Purpose
To report logarithm of the minimal angle of resolution (logMAR) visual outcomes of the Boston keratoprosthesis type 1.
Design
Prospective cohort study.
Methods
Preoperative, intraoperative, and postoperative parameters of 300 eyes of 300 patients who underwent implantation of a Boston keratoprosthesis type 1 device between January 2003 and July 2008 by 1 of 19 surgeons at 18 medical centers were collected.
Results
After an average of 17.1 ± 14.8 months, visual acuity improved significantly ( P < .0001) to a mean final value of 0.89 ± 0.64 (20/150). There were also significantly fewer eyes with light perception (6.7%; n = 19; P < .0001), although 3.1% (n = 9) progressed to no light perception. There was no association between age ( P = .08), sex ( P = .959), operative side ( P = .167), or failure ( P = .494) and final visual acuity. The median time to achieve 20/200 visual acuity was 1 month (95% confidence interval 1.0–6.0) and it was retained for an average of 47.8 months. Multivariate analysis, controlling for preoperative visual acuity, demonstrated 2 factors associated with final visual outcome: chemical injury was associated with better final vision ( P = .007), whereas age-related macular degeneration was associated with poorer vision ( P < .0001).
Conclusions
The Boston keratoprosthesis type 1 is an effective device for rehabilitation in advanced ocular surface disease, resulting in a significant improvement in visual acuity. Eyes achieved a mean value of 20/150 (0.89 ± 0.64 logMAR units) after 6 months and this was relatively stable thereafter. The best visual prognosis is observed in chemical injury eyes, whereas the worst prognosis is in aniridia, although the latter has limited visual potential.
Keratoprostheses (KPro) were first implanted in animals in 1853, followed by the first human implantation by Heusser in 1859. The initial devices were made from glass and had poor retention, with none retained more than 6 months. While other investigators attempted to improve on the design, long-term retention was still poor, with most KPros extruded. With improvement in both donor corneal storage and techniques of full-thickness corneal transplantation, the interest in an artificial cornea waned. As with intraocular lenses, the discovery of inert plastics (eg, polymethyl methacrylate) in the mid-20th century prompted a resurgence in both the design of and interest in KPros.
Numerous KPro designs emerged in the latter part of the 20th century, with the Boston KPro being the most popular. Originally called the Dohlman-Doane keratoprosthesis, it received US Food and Drug Administration clearance in 1993. Further advances in the refinement of the design and use of topical antibiotic prophylaxis improved the overall retention rate and made keratoprosthesis surgery a viable option for patients who would be poor candidates for penetrating keratoplasties.
In spite of these improvements, keratoprosthesis surgery remained a rarity. In 2003 although over 50,000 transplants were being performed annually in the United States, only 57 Boston KPros were implanted worldwide. Over the last 10 years the acceptance of the Boston KPro and its use has grown exponentially; in 2012 over 1500 Boston KPros were implanted worldwide.
The increased utilization of the Boston KPro led to the first large-scale multicenter study in 2006. The original paper analyzed 141 procedures and reported an overall retention rate of 95%. The study was limited by a short follow-up time of 8.5 months (range 3–24 months). In 2013 we reported on the long-term retention in an expanded study population of over 300 eyes with an additional 4 years of follow-up. Additional papers from the expanded study database analyzed factors that were associated with retroprosthetic membrane formation. The purpose of our current paper is to report on the long-term visual outcomes in 300 Boston KPro eyes.
Methods
The Boston keratoprosthesis type 1 is obtained from the Massachusetts Eye and Ear Infirmary (Boston, Massachusetts, USA). The technique for implanting the Boston keratoprosthesis has been previously described and all surgeons reported using a similar technique.
Data Collection
The Boston Keratoprosthesis Multicenter Study is a large prospective cohort study gathering data under institutional review board approval (Cornea Consultants of Albany, Albany Medical Center Department of Ophthalmology) on Boston keratoprostheses type 1 implanted since January 1, 2003. This study was initiated 2 years prior to the public launch of the clinicaltrials.gov website and is therefore not registered on the site. At the time the study was initiated, all surgeons known to be performing multiple procedures were contacted and encouraged to participate. Surgeons reported data using a mail-in case report form evaluating approximately 70 perioperative variables. Data submissions were voluntary, although all participating surgeons were encouraged to submit data that were as complete as possible, regardless of the outcome. In compliance with Health Insurance Portability and Accountability Act regulations, patients were assigned a unique study number. These forms were sent to a data coordinating center, under institutional review board approval. In general, follow-up visits at 1 month, 6 months, and 12 months and annually thereafter were reported by participating surgeons.
For patients who underwent repeat implantation after keratoprosthesis failure, only data from the first implant were included in this study. If a patient underwent bilateral keratoprosthesis implantation, then only the first eye of the patient was included in the study because the eyes are not independent data points.
Analysis
Based on previously published prognostic categories, the patients were categorized into the following pathologic groups: severe autoimmune disease (ocular cicatricial pemphigoid and Stevens-Johnson Syndrome), chemical injuries, herpes simplex keratitis, Fuchs endothelial dystrophy, keratoconus, infectious keratitis, neurotrophic ulcers, limbal stem cell deficiency (LSCD), pseudophakic bullous keratopathy (PBK), trauma, aniridia, miscellaneous, failed penetrating keratoplasty (PK), and unknown.
A Microsoft Excel spreadsheet was used to compile the data and SAS version 9.2 (SAS Institute Inc, Cary, North Carolina, USA) was used for all data analyses. Because some surgeons provided data at follow-up time points (eg, 6 months, 1 year, etc) without a specific date, a follow-up date was imputed for these patients. Associations between categorical variables were examined using Fisher exact test or χ 2 test. For comparisons of continuous variables between 2 groups, 2-sample t tests were used, and when comparisons of pre/post visual acuity measurements were performed, paired t tests were employed. To analyze the relationship between 2 continuous variables, simple linear regression was performed.
Visual acuity measurements were obtained using a standard Snellen chart viewed from a distance of 6 meters and were converted to logarithm of the minimal angle of resolution (logMAR) units for the analysis, which was the primary outcome of interest. Visual acuity measurements that were recorded as counting fingers were converted to a Snellen equivalent using the conversion algorithm described by Holladay, although a lower limit of 20/2000 was used. When a distance at which finger counting was measured was not recorded, the distance was assumed to be 2 feet, which is equivalent to 20/2000. One research group has calculated that hand motions acuity ranges between 2.28 and 3.60 logMAR units; the upper limit was used for this study.
Eyes with light perception (LP) and no light perception (NLP) visual acuity were excluded in initial analyses of visual acuity, although they were summarized in the figures using the format previously published for Boston keratoprostheses. In addition, the same analysis was performed by assigning Snellen values of 20/40 000 for LP and 20/60 000 for NLP. Results for these analyses are presented only when they differed from the primary approach.
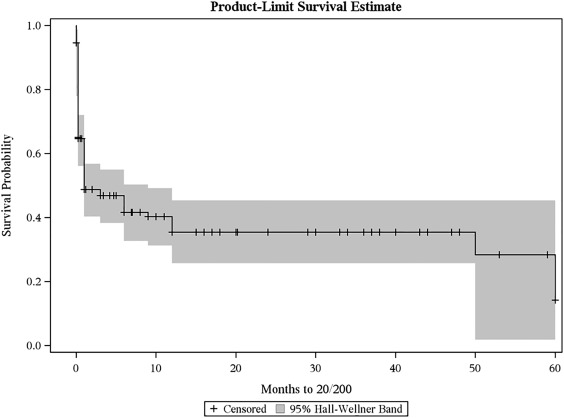
Based on the recommendations by Jabs, the time to achieve 20/200 vision was analyzed using Kaplan-Meier curves with 95% Hall-Wellner Bands and the log-rank test was used for group comparisons. Time to achieve 20/200 visual acuity was defined as from the date of surgery to the first follow-up visit at which 20/200 visual acuity was observed, including preoperatively. As such, eyes with better than 20/200 visual acuity prior to KPro placement were censored at time zero. For those eyes, time to loss of 20/200 visual acuity was analyzed as well. For the latter analysis, fluctuation in vision was discounted such that, for example, an eye that had hand motions vision preoperatively, 20/50 vision at 1 week, 20/200 at 1 month, 20/400 at 6 months, 20/100 at 1 year, and hand motions at 2 years would be recorded as having lost 20/200 vision after 22.77 months. Cox proportional hazards regression analysis, using stepwise selection, was used to determine which surgical indication was significantly associated with the time to achieve 20/200 visual acuity. The Kaplan-Meier curves and cumulative residuals were used to evaluate the proportional hazards assumption.
Pearson correlation coefficients were calculated to evaluate the correlation of final visual acuity with preoperative visual acuity. A scatterplot was created to demonstrate the relationship between preoperative and final logMAR visual acuity levels and linear regression was used to fit a summary line with 95% confidence interval (CI). A multivariate linear regression model, controlling for baseline visual acuity, was fit using stepwise selection to determine which covariates were significantly associated with final visual acuity. Covariates were considered for inclusion in the model if univariate analyses demonstrated a P ≤ .1.
Results
Between January 2003 and July 2008, information on 321 Boston keratoprosthesis type 1 implants placed in 303 patients by 19 surgeons at 18 medical centers was collected. Thirteen eyes were excluded because they represented reimplantation of a keratoprosthesis; only data from the first implant were included. Eight eyes of 8 patients that underwent sequential bilateral keratoprosthesis implantation were also excluded. The final analysis included 300 eyes of 300 patients.
The mean age at the time of implantation was 62.6 ± 18.9 years (range 10.5–96.7 years); age was not associated with final visual acuity outcome ( P = .09). Patients were nearly equally split between sexes, with 48.1% (n = 102) being female; there was no association between final visual acuity and sex ( P = .959). The procedure was performed in the right eye for 53.6% (n = 158) of the cases and there was no relationship to final visual acuity ( P = .167). Overall, 7.0% (21/300) of keratoprostheses failed; our initial analysis with actual visual acuity measurements showed that final visual outcome was not associated with failure ( P = .494). However, 4 eyes that failed had non-numeric visual acuity (LP or NLP) at the final visit. We performed additional analyses assigning Snellen values of 20/40 000 for LP and 20/60 000 for NLP and there was still no association between final visual outcome and failure ( P = .167).
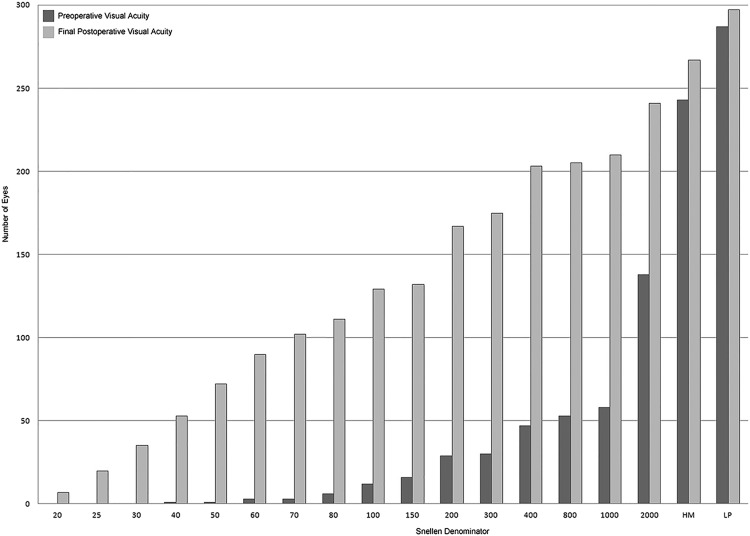
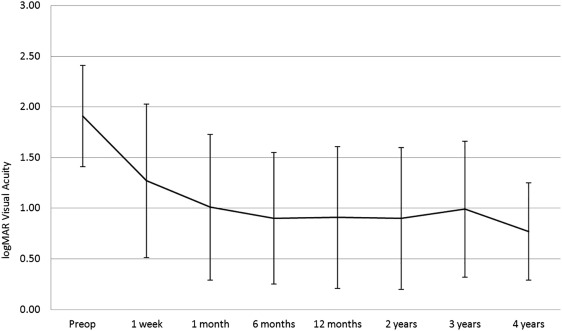
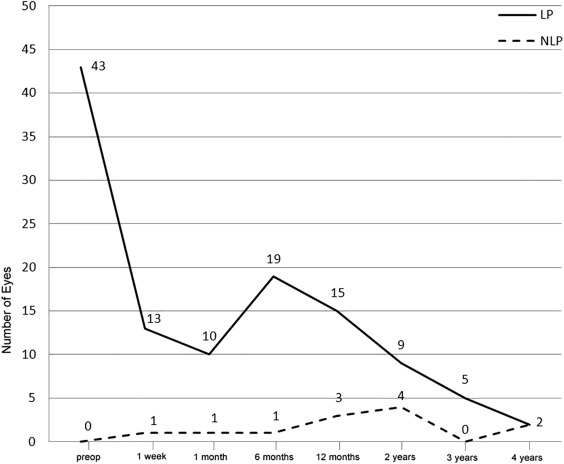
Preoperative visual acuity was recordable on the Snellen chart for 47.3% of eyes (n = 142) and was equivalent to 1.78 ± 0.62 logMAR units (20/1205). Of those eyes that did not have vision measurable using a Snellen chart, 33.3% of eyes (n = 100) had hand motions acuity and 14.3% (n = 43) had only LP. Over the follow-up period, which averaged 17.1 ± 14.8 months and ranged from 1 week to over 6.1 years, visual acuity improved ( Figures 1 and 2 ) significantly ( P < .0001) for 84.7% of eyes (n = 254), to a mean final value of 0.89 ± 0.64 (20/150). At the final visit, there were significantly fewer eyes with hand motions acuity (8.5%; n = 24; P = .263) and light perception (6.7%; n = 19; P < .0001), but 3.1% (n = 9) progressed to no light perception ( Figure 3 ). The mean change in visual acuity was −0.89 ± 0.91 logMAR units, representing a significant improvement in vision.
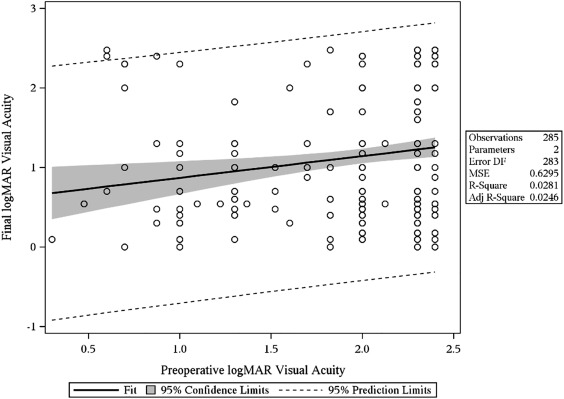
The relationship between preoperative and final logMAR visual acuity is illustrated in Figure 4 ; the Pearson correlation coefficient is 0.13 ( P = .05), indicating the positive correlation between these 2 variables.
The median time to achieve 20/200 visual acuity was 1 month (95% CI 1.0–6.0; Figure 5 ). Of those, the median time to loss of 20/200 visual acuity was 47.8 months ( Figure 6 ). From the Kaplan-Meier curve itself, approximately three-quarters of eyes retained 20/200 acuity for most of the study period. Eyes with a preoperative etiology of multiple failed graft and limbal stem cell deficiency achieved 20/200 faster, with median times of 1.0 ( P = .012; 95% CI 1.0–6.0) and 0.23 ( P = .002; 95% CI 0.00–0.23) months, respectively. Multivariate (Cox) proportional hazards regression analysis confirmed that eyes with limbal stem cell deficiency (hazard ratio [HR] = 3.73; 95% CI 1.81–7.70; P = .0004), multiple failed grafts (HR = 1.98; 95% CI 1.33–2.94; P = .0008), chemical injuries (HR = 1.81; 95% CI 1.16–2.85; P = .0096), and traumatized eyes (HR = 2.27; 95% CI 1.17–4.40; P = .015) experience rapid recovery. It is also noteworthy that no aniridia patients achieved 20/200 vision, which is not surprising given the coexisting macular pathology associated with the condition. For time to loss of 20/200, eyes that received a KPro for limbal stem cell deficiency did the best ( P < .0001), with none losing 20/200 acuity once it was achieved.
There were 3 categories of implant types: primary (no previous PK; 13.3%), repeat (previous keratoprosthesis undergoing replacement; 7.5%) and de novo (previous PK; 79.2%). Of those that had a failed PK prior to keratoprosthesis (n = 244), an average of 2.3 ± 1.3 prior corneal transplants (range 1–8) were performed. There was no difference in final visual acuity by implant category ( P = .621) or in relation to the number of prior failed PK ( P = .567).
The Boston keratoprosthesis type 1 was used for a wide variety of ocular surface diseases ( Table 1 ). All etiologies except aniridia ( P = .08) demonstrated a statistically significant improvement ( P ≤ .04) in visual acuity after keratoprosthesis implantation. The best final visual acuity was observed in (nonaniridic) limbal stem cell deficiency patients, with a mean final logMAR acuity of 0.47 ± 0.30. Chemical injuries (0.55 ± 0.56 logMAR units) and keratoconus (0.68 ± 0.44 logMAR units) were similarly successful.
| Diagnosis | N | Preoperative LogMAR Acuity (± SD) | Final LogMAR Acuity (± SD) | Snellen Equivalent Final Visual Acuity | P Value a |
|---|---|---|---|---|---|
| Autoimmune | 31 | 1.83 ± 0.63 | 1.20 ± 0.76 | 20/317 | <.0001 |
| Chemical injury | 31 | 2.03 ± 0.40 | 0.55 ± 0.56 | 20/71 | .006 |
| HSV | 21 | 2.00 ± 0.44 | 1.14 ± 0.80 | 20/276 | <.0001 |
| Fuchs endothelial dystrophy | 8 | 1.64 ± 0.50 | 1.24 ± 0.79 | 20/348 | .04 |
| Keratoconus | 11 | 1.84 ± 0.47 | 0.68 ± 0.44 | 20/96 | .04 |
| Infectious keratitis | 19 | 1.76 ± 0.67 | 1.04 ± 0.61 | 20/219 | .0006 |
| Neurotrophic keratopathy | 4 | 2.30 ± 0 | 1.21 ± 0.41 | 20/325 | .01 |
| Limbal stem cell deficiency | 9 | 1.76 ± 0.47 | 0.47 ± 0.30 | 20/59 | .0004 |
| Bullous keratopathy | 55 | 1.92 ± 0.46 | 1.14 ± 0.67 | 20/276 | <.0001 |
| Trauma | 13 | 1.83 ± 0.59 | 0.87 ± 0.55 | 20/148 | .0004 |
| Aniridia | 7 | 2.10 ± 0.45 | 1.54 ± 0.48 | 20/693 | .08 |
| Miscellaneous | 35 | 1.94 ± 0.50 | 1.03 ± 0.81 | 20/215 | .0004 |
| Unknown | 6 | 2.06 ± 0.34 | 1.00 ± 1.19 | 20/200 | Undefined b |
| Multiple failed graft | 50 | 1.93 ± 0.49 | 0.97 ± 0.69 | 20/187 | <.0001 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


