appropriate. The VA was converted to logarithm of the minimal angle of resolution units for statistical analysis. Statistical analysis was conducted using SPSS software version 11.5.1 for Windows (SPSS, Inc, Chicago, Illinois, USA). A P value less than .05 was considered significant.
Results
The Table shows the demographic and baseline characteristics of the eyes in the 2 study groups. No significant differences were found among the preoperative characteristics between the 2 groups.
A posterior vitreous detachment was created during surgery in 38 (76%) eyes in the 23-gauge group and 37 (74%) eyes in the 20-gauge group. Retinal breaks associated with the maneuvers performed to create a posterior vitreous detachment developed in 6 (16%) of 38 eyes and ion 7 (19%) of 37 eyes in the 23- and 20-gauge groups, respectively, and were treated effectively with photocoagulation during surgery. No retinal detachments developed during the follow-up period. No other complications developed during surgery. No patient required a suture at the 23-gauge sclerotomy site because of leakage, and no sclerotomies were converted to the 20-gauge procedure. No severe postoperative hypotony such as wound leakage confirmed by the Seidel test or an IOP of less than 6 mm Hg developed in either group, although in 2 eyes in the 23-gauge group, the IOP was less than 8 mm Hg 1 day after surgery and increased to more than 10 mm Hg 3 days after surgery in the 2 eyes. No choroidal detachment or bacterial endophthalmitis developed in either group. No eye in either group except 1 eye (2%) in the 20-gauge group had a rupture of the posterior capsule of the crystalline lens during phacoemulsification surgery. No eyes had posterior synechiae of the iris to the intraocular lens or iris capture after surgery. The mean surgical time (± standard deviation) was 37 ± 8 minutes in the 23-gauge group, which was significantly ( P = .01) shorter than in the 20-gauge group (50 ± 9 minutes).
The macular holes successfully closed after the primary vitrectomy in all eyes in both groups. The VAs 1 and 3 months after surgery were significantly better in the 23-gauge group than in the 20-gauge group ( P = .01 and P = .03, respectively); however, the VAs did not differ significantly before surgery or 1 week and 6 and 12 months after surgery between the 2 groups ( Figure 1 ). In both groups, the VA improved significantly 12 months after surgery compared with before surgery ( P = .01). Although the VA improvement was greater 1 and 3 months after surgery in the 23-gauge group compared with the 20-gauge group ( P = .02), no significant difference was found in the VA improvement 1 week and 6 and 12 months after surgery between the groups ( Figure 2 ). The induced corneal astigmatism was lower 1 week and 1 and 3 months after surgery in the 23-gauge group compared with the 20-gauge group ( P = .01, P = .01, and P = .03, respectively; Figure 3 ).

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


