Purpose
To characterize the clinical spectrum of class 1 and class 2 uveal melanomas and their relationship with intraocular proton radiation response.
Design
Masked retrospective case series of uveal melanoma patients with fine needle biopsy–based molecular profiles.
Methods
A total of 197 uveal melanoma patients from a single institution were analyzed for pathology, clinical characteristics, and response to radiation therapy.
Results
A total of 126 patients (64%) had class 1 tumors and 71 (36%) had class 2 tumors. Patients with class 2 tumors had more advanced age (mean: 64 years vs 57 years; P = .001), had thicker initial mean ultrasound measurements (7.4 mm vs 5.9 mm; P = .0007), and were more likely to have epithelioid or mixed cells on cytopathology (66% vs 38%; P = .0004). Although mean pretreatment and posttreatment ultrasound thicknesses were significantly different between class 1 and class 2 tumors, there was no difference in the mean change in thickness 24 months after radiation therapy (mean difference: class 1 = −1.64 mm, class 2 = −1.47; P = .47) or in the overall rate of thickness change (slope: P = .64). Class 2 tumors were more likely to metastasize and cause death than class 1 tumors (DSS: P < .0001).
Conclusions
At the time of radiation therapy, thicker tumors, epithelioid pathology, and older patient age are significantly related to class 2 tumors, and class 2 tumors result in higher tumor-related mortality. We found no definitive clinical marker for differentiating class 1 and class 2 tumors.
Many uveal melanoma parameters are related to an increased risk of metastatic death. Genomic changes, most importantly loss of a single copy of chromosome 3, are associated with an increased risk of metastasis. Gene expression profiling of uveal melanomas has revealed 2 distinct classes of tumors that accurately predict prognosis. Class 1 tumors are unlikely to metastasize, while class 2 tumors are more likely to metastasize and cause death. The genetic signature of class 1 and class 2 tumors can be assessed from a traditional biopsy or from a fine needle aspiration biopsy (FNAB). This gene expression profiling has been shown to have superior predictive accuracy when compared to the analysis using fluorescence in situ hybridization or array comparative genomic hybridization looking only at monosomy 3.
The goal of this study was to better characterize the clinical spectrum of class 1 and class 2 uveal melanomas and its possible relationship with intraocular proton radiation response.
Methods
This study was a retrospective case series of patients with uveal melanoma treated with proton beam radiation therapy between November 13, 1997 and November 12, 2010, with the last follow-up in January 2011. All patients were recruited from a single institution. Patients who did not receive proton radiation, because of primary enucleation or iridocyclectomy without radiation, were excluded from the analysis (n = 25). The study examined the clinical characteristics and radiation response of 197 evaluable uveal melanoma patients. Each patient underwent FNAB (185) or tumor resection with biopsy (12), followed by proton radiation. The tissues from each case underwent molecular studies and were delineated into class 1 and class 2 tumors based on the genetic characteristics at 15 loci. The specifics of this gene expression profiling have been previously discussed. In addition, cytologic or histopathologic examination was performed to determine the predominant tumor cell type. Evaluation of the tissue was performed masked to all patient, disease, and outcome features.
Clinical data and fluorescein angiographic and ultrasound characteristics were obtained from patients’ initial visits. For each case, information obtained from clinical records and photographs included sex, age, ocular location, tumor area, and presence of orange pigmentation. Ultrasound data collected included tumor thickness, the presence of a collar button, and the presence of subretinal fluid.
To assess if an in-depth characterization of fluorescein angiographic or ultrasonographic characteristics might be related to molecular class, a subset of 36 irradiated patients with posterior class 1 or class 2 choroidal melanomas were selected based on tumor size and location (posterior to the equator). For these patients, additional ultrasound data (homogeneity, orbital shadowing, shape of posterior A-wave spike, and internal reflectivity) and fluorescein angiographic features (degree of blockage and leakage, presence of hot spots, tumor coloration, and the presence of intrinsic tumor vascularity) were assessed by 2 observers masked to all other clinical features and disease outcomes (M.C., D.H.C.).
Patients treated with proton beam radiation therapy had their follow-up clinical, photographic, and ultrasound thickness measurements gathered at each postoperative appointment. These data were then used to compare changes over time after radiation therapy.
Comparability between molecular classes in baseline disease and patient features was determined using Fisher exact test for categorical variables and a 2-group t test for continuous variables. Overall results were summarized using a logistic regression model to identify independent predictors of molecular class. Variables considered as potential predictors were those found to be related to molecular class univariately with a probability of less than 0.10 ( Table ). A likelihood ratio (LLR) test was used with a forward stepwise approach with a probability to be included in the model of .05 and a probability to be removed of .10. Linear regression analysis was performed to estimate the rate of change in tumor thickness over time using all follow-up measurements for each patient. The overall rate of change was summarized by the slope from this analysis. In addition, the estimate of change 24 months after proton therapy, the velocity, was calculated by the difference in tumor thickness at approximately 24 months from the pretreatment measurement divided by the follow-up time interval. A t statistic was used to compare the 2 molecular classes in terms of the velocity at 24 months, the mean change from baseline in tumor thickness to 24 months after therapy, and the mean rate of change using the slope for each patient.
| Class 1 (n = 126) | Class 2 (n = 71) | P Value | |
|---|---|---|---|
| Mean age at RT (y) | 57.2 | 64.1 | .001 |
| Range | 20.5–94.2 | 17.3–91.6 | |
| Number male (%) | 65 (52%) | 39 (55%) | .66 |
| Eyewall resection/irido, n (%) | 8 (6%) | 8 (11%) | .28 |
| Enucleation (post-RT), n (%) | 14 (11%) | 11 (15%) | .38 |
| FNAB, n (%) | 120 (95%) | 65 (92%) | .36 |
| Tumor location, n (%) | |||
| Crosses equator | 110 (89%) | 59 (83%) | .28 |
| Posterior to equator | 74 (60%) | 35 (49%) | .18 |
| Mean tumor area, mm | 61.2 | 68.8 | .25 |
| Range | 7.2–179.7 | 4.9–192.2 | |
| Ultrasound thickness, mm | 5.9 | 7.4 | .0007 |
| Range | 2.0–13.7 | 3.2–13.8 | |
| Number with OP (%) | 20 (16%) | 12 (17%) | 1.00 |
| Number with drusen (%) | 17 (14%) | 6 (8%) | .36 |
| Number with collar button (%) | 24 (19%) | 19 (27%) | .28 |
| Number with SRF (%) | 65 (52%) | 35 (50%) | .88 |
| Tumor cell type, n (%) | |||
| Spindle | 74 (62%) | 23 (34%) | |
| Epithelioid/mixed | 45 (38%) | 44 (66%) | .0004 |
The durations of time to metastasis and disease-specific survival (DSS) were each measured from the start of proton beam radiation therapy. Failure for DSS was the date of death attributable to metastatic disease. Patients who were alive were censored at the date of last follow-up. Univariate analyses using the Cox proportional hazards model were carried out to determine which factors were predictors of time to metastasis and DSS. Statistical significance was analyzed using the LLR test. Variables found to be predictive of outcome with a probability value of less than .10 univariately were considered in multivariate analyses, again using the Cox proportional hazards model to identify independent predictors of time to metastases and DSS. A forward stepwise approach was used to build a model with a probability of .05 to include a variable and a probability of .10 to remove a variable in the model. Independent predictors were determined by a probability value of less than .05 using the LLR test, with the results summarized by the hazard ratio (HR). The Kaplan-Meier product limit method was used to graphically display the results and estimate 5-year time to metastases and DSS outcomes.
Results
There were 197 evaluable patients with uveal melanoma defined by molecular classification treated with proton radiation between November 1997 and November 2010. One hundred and twenty-six (64%) had class 1 tumors and 71 (36%) had class 2 tumors. One hundred and four patients (53%) were male and 93 patients (47%) were female. The mean age of patients at the time of proton radiation therapy was significantly less in patients with class 1 tumors (57 years old) as compared to patients with class 2 tumors (64 years old) ( P = .001). The mean tumor area for class 1 tumors (61.2 mm) was smaller than for class 2 tumors (68.8 mm). This difference was not statistically significant ( P = .25). Mean pretreatment ultrasound thickness was greater for class 2 patients (7.4 mm) as compared to class 1 patients (5.9 mm) ( P < .001) ( Table ).
There was no statistically significant difference between the 2 classes of tumors based on the presence of orange pigment ( P = 1.00), drusen ( P = .36), a collar button ( P = .28), or subretinal fluid ( P = .88). Analysis of gene profile and tumor cell type showed that a significantly greater number of tumors in class 1 were spindle cell (62%) as compared to class 2 (34%) ( P = .0004) ( Table ).
A logistic regression model was developed to identify independent predictors of molecular class. The same 3 factors identified with the univariate analyses were found to be independent predictors of molecular classification. Using a LLR test the 3 variables were significant predictors in the following order. Having a class 2 tumor reflected an epithelioid cell type ( P = .0003), being older at the time of radiotherapy ( P = .002), and increased tumor thickness on ultrasound pretreatment ( P = .03). Tumor location entirely posterior to the equator was not an independent predictor of molecular class.
Of the 197 patients treated with proton radiation, 187 had follow-up information available after radiation treatment (118 class 1, 69 class 2). There was no difference in the mean number of follow-up measurements between the 2 classes ( P = .65). Overall, the median duration of disease follow-up for irradiated patients was 21.7 months (range 2.0-111.9 months) with no difference between the 2 classes (medians: class 1 = 25.7 months [2.6-111.9 months], class 2 = 18.0 months [2.0-94.3 months]) ( P = .23). Univariate analyses were performed using the Cox proportional hazards model to evaluate factors predictive of time to metastases and DSS. The results indicated that molecular class ( P < .0001), cell type ( P = .05), and age of the patient at the time of radiation therapy ( P = .04) were significantly predictive of time to metastases, whereas pretreatment ultrasound measurement ( P = .11) was not.
Molecular class ( P < .0001) was predictive of DSS with univariate analysis, while cell type ( P = .24), age of the patient at time of radiation therapy ( P = .056), and pretreatment ultrasound measurement ( P = .21) were not. Multivariate analysis using the Cox proportional hazards model was performed to identify independent predictors of time to metastases and DSS. Variables found to be significant predictors univariately with a probability of less than .10 were considered as potential predictors. Only molecular class was an independent significant predictor of outcome for remaining metastasis-free (HR = 8.4; LLR: P < .0001) and DSS (HR = 12.3; LLR: P < .0001).
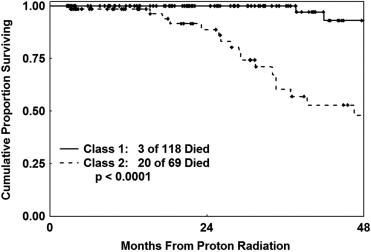
As of this analysis, 30 of the irradiated patients developed metastatic disease after proton radiation therapy, 6 of the class 1 patients and 24 of the class 2 patients (LLR test: P < .0001). Twenty-three patients died as a result of their metastatic melanoma, 3 patients with class 1 melanomas and 20 patients with class 2 melanomas (LLR test: P < .0001) (DSS: Figure 1 ) . One of the class 1 patients whose disease metastasized (and who died of metastatic melanoma) was later reclassified as class 1B. It is unknown whether the other 5 class 1 patients were class 1A or 1B. At 5 years, the Kaplan-Meier estimate of the probability of metastatic death from uveal melanoma was 62% (95% CI: 44%-80%) for class 2 patients as compared to 7% (95% CI: 2%-25%) for class 1 patients.