Purpose
To compare the clinical outcomes of wavefront-guided and wavefront-optimized laser in situ keratomileusis (LASIK).
Design
Prospective, randomized, fellow-eye–controlled study.
Methods
The setting was a single academic institution. The study population included 110 eyes of 55 patients with myopia with and without astigmatism. One eye of each patient was randomized to undergo wavefront-guided LASIK by the AMO Visx CustomVue S4 IR excimer laser system; the fellow eye received wavefront-optimized LASIK by the Alcon Allegretto Wave Eye-Q 400 Hz excimer laser system. Corneal flaps were constructed using the Intralase FS 60 Hz femtosecond laser. Patients were followed at postoperative months 1, 3, 6, and 12. The study’s main outcome measures were uncorrected visual acuity, stability of refractive correction, contrast sensitivity, and wavefront aberrometry.
Results
After 12 months, LASIK eyes had achieved visual acuity of 20/12.5 or better (30 eyes, 56%) in the wavefront-guided group compared to those receiving wavefront-optimized treatment (22 eyes, 41%) ( P = 0.016). Average spherical equivalent refractions were −0.13 ± 0.46 diopters in wavefront-guided eyes whereas in wavefront-optimized eyes the refractions were −0.41 ± 0.38 diopters at 12 months. Wavefront-guided eyes also achieved better best-corrected visual acuity at both the 5% and 25% contrast levels ( P = 0.022 and P = 0.004, respectively). There were no differences in levels of residual astigmatism ( P = 0.798) or in higher order aberrations ( P = 0.869).
Conclusions
Both wavefront-guided and wavefront-optimized treatments are able to correct myopia safely and effectively in eyes with and without astigmatism. However, wavefront-guided treatment platforms appear to offer significant advantages in terms of residual refractive error, uncorrected distance acuity and contrast sensitivity.
Conventional excimer ablations are determined only by an eye’s existing lower order aberrations and generally change the shape of the cornea from prolate to a more oblate profile, increasing patients’ higher order aberrations (HOAs). These induced HOAs can be associated with undesired symptoms, such as glare, halo and starbursts and a decrease in contrast sensitivity. Newer techniques, including wavefront-guided excimer ablations, which take into account patients’ prior HOAs in addition to lower order aberrations, have been developed. Wavefront-guided ablations are based on preoperative measures of HOAs with the intent to decrease existing HOAs. Wavefront-guided ablations can produce a reduction in prior existing HOAs and less induction of new HOAs when compared to conventional treatment. The wavefront-optimized platform was developed to address variations in ablation depth that arise from corneal shape when eccentric laser pulses are applied to the cornea. This should decrease the degree of induction to an oblate profile as well as other HOAs during surgery, which have been noted in conventional treatment and which could improve visual outcomes. The wavefront-optimized platform generates ablation profiles that are not only based on corneal shape but also use population norms of aberration measures including HOAs. The wavefront-optimized technique has been found to decrease induced HOAs when compared to conventional laser in situ keratomileusis (LASIK).
Previous studies comparing wavefront-guided and wavefront-optimized treatments using different excimer laser platforms have yielded mixed results. Several of these studies have shown no difference between the two profiles. However, some studies have suggested improved outcomes with wavefront-guided treatment. These studies have been limited by small sample sizes, retrospective design or short duration of follow-up. Our study used a prospective randomized contralateral-eye–controlled design with a large cohort size and a year of follow-up in order to compare the refractive results of the 2 treatment platforms.
Methods
This prospective, randomized, fellow-eye–controlled study protocol was approved by the Stanford University Institutional Review Board to compare the safety, efficacy, predictability, and contrast acuity outcomes of wavefront-guided and wavefront-optimized LASIK for patients with myopia. Patients were consecutively recruited at the Stanford Laser Eye Center between April 2009 and March 2011. The trial was publically registered with the National Institutes of Health at http://www.clinicaltrials.gov/ ( NCT01135719 ). Inclusion criteria included patients 21 years of age or older, with myopia between −0.25 diopters (D) and −12.00 D, with or without astigmatism, up to 3.00 D, stable refraction with a <0.50 D change in sphere and cylinder recorded over the prior 12 months; best correctable visual acuity of ≥20/20 in both eyes; cessation of soft contact lens use for ≥7 days before the preoperative visit; and ability to participate in follow-up examinations for 12 months following refractive surgery. Exclusion criteria included age younger than 21; excessively thin corneas (<500 μm central corneal thickness); topographic evidence of keratoconus ; eyes with ectactic disorders ; histories of autoimmune diseases, pregnancy or current nursing of an infant; and greater than 2.5 D of difference in sphere and cylinder between eyes. Each patient meeting these criteria and willing to participate in the study signed an Institutional Review Board-approved informed consent form prior to participating in the study, and the protocol adhered to the tenets of the Declaration of Helsinki.
Patients enrolled in the study underwent a comprehensive preoperative evaluation, including history, manifest and cycloplegic refraction using the Early Treatment of Diabetic Retinopathy Study visual acuity charts; examination by slit-lamp biomicroscopy; Goldmann applanation tonometry; infrared pupillometry (Neuroptics, Irvine, California) under photopic and scotopic conditions; dilated fundus examination; VISX Wavescan Wavefront (Abbott Medical Optics, Santa Ana, California); and computerized corneal topography using the Pentacam (Oculus, Wetzlar, Germany). Measurements of best corrected visual acuity (BCVA) were also made under controlled ambient conditions with 5% and 25% contrast sensitivity conditions (Precision Vision, La Salle, Illinois).
Wavefront aberrations were measured in an undilated pupil. HOAs were subject to variability due to measuring at different pupil diameters, thus for data analysis we used aberrometry images that were within 0.25 mm of the preoperative measurements. Although luminance was not measured, all measurements were made in the same room with the same dark lighting conditions for all patients. To assess aberrometry readings, 6 readings were performed on each visit before and after surgery. In all patients aberrometry data were analyzed and treatment was planned for both eyes prior to the actual treatment. The randomization envelope was opened on the day of surgery, and the eye selected for wavefront-guided treatment received the preplanned treatment in that eye and wavefront-optimized treatment in the fellow eye. Preoperatively, the aberrometry reading most closely matching the manifest refraction was chosen for treatment. Postoperatively, the scan within 0.25 mm of the preoperative pupil diameter scan was chosen for analysis. All scans had to be rated as high quality (4 greens) to be used for preoperative surgery planning and postoperative analysis.
Of 55 patients, 110 eyes were randomized to receive either wavefront-guided or wavefront-optimized LASIK. Randomization was performed by assigning the dominant eye, as determined by the Dolman method, to a treatment based on a computer-generated schedule. Each dominant eye was randomly assigned to receive wavefront-guided or wavefront-optimized LASIK, and the contralateral eye received the alternative procedure. Topical proparacaine hydrochloride 0.5% (Ophthetic; Allergan, Irvine, California); moxifloxacin hydrochloride ophthalmic solution 0.5% (Vigamox; Alcon, Fort Worth, Texas); and ketorolac tromethamine ophthalmic solution 0.4% (Acular LS; Allergan) were administered immediately before the procedure. Wavefront-guided procedures were performed with the VISX Star Custom Vue S4 IR excimer laser (Abbot Medical Optics, Santa Ana, California). Wavefront-optimized procedures were performed with the Wavelight Allegretto Wave Eye-Q 400 Hz excimer laser (Alcon, Huenberg, Switzerland). Cornea flaps were constructed using the Intralase FS 60 Hz femtosecond laser (Abbot Medical Optics) in all eyes using identical laser settings in both groups. Flaps were 9.0 mm in diameter with an intended thickness of 105 μm. Flaps were created in both eyes prior to any excimer laser treatment. Flap creation was performed first in the right eye in all patients. In order to eliminate any potential effects of the second eye surgery, an equal number of eyes underwent wavefront-guided ablation first, followed by wavefront-optimized ablation second (ie, 50 eyes were treated with wavefront-guided ablation first and 50 eyes were treated with wavefront-optimized ablation first). Ultrasound pachymetry was performed preoperatively and intraoperatively to determine flap thickness by subtraction pachymetry. Postoperative medications included topical moxifloxacin 4 times daily for 4 days and prednisolone acetate 1.0% (Pred Forte; Allergan) 4 times daily for 7 days.
The AMO (Abbot Medical Optics) VISX CustomVue Star S4IR excimer laser system uses variable spot scanning technology in which the beam sizes range from a minimum of 0.65 mm to a maximum of 6.5 mm. It also uses a variable repetition rate in which the repetition rate varies from a minimum of 6 Hz to a maximum of 20 Hz. The system utilizes iris registration to account for any cyclorotational misalignments as well as centroid shift. The system uses a 60 Hz active infrared eye tracking that compensates for eye movements along the x, y and z axes. The standard default optical treatment zone is 6.0 mm with an 8 mm transition blend zone.
The Alcon Wavelight Allegretto Wave Eye-Q 400 Hz excimer laser utilizes a fixed 0.95 μm scanning spot technology. The system uses a 400 Hz repetition rate and a 200 Hz active infrared eye tracking that compensates for eye movements. The standard default optical treatment zone is 6.5 mm with an 8 mm transition blend zone.
Patients were evaluated at 1 day, 1 week, 1 month, 3 months, 6 months, and 12 months following LASIK surgery. Primary outcome measures included uncorrected visual accuity, refractive stability, predictability, loss of best-spectacle corrected visual acuity, aberrometry, contrast sensitivity, and adverse event profile. Statistical analysis was performed using a 2-tailed, paired t test and a Wilcoxon signed rank test (Microsoft Excel ToolPak Software; Microsoft, Redmond, Washington). For all statistics, a P value of <0.05 was considered significant.
Results
There was excellent retention of patients through the trial; all 55 returned for the postoperative 1-month exam, 54 returned at 3 months, 53 at 6 months, and 54 at 12 months. Table 1 shows the preoperative refractive characteristics of the eyes, which were not significantly different between the 2 groups ( P > 0.500). The mean age of patients was 37 ± 9.4 (range, 24 to 55) years. Of the subjects 47% (n = 26) were male, and 53% (n = 29) were female. Preoperatively, the average corneal thickness was 552 ± 31 (range, 500–656) μm in the wavefront-guided group and 552 ± 31 (range, 507–658) μm in the wavefront-optimized group. The calculated mean ablation depth was 78 ± 27 (range, 27–124) μm in the wavefront-guided group, a significant 5.27 μm ( P = 0.012) more, on average, than the wavefront-optimized group in whom the mean ablation depth was 73 ± 71 (range, 23–120) μm.
| Wavefront-Guided (Mean ± SD) | Wavefront-Optimized (Mean ± SD) | P- Value | |
|---|---|---|---|
| Scotopic pupil size (mm) | 6.59 ± 0.94 | 6.56 ± 1.01 | 0.536 |
| Sphere | −5.22 ± 2.38 | −5.26 ± 2.07 | 0.763 |
| Cylinder | 0.94 ± 0.86 | 0.91 ± 0.91 | 0.733 |
| Spherical equivalent | −4.75 ± 2.22 | −4.81 ± 1.95 | 0.669 |
| Coma | 0.18 ± 0.12 | 0.18 ± 0.11 | 0.930 |
| Trefoil | 0.16 ± 0.10 | 0.16 ± 0.10 | 0.869 |
| Spherical aberration | 0.12 ± 0.15 | 0.13 ± 0.15 | 0.669 |
| Higher order aberrations | 0.35 ± 0.14 | 0.35 ± 0.15 | 0.757 |
Efficacy
Table 2 summarizes the residual refractive errors at the 1-, 3-, 6-, and 12-month follow-up visits after treatment. There was significantly more negative sphere and spherical equivalent in the wavefront-optimized eyes at all times except at 3 months ( P < 0.007). There was also significantly more trefoil in the wavefront-optimized eyes at 1, 3 and 6 months ( P < 0.001), with the difference just missing significance at 12 months ( P = 0.051).
| Wavefront-Guided (Mean ± SD) | Wavefront-Optimized (Mean ± SD) | P- Value | |
|---|---|---|---|
| Postoperative month 1 (n = 55) | |||
| Sphere | −0.17 ± 0.41 | −0.39 ± 0.32 | <0.001 |
| Cylinder | 0.17 ± 0.24 | 0.29 ± 0.30 | 0.026 |
| Spherical equivalent | −0.09 ± 0.43 | −0.24 ± 0.34 | 0.006 |
| Coma | 0.23 ± 0.14 | 0.25 ± 0.16 | 0.241 |
| Trefoil | 0.11 ± 0.07 | 0.16 ± 0.09 | <0.001 |
| Spherical aberration | 0.20 ± 0.19 | 0.19 ± 0.17 | 0.639 |
| Higher order aberrations | 0.40 ± 0.18 | 0.43 ± 0.19 | 0.117 |
| Postoperative month 3 (n = 54) | |||
| Sphere | −0.40 ± 1.1 | −0.48 ± 0.86 | 0.265 |
| Cylinder | 0.23 ± 0.34 | 0.25 ± 0.38 | 0.677 |
| Spherical equivalent | −0.29 ± 0.99 | −0.35 ± 0.74 | 0.334 |
| Coma | 0.23 ± 0.13 | 0.24 ± 0.14 | 0.959 |
| Trefoil | 0.10 ± 0.06 | 0.15 ± 0.09 | <0.001 |
| Spherical aberration | 0.21 ± 0.19 | 0.17 ± 0.18 | 0.133 |
| Higher order aberrations | 0.40 ± 0.17 | 0.41 ± 0.16 | 0.662 |
| Postoperative month 6 (n = 53) | |||
| Sphere | −0.21 ± 0.43 | −0.44 ± 0.39 | <0.001 |
| Cylinder | 0.19 ± 0.23 | 0.23 ± 0.24 | 0.296 |
| Spherical equivalent | −0.12 ± 0.41 | −0.33 ± 0.41 | <0.001 |
| Coma | 0.26 ± 0.15 | 0.27 ± 0.15 | 0.949 |
| Trefoil | 0.11 ± 0.07 | 0.17 ± 0.12 | <0.001 |
| Spherical aberration | 0.24 ± 0.20 | 0.22 ± 0.19 | 0.211 |
| Higher order aberrations | 0.46 ± 0.18 | 0.47 ± 0.19 | 0.639 |
| Postoperative month 12 (n = 54) | |||
| Sphere | −0.28 ± 0.45 | −0.56 ± 0.40 | <0.001 |
| Cylinder | 0.28 ± 0.3 | 0.30 ± 0.31 | 0.633 |
| Spherical equivalent | −0.13 ± 0.46 | −0.41 ± 0.38 | <0.001 |
| Coma | 0.28 ± 0.16 | 0.28 ± 0.17 | 0.809 |
| Trefoil | 0.13 ± 0.08 | 0.17 ± 0.11 | 0.051 |
| Spherical aberration | 0.23 ± 0.19 | 0.21 ± 0.17 | 0.119 |
| Higher order aberrations | 0.46 ± 0.19 | 0.47 ± 0.19 | 0.869 |
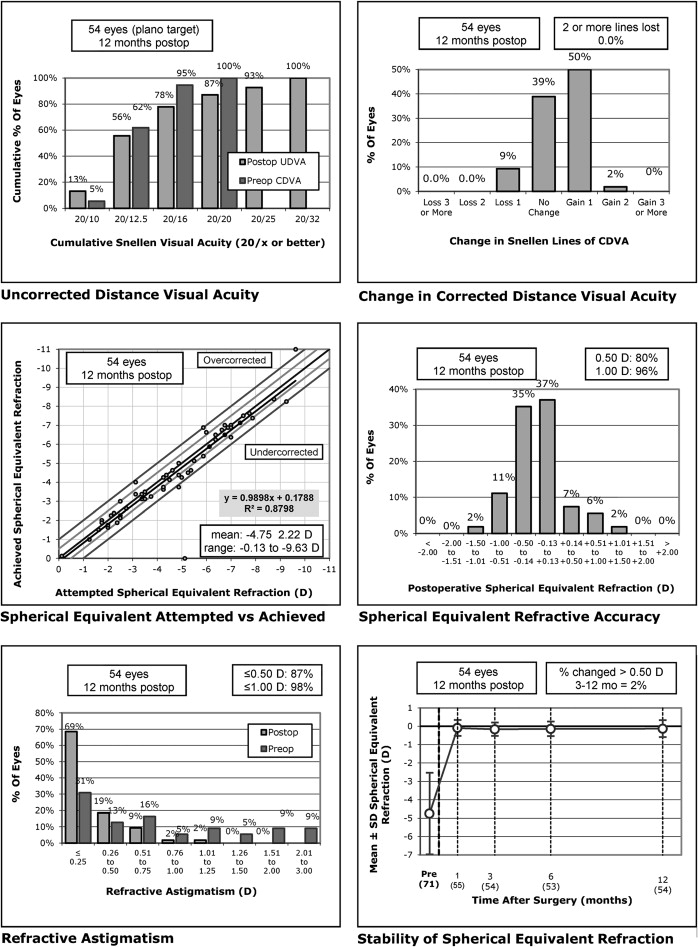
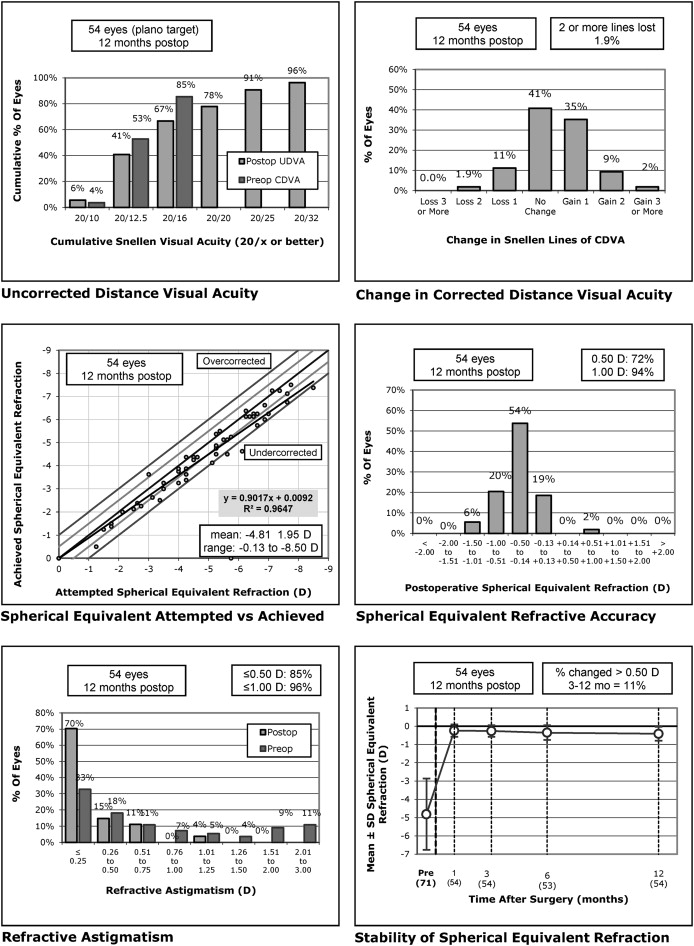
Figure 1 (top left) shows the distribution of uncorrected distance acuity at 12 months compared to BCVA preoperatively for the wavefront-guided group and Figure 2 (top left) shows the same for the wavefront-optimized group. At 12 months, the uncorrected distance acuity was 20/32 or better in 100% and 20/20 or better in 87% of eyes in the wavefront-guided group. In the wavefront-optimized group, 100% of eyes were 20/40 or better, and 78% of eyes were 20/20 or better. There were no statistically significant differences between the groups when comparing change from preoperative uncorrected visual acuity at any of the months of follow-up ( P > 0.126). At 12 months, significantly more eyes that had received wavefront-guided treatment achieved vision of 20/10 (7 eyes, 13%) and 20/12.5 (23 eyes, 43%) than eyes receiving wavefront-optimized treatment (3 eyes (6%) achieved 20/10 and 19 eyes (25%) achieved 12/12.5) ( P = 0.016).
Safety
There were no intraoperative or postoperative flap complications in either group. No eyes were retreated within the first 12 months. Figure 1 (top right) and Figure 2 (top right) show the change in BCVA at 12 months compared to preoperatively for the wavefront-guided and wavefront-optimized groups, respectively. No eyes lost more than 2 lines of BCVA in either group. In the wavefront-guided group, 5 eyes (9%) lost 1 line of BCVA, 21 eyes (39%) were unchanged, and 27 eyes (52%) gained 1 or more lines of BCVA. In the wavefront-optimized group, 1 eye (2%) lost 2 lines of BCVA, 6 eyes (11%) lost 1 line of BCVA, 22 eyes (41%) were unchanged, and 25 eyes (46%) gained 1 or more lines of BCVA. There were no statistically significant differences in the change in BCVA between the 2 groups at any time ( P > 0.200). There were significantly more eyes in the wavefront-guided treatment group with visual acuities of 20/10 and 20/16 or better BCVA ( P = 0.008).
Accuracy, Predictability, and Stability
Figure 1 (middle left) and Figure 2 (middle left) show the spherical equivalent correction attempted compared to that which was achieved at 12 months for the wavefront-guided and wavefront-optimized groups, respectively. In the first month, 86% of eyes were within 0.5 D and 98% were within 1.0 D of attempted correction in the wavefront-guided group. In the wavefront-optimized group, 88% of eyes were within 0.5 D, and 98% were within 1.0 D of attempted correction. By the third month, 91% of eyes were within 0.5 D and 100% were within 1.0 D of attempted correction in the wavefront-guided group. In the wavefront-optimized group, 86% of eyes were within 0.5 D and 100% were within 1.0 D of attempted correction. This was slightly reduced at 12 months for both groups, as shown in Figure 1 (middle right) and Figure 2 (middle right) for the wavefront-guided and wavefront-optimized groups, respectively. A significantly higher proportion of eyes were closer to emmetropia in the wavefront-guided group ( P = 0.031).
Figure 1 (bottom right) and Figure 2 (bottom right) show the stability of the spherical equivalent refraction at 12 months for the wavefront-guided and wavefront-optimized eyes, respectively. A higher proportion of eyes had 0.5 D or more change compared to the refraction achieved at 3 months in the wavefront-optimized group (11% compared to 2% in the wavefront-guided group). Over time, the average spherical equivalent remained close to emmetropia in the wavefront-guided group, with an average of −0.13 ± 0.46 D at 12 months from −0.09 ± 0.43 D at 1 month, whereas eyes in the wavefront-optimized group became slightly more myopic, from −0.24 ± 0.34 D at 1 month to an average of −0.41 ± 0.38 D at 12 months.
Astigmatism and Higher Order Aberrations
Figure 1 (bottom left) and Figure 2 (bottom left) show the residual astigmatism in eyes after treatment compared to preoperatively. In 85% or more of eyes there was 0.5 D or less of astigmatism after treatment, with no significant difference between the 2 groups ( P = 0.798). There were no significant differences in HOAs between the 2 groups at any time studied ( P > 0.116). There was an average increase in HOAs of 0.11 ± 0.17 μm at 12 months compared to baseline for both groups ( P = 0.936).
Contrast Sensitivity
There were no statistically significant differences in distribution of 25% contrast BCVA in eyes receiving wavefront-optimized or wavefront-guided treatment preoperatively or at 1 and 3 months postoperatively ( P > 0.058). However, at 6 and 12 months, more eyes that had received wavefront-guided treatment had 20/32 or better 25% contrast BCVA ( P = 0.032 and 0.004, respectively). In terms of 5% contrast BCVA, the groups had a similar distribution preoperatively and at 1 month ( P = 0.082 and 0.066, respectively). However, at 3, 6 and 12 months, a higher percentage of wavefront-guided eyes had 20/20 or better BCVA ( P = 0.033, 0.033, and 0.022, respectively). There were no differences between the groups in terms of change compared to preoperative 5% or 25% contrast best-spectacle corrected visual acuity at 1, 3, 6, or 12 months postoperatively ( P > 0.209). Table 3 summarizes the 5% and 25% contrast BCVA results at each time.



