Purpose
To evaluate reactivation of pediatric uveitis during/following treatment with tumor necrosis factor alpha inhibition (anti-TNFα).
Design
Retrospective cohort study.
Methods
We assessed the incidence of uveitis reactivation in children ≤18 years who had achieved uveitis quiescence under anti-TNFα. Survival analysis was used to calculate reactivation rates while still on (primary outcome), and following discontinuation of (secondary outcome), anti-TNFα. Potential predictive factors were assessed.
Results
Among 50 children observed to develop quiescence of uveitis under anti-TNFα, 39 met criteria to be “at risk” of the primary (19 for the secondary) outcome. 60% were female, ∼half had juvenile idiopathic arthritis, and most were treated with infliximab. Overall, the estimated proportion relapsing within 12 months was 27.8% (95% confidence interval [CI]: 15.9%–45.8%); the estimated probability of reactivation was higher following (63.8% [95% CI: 38.9%–87.7%]) vs before (21.6% [95% CI: 10.8%–40.2%]) anti-TNFα discontinuation. Among those who discontinued anti-TNFα, the likelihood of reactivation was higher for those treated with adalimumab vs infliximab (hazard ratio [HR] 13.4, P = .01, 95% CI: 2.2–82.5) and those with older age at uveitis onset (HR 1.3, P = .09, 95% CI: 1.0–1.7). The duration of suppression, on medication, did not significantly affect the likelihood of reactivation when quiescence was maintained for ≥1.5 years.
Conclusions
Approximately 75% of children remaining on anti-TNFα following achievement of uveitis quiescence remain quiescent at 1 year. However, most reactivate following anti-TNFα discontinuation. These results suggest that infliximab more often is followed by remission, off medication, than adalimumab. The data do not suggest that maintenance of suppression for more than 1.5 years decreases the reactivation risk.
Uveitis is an important cause of visual loss. The incidence may range from 7/100 000 to 27/100 000 among children and adolescents. In the developed world, the majority of pediatric uveitis is noninfectious, chronic, anterior uveitis, and the preponderance is undifferentiated or associated with juvenile idiopathic arthritis (JIA). In most cases, topical corticosteroids are the first line of treatment. When uveitis is corticosteroid-resistant or requires sufficient corticosteroid therapy to make morbidity likely, conventional immunomodulators, most often methotrexate, are added. If these are inadequate, biologic immunomodulators that block tumor necrosis factor alpha (anti-TNFα) are often the next treatment choice. A number of studies have demonstrated that the majority of corticosteroid- and methotrexate-resistant uveitis improved under anti-TNFα. We previously demonstrated that 75% of children with noninfectious uveitis achieved quiescence within 1 year of treatment with anti-TNFα (infliximab or adalimumab), and that the children with JIA-associated anterior uveitis were most likely to respond. While this study showed that anti-TNFα were useful for uveitis treatment in this setting, it did not address how long we should continue to treat children with anti-TNFα for uveitis.
A common practice among pediatric rheumatologists is to maintain JIA quiescence for 1–2 years before discontinuing systemic medications. Similarly, many physicians who care for patients with pediatric uveitis believe that the risk of reactivation is lowered by a more prolonged period of suppression while on medication (suppression). There are scant, conflicting data on whether a longer duration of suppression results in decreased risk of disease reactivation after withdrawal for either JIA or JIA-associated uveitis. All but 1 of these studies focused on reactivation following conventional immunomodulator withdrawal. Whether this is the case in uveitis controlled under anti-TNFα has not been directly examined.
To provide guidance regarding management of children with uveitis while under treatment with anti-TNFα, we provide additional follow-up of patients from our previously published cohort. Here, we report estimates of the risk of uveitis reactivation while patients were still on, and after they had discontinued, anti-TNFα. In addition, we examine potential risk factors for reactivation of uveitis after anti-TNFα discontinuation.
Patients and Methods
Study Setting and Study Population
This was a retrospective cohort study of patients managed in the Division of Rheumatology of The Children’s Hospital of Philadelphia (CHOP) between January 2000 and July 2012 inclusive. This study includes expanded data collection for patients previously described and patients who began care from July 1, 2009 to July 31, 2012. Subjects were identified by searching the electronic medical record (EMR) for ICD-9 codes possibly indicating noninfectious uveitis (ICD-9 363.x, 364.x). The charts of patients thus identified were reviewed to determine if each patient had noninfectious nontraumatic uveitis (uveitis), if ophthalmologic records were available, and if the patient had been treated with an anti-TNFα agent. The study was undertaken after the governing institutional review board (The Children’s Hospital of Philadelphia) granted approval with waiver of informed consent for this retrospective study, which did not involve any contact with patients. HIPAA-compliant procedures were used for the research described herein.
Inclusion Criteria
Subjects selected for this analysis had achieved inactive uveitis following treatment with anti-TNFα medications and had ophthalmologic records available for review after achieving quiescence. Inclusion criteria for analysis of achievement initial of quiescence were described previously. Briefly, quiescence was defined as achieving either “slightly active” or “inactive” uveitis while on ≤2 drops/day topical corticosteroids and no oral corticosteroids sustained for ≥2 visits spanning ≥28 days. In that analysis, if a patient discontinued medication because of failure or adverse events, a second drug episode could be included per patient, but second rounds of anti-TNFα therapy following initially successful treatment were not included. For this analysis, patients could meet criteria for inactive disease while being on ≤2 drops/day of topical corticosteroids. Infliximab and adalimumab were the only anti-TNFα therapies observed. Concomitant treatment with corticosteroids, methotrexate (MTX), and/or mycophenolate mofetil was recorded.
Data Collection
Data regarding demographics, treatment, and disease activity were obtained from electronic medical or paper records through the CHOP Division of Rheumatology, the CHOP Department of Ophthalmology, and community ophthalmologist reports available in these records. Data were entered into a custom electronic Microsoft Access 2007 (Microsoft Corporation, Redmond, Washington, USA) database. JIA patients were characterized further into subtypes according to the International League of Associations for Rheumatology criteria. Age at uveitis was dichotomized (≤6 vs >6 years), following the American Academy of Pediatrics ophthalmologic screening guidelines for children with JIA.
Definition of Disease Activity and Location
Disease activity at each visit was characterized as an ordered categorical outcome, using the approach the Systemic Immunosuppressive Therapy for Eye Disease Cohort Study has used previously: inactive, slightly active, or active, incorporating information from sites of inflammation including, but not limited to, the anterior chamber (AC) into a single activity variable (including AC cell, vitreous cells, vitreous haze). Categories closely parallel the uveitis definitions of the Standardization of Uveitis Nomenclature (SUN) Working Group. Chart reviewers ascertained disease status from a combination of the physician’s overall assessment at the visit through the use of descriptors such as quiet or quiescent, as well as quantitative descriptors of cell grade and vitreous haze. “Inactive” reflected designations such as no cells, rare cells, no vitreous haze, and no corticosteroids; “slightly active” reflected gradings such as trace or fewer AC cells (≤0.5 + ) and minimal vitreous haze or cells; and “active” reflected higher levels of inflammation. Disease state was recorded by eye; a patient’s disease state was considered as the worse level of the 2 eyes if both had uveitis, or else the level of the single eye with uveitis. Uveitis location was categorized as anterior, intermediate (±anterior), posterior, or panuveitis.
Outcome Definition
The outcome was reactivation, defined as observation of “active” uveitis after achievement of quiescence. The incidence of reactivation was calculated following achievement of quiescence while still on treatment (primary analysis) and following complete discontinuation of anti-TNFα (secondary analysis). When uveitis was inactive, it was termed “suppression” when a patient remained on anti-TNFα and “remission” following discontinuation of anti-TNFα.
Follow-up Time
For the primary analysis, follow-up time began with the first visit at which “inactive uveitis” was identified; time to reactivation was measured to the first visit at which “active” uveitis reoccurred. Data were not censored when medication was discontinued. For the secondary analysis (time to reactivation after stopping anti-TNFα), follow-up time began on the date of the clinic visit at which medication discontinuation was advised. Uveitis information was collected from all available visits to an ophthalmologist, but visit frequency varied in this retrospective, observational study. Follow-up was completed when reactivation occurred, when the subject was lost to follow-up without redeveloping “active” uveitis (censoring), or at the end of the study (administrative censoring).
Covariates
Demographic and clinical characteristics (summarized in Table 1 ) were assessed for their relationship to incidence of reactivation of uveitis while continuing anti-TNFα or after discontinuing anti-TNFα therapy. Severity of disease activity was analyzed with a surrogate marker, use of <4 vs ≥4 drops of topical corticosteriods daily.
| Characteristic | Whole Cohort a (n = 39 b , PY = 64.4) | Subset Continuing on Anti-TNFα (n = 20 b , PY = 32.1) | Subset Discontinuing Anti-TNFα (n = 19, PY = 13.2) |
|---|---|---|---|
| Sex (% female) | 59.0 | 60.0 | 57.9 |
| Race (% white) | 64.1 | 70.0 | 57.9 |
| Age at uveitis diagnosis (% >6 years) c | 61.6 | 64.7 | 58.8 |
| Median age at uveitis diagnosis (range) c | 6.9 (1.4–16.3) | 7.4 (2.0–16.3) | 6.25 (1.4–13.1) |
| Systemic diagnosis (% JIA) | 43.6 | 40.0 | 47.4 |
| Uveitis location (% anterior) | 80.1 | 80.0 | 84.2 |
| Anti-TNFα (% infliximab) d | 89.7 | 90.5 | 84.2 |
| Median years from disease diagnosis to initiation of anti-TNFα (IQR) c | 0.6 (0.2, 2.1) | 0.9 (0.3, 2.0) | 0.3 (0.2, 2.1) |
| Median years to achieve uveitis quiescence on anti-TNFα (IQR) | 0.2 (0.1, 0.3) | 0.2 (0.1, 0.3) | 0.2 (0.1, 0.3) |
| Median years uveitis quiescence on anti-TNFα to discontinuation (IQR) | — | — | 1.9 (0.6, 2.4) |
a The cohort was followed until either reactivation or the end of follow-up data, regardless of whether discontinued anti-TNFα.
b One patient with 2 drug episodes, eg, 39 patients but 40 drug episodes.
c Four cases were missing age of uveitis diagnosis (available, n = 35).
d Percentage of treatment courses, not patients (n = 40 for whole cohort).
Data Analysis
Data were analyzed using Stata 11.0 (Stata Corp, College Station, Texas, USA). Differences in categorical demographic and clinical characteristics between subcohorts were assessed using the χ 2 test. Nominal statistical significance was defined as a 2-tailed P value ≤.05. Kaplan-Meier methods evaluated time to reactivation (failure) after observation of quiescence. As a subject could have more than 1 anti-TNFα treatment episode, analyses were performed using a robust variance estimator to account for clustering by subject (1 subject had 2 treatment episodes). Cox proportional hazards model–derived hazard ratios (HR) estimated the association of each independent patient-level or treatment-level variable with each outcome variable. Being on additional immunomodulatory therapy varied during treatment (eg, being on one at quiescence did not ensure continued treatment at reactivation); as this was a time-varying covariate, associations with other variables were analyzed with χ 2 testing rather than through regression analysis.
Results
Among 50 children and adolescents observed to develop quiescence of uveitis under anti-TNFα treatment during the period of observation, encompassing 53 drug episodes (ie, 3 were treated twice), 39 subjects—observed over 40 drug episodes—met inclusion criteria for assessment of the primary outcome of time to reactivation of uveitis while under anti-TNFα treatment (64.4 patient-years at risk). Nineteen subjects discontinued anti-TNFα following achievement of quiescence and met inclusion criteria for the secondary outcome of time to reactivation of uveitis after cessation of anti-TNFα therapy, covering 13.2 patient-years at risk.
Demographic and Treatment Characteristics of Subjects who Achieved Quiescence
Of the 39 subjects who achieved quiescence, 60.0% were female, 64.1% were white, and 61.6% were diagnosed with uveitis at older than 6 years of age ( Table 1 ). Less than half the cohort, 43.6% of subjects, had JIA; the remainder had sarcoidosis (10%) or uveitis without a known systemic association (46%). The majority had anterior uveitis (n = 32, 82.1%), 1 had intermediate uveitis (2.6%), and 6 had posterior uveitis (15.4%). Of those with sarcoidosis, 3 had anterior uveitis and 1 panuveitis. Treatment with anti-TNFα was initiated within 1 year of uveitis onset in 62.9% of the subjects, and 87.5% achieved quiescence within a half a year of starting medication. The majority of patients were treated with infliximab, although 5 patients were treated with adalimumab (1 of whom switched to adalimumab after having an adverse reaction to infliximab). While a greater percentage of patients treated with adalimumab had an underlying diagnosis of JIA (80%) compared to those treated with infliximab (40%), there was no statistically significant association between underlying diagnosis (JIA vs other) and the anti-TNFα used ( P = .09) (data not shown). Nor was there a statistically significant difference in the type of uveitis children treated with infliximab had relative to those treated with adalimumab (anterior uveitis: 79% vs 100%, P = .53) or the underlying disease activity, as reflected by ≥4 drops topical corticosteroids/day, at the onset (more severe uveitis 60% vs 40%, P = .4) (data not shown).
Characteristics of Subjects who Continued on Anti-TNFα Relative to Those who Discontinued
The demographic characteristics of those who continued on and discontinued anti-TNFα were similar: 60.0% vs 57.9% were female ( P = .8), 70.0% vs 57.9% were white ( P = .4), 64.7% vs 58.8% ( P = .9) were diagnosed with uveitis after 6 years of age, and 80.0% vs 84.2% ( P = .4) had anterior uveitis ( Table 1 ). Almost half the cohort, 40.0% vs 47.4%, had JIA ( P = .6). Treatment with anti-TNFα was initiated within 1 year in 38.9% vs 35.3% of the subjects ( P = .9, data not shown); 90.5% vs 84.2% were treated with infliximab ( P = .6) and the remainder with adalimumab. Of those who discontinued anti-TNFα, two thirds of subjects (68.4%) were on anti-TNFα for more than 1 year after achieving quiescence, but only one third were on anti-TNFα for more than 2 years after achieving quiescence (36.8%). The median time on anti-TNFα from achievement of quiescence to discontinuation was 1.73 years (interquartile range [IQR]: 0.25–2.15). The median total time on anti-TNFα was 2.26 years (IQR: 1.45–3.17) in those who discontinued the drug relative to 1.24 (IQR 0.78–2.38) in those who remained on anti-TNFα ( P = .16). By the time of disease reactivation or censoring, 47% (56% infliximab, 0% adalimumab) of the cohort who discontinued anti-TNFα remained on either traditional immunomodulators (methotrexate [n = 7] or mycophenolate mofetil [n = 2]).
Incidence of Uveitis Reactivation
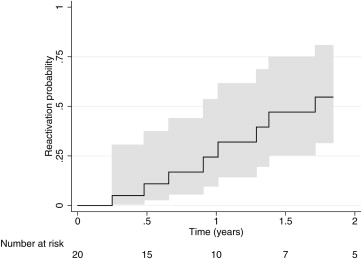
Among the 39 subjects at risk of the primary outcome, the estimated proportion of those in whom uveitis reactivated within 12 months of quiescence was 27.8% (95% confidence interval [CI]: 15.9%–45.8%) (data not shown). The estimated probability of a uveitis reactivation was 2.5% by 3 months (95% CI: 0%–16.8%), 18.4% by 6 months (95% CI: 9.2%–34.9%), and 21.3% by 9 months (95% CI 11.2%–38.1%). The median time to failure was not observed; the estimated median time to failure was 23.5 months.
In a sensitivity analysis of the failure rate among only those who continued on anti-TNFα, the estimated probability of a uveitis reactivation per subject by 12 months was 24.4% (95% CI 9.7%, 53.5%) ( Figure 1 ) and the estimated median time to failure was 20.5 months (32.1 patient-years).