FIGURE 2.1 Artist diagram of hyaloid artery at its pinnacle in a 40-mm human embryo. a, pupillary membrane; b, anterior tunica vasculosa lentis; c, outer set of vasa hyaloidea propria; d, inner set of vasa hyaloidea propria; e, main trunk of hyaloid artery emerging from optic cup. (Reprinted from Mann I. Development of the human eye, 3rd ed. Australia: Grune & Stratton, 1964, with permission.)
The structure of the hyaloid artery consists of three layers, the intima, the media, and the adventitia (3). The intima consists of flattened nonfenestrated endothelial cells connected by tight junctions that have a basement membrane and an incomplete layer of pericytes. The media has concentric layers of smooth muscle with basement membranes around each fiber. The adventitia contains scattered fibroblasts and collagen.
The walls of the vasa hyaloidea propria and the tunica vasculosa lentis are small capillaries that consist of a complete layer of nonfenestrated endothelium with intervening tight junctions between adjacent endothelial cells encircled by a continuous basement membrane and incomplete layer of pericytes in primates (4).
The formation of the hyaloid has been proposed to be by hemovasculogenesis (5) and becomes most prominent between 8 and 12 weeks (2). Regression of the hyaloidal circulation begins at 12 weeks (6) with the vasa hyaloidea propria, followed by the tunica vasculosa lentis, and then the pupillary membrane and finishes at about 35 to 36 weeks’ gestation with the complete loss of blood flow in the hyaloidal artery (2). As the hyaloidal vasculature regresses, there is contemporaneous development of the retinal vasculature. The primary vitreous retracts and collagen fibers and hyaluronic acid are produced, which make up the secondary vitreous. The posterior segment is composed largely of the secondary vitreous by the 6th month of gestation, and the primary vitreous at this point is reduced to a central extension from the optic disc to the posterior lens surface known as Cloquet canal (Fig. 2.2).
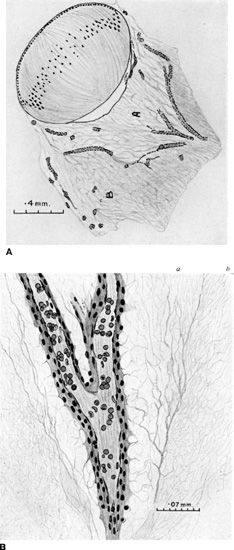
FIGURE 2.2 A: Artist diagram of a 48-mm human embryo with hyaloidal artery regression and early formation of primary and secondary vitreous. A, primary vitreous; B, secondary vitreous being formed by disappearance of vasa hyaloidea propria. B: Artist diagram of a 65-mm human embryo demonstrating Cloquet canal and the contained hyaloid artery in central primary vitreous. The condensation between primary and secondary vitreous is seen forming the wall of Cloquet canal. a, primary; b, secondary itreous. (Reprinted from Mann I. Development of the human eye, 3rd ed. Australia: Grune & Stratton, 1964, with permission.)
Ida Mann described the regression of the human hyaloid through detailed observations (2). She noted that as the lens increased in size, the mesh of intertwined vessels that made up the tunica vasculosa lentis along the posterior capsule became stretched and developed decreased vessel caliber. At the same time, the vasa hyaloidea propria branches had reduced caliber at their proximal ends near the origin at the main hyaloid artery and eventually lost connection with it, although the distal ends of the vasa hyaloidea propria remained continuous with vessels on the posterior surface of the lens (Fig. 2.3). The atrophy of the vasa hyaloidea propria and of the more central set of vitreous branches is normally complete by 8½ months. During the 7th month, atrophy of the anterior tunica vasculosa lentis or pupillary membrane begins. The atrophy of the pupillary membrane begins centrally over the lens where the vessels are thinner and consists of gradual shrinkage of vessel walls with decreasing lumen size and, finally, cessation of blood flow. Once cessation of blood flow occurs, the central vessels shrink away (Fig. 2.4). By 8½ months most of the central loops of the pupillary membrane have atrophied. Endothelial cell processes then fill the lumen, and macrophages form a plug that occludes the vessel. The cells in the vessel wall then undergo necrosis and are phagocytized by mononuclear phagocytes (7). Frequently, remains of the pupillary membrane can be seen in fine strands that extend along the pupil/iris margin.
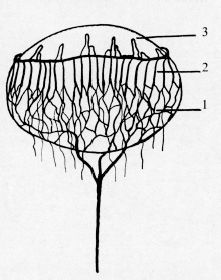
FIGURE 2.3 Artist diagram showing the relation of the hyaloid vessels on the lens during atrophy of the vasa hyaloidea propria. 1, remains of the vasa hyaloidea propria; 2, vessels of tunica vasculosa lentis; 3, pupillary membrane vessels. Note main trunk of hyaloid posterior to lens. (Reprinted from Mann I. Development of the human eye, 3rd ed. Australia: Grune & Stratton, 1964, with permission.)
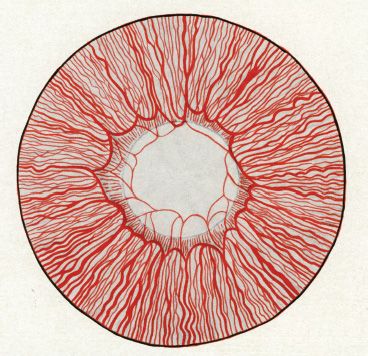
FIGURE 2.4 Artist representation of the appearance of the iris and the pupillary membrane of a 7-month human fetus. Note the thinner caliber of the central loops of the pupillary membrane. These eventually shrink away. Not infrequently, remnants of the pupillary membrane can be seen at the iris edge. (Reprinted from Mann I. Development of the human eye, 3rd ed. Australia: Grune & Stratton, 1964, with permission.)
Regression of the hyaloid artery and its associated branches usually occurs completely and without complications. Persistence of the hyaloid vascular system occurs in 3% of full-term infants and in 95% of premature infants. A persistent hyaloid artery can be associated with prepapillary or vitreous hemorrhage (8). Anomalies involving incomplete regression of the embryonic hyaloid vascular system occur in more than 90% of infants born younger than 36 weeks of gestation and in over 95% of infants weighing <5 pounds at birth (9).
MOLECULAR EVENTS INVOLVED IN THE HYALOID VASCULATURE
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


