Purpose
To assess for activation of the unfolded protein response in corneal endothelium of Fuchs endothelial corneal dystrophy patients.
Design
Retrospective, comparative case series of laboratory specimens.
Methods
Corneal specimens of patients with Fuchs dystrophy and controls with corneal pathologic features other than Fuchs dystrophy were evaluated by transmission electron microscopy (TEM) to evaluate for structural changes of the rough endoplasmic reticulum in corneal endothelium. TEM images were evaluated for alterations of rough endoplasmic reticulum as a sign of unfolded protein response. Normal autopsy eyes, Fuchs dystrophy corneas, and keratoconus corneas were used for immunohistochemistry. Immunohistochemistry was performed on formalin-fixed, paraffin-embedded sections of patient corneas for 3 unfolded protein response markers (GRP78, the α subunit of eukaryotic initiation factor 2, C/EBP homologous protein) and 2 apoptosis markers (caspase 3 and 9). Immunohistochemistry signal quantitation of corneal endothelium for evaluation of marker expression was performed using automated software. Corneal sections were assessed quantitatively for levels of immunohistochemistry marker expression.
Results
TEM showed enlargement of rough endoplasmic reticulum in corneal endothelium of all Fuchs dystrophy specimens. Immunohistochemistry quantitation demonstrated a significant increase in mean signal in corneal endothelium from Fuchs dystrophy patients for markers GRP78, the α subunit of eukaryotic initiation factor 2, C/EBP homologous protein, and caspase 9 compared with non-Fuchs dystrophy corneas ( P < .05).
Conclusions
Results of both TEM and immunohistochemistry indicate activation of unfolded protein response in Fuchs dystrophy. Unfolded protein response activation leads to endothelial cell apoptosis in Fuchs dystrophy and may play a central pathogenic role in this disease.
Fuchs endothelial corneal dystrophy (Fuchs dystrophy) is a primary disease of the corneal endothelium characterized by loss of endothelial cells and abnormalities of Descemet membrane. These pathologic changes lead progressively to corneal edema and loss of vision, occurring approximately 20 years after disease onset. Fuchs dystrophy recently has been characterized as having an early-onset form, with diagnosis as early as the first decade of life, and a late-onset form with diagnosis typically between the third and fourth decades of life. Previous studies of Fuchs dystrophy pathogenesis indicate increased endothelial cell apoptosis. However, early disease stages are asymptomatic, and studies of Fuchs dystrophy tissues are limited to failed patient corneas undergoing corneal transplant surgery. Thus, the cellular pathophysiologic features of Fuchs dystrophy remain poorly understood.
Protein folding is critical for cellular functioning, and all cells have mechanisms to ensure proper protein folding and disposal of irreversibly misfolded proteins. Accumulation of misfolded proteins can result in endoplasmic reticulum stress, a condition that is toxic to cells. To counteract this stress, cells initiate the unfolded protein response, which is a comprehensive program to reduce the accumulation of toxic unfolded proteins. Endoplasmic reticulum stress and unfolded protein response have been shown to play important roles in the pathogenesis of multiple human diseases, including diabetes, Alzheimer and Parkinson diseases, and atherosclerosis. In addition, endoplasmic reticulum stress and unfolded protein response have been implicated in a variety of ophthalmologic diseases, including cataract, retinitis pigmentosa, and glaucoma.
The unfolded protein response consists of 3 effector arms mediated by 3 endoplasmic reticulum transmembrane receptors: pancreatic endoplasmic reticulum kinase–like endoplasmic reticulum kinase, activating transcription factor 6, and inositol requiring enzyme 1. These effectors are maintained in an inactive state by binding to the intraluminal endoplasmic reticulum chaperone protein GRP78. Additional important markers of unfolded protein response activation include the α subunit of eukaryotic initiation factor 2 (phospho-eIF2α), which is phosphorylated by pancreatic endoplasmic reticulum kinase–like endoplasmic reticulum kinase under endoplasmic reticulum stress conditions and C/EBP homologous protein (CHOP). Levels of GRP78, phospho-eIF2α, and CHOP are increased in cells undergoing unfolded protein response. Failure to alleviate endoplasmic reticulum stress by unfolded protein response can lead to cellular apoptosis. The complex molecular mechanisms involved in the apoptotic death of endoplasmic reticulum–stressed cells have not been elucidated fully. However, downstream effectors of unfolded protein response–mediated apoptosis in humans may include caspase 4, caspase 3, and caspase 9 ( Figure 1 ).
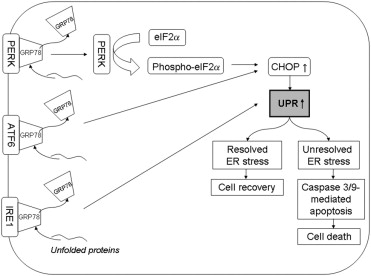
We hypothesized that unfolded protein response is activated in Fuchs dystrophy by the abundant production of misfolded protein, as indicated by thickening of Descemet membrane and the accumulation of wide-spaced collagen aggregates, which are hallmark findings in Fuchs dystrophy. We tested this hypothesis by electron microscopic assessment of endoplasmic reticulum in Fuchs dystrophy patients to evaluate for structural evidence of unfolded protein response activation. In addition, we performed immunohistochemistry of Fuchs dystrophy corneal endothelium for 3 unfolded protein response markers (GRP78, phospho-eIF2α, and CHOP) and 2 apoptosis markers (caspase 3 and 9).
Methods
Patients and Controls
The diagnosis of Fuchs dystrophy and keratoconus was made by fellowship-trained corneal specialists. For Fuchs dystrophy specimens, diagnosis was based on the presence of confluent endothelial guttae in both eyes and presence of corneal edema (stromal, epithelial, or both) in the operative eye. Diagnosis of keratoconus was based on corneal topography along with the presence of standard clinical signs. Corneas from autopsy cases with no history of corneal disease or pathologic corneas with diagnoses other than Fuchs dystrophy served as controls. Patient and control characteristics are summarized in Table 1 (TEM experiments) and Table 2 (immunohistochemistry).
| Prominent RER +/− | Diagnosis | |
|---|---|---|
| Controls | ||
| 1 | − | Granular dystrophy |
| 2 | − | Lattice dystrophy |
| 3 | + | Macular dystrophy |
| 4 | +/− | Lattice dystrophy |
| 5 | − | Macular dystrophy |
| 6 | − | Lattice dystrophy |
| 7 | − | Macular dystrophy |
| 8 | +/− | Granular dystrophy |
| 9 | − | Granular dystrophy |
| Fuchs dystrophy | ||
| 1 | + | Fuchs dystrophy |
| 2 | + | Fuchs dystrophy |
| 3 | + | Fuchs dystrophy |
| 4 | + | Fuchs dystrophy |
| 5 | + | Fuchs dystrophy |
| 6 | + | Fuchs dystrophy |
| 7 | + | Fuchs dystrophy |
| 8 | + | Fuchs dystrophy |
| 9 | + | Fuchs dystrophy |
| 10 | + | Fuchs dystrophy |
| Autopsy Corneas | |||||
|---|---|---|---|---|---|
| Age (yrs) | Cause of Death | Corneal Findings | Gender | Death to Preservation Time (hours) | |
| 19 | Cystic fibrosis | Normal | Male | 16 | |
| 54 | Larynx carcinoma | Normal | Female | 11 | |
| 70 | Liver cirrhosis | Normal | Male | 44 | |
| 92 | Aspiration pneumonia | Normal | Male | 49 | |
| 58 | Human immunodeficiency virus | Normal | Male | 13 | |
| 65 | Pneumonia | Normal | Female | 58 | |
| 59 | Adenocarcinoma gall bladder | Normal | Female | 15 | |
| 53 | Multisystem organ failure | Normal | Male | 21 | |
| 6 | Cerebral infarctions | Normal | Male | 23 | |
| 63 | Metastatic carcinoma | Normal | Male | 52 | |
| 75 | Aneurysm rupture | Normal | Female | 53 | |
| 80 | Aspiration pneumonia | Normal | Male | 46 | |
| 47 | Lung cancer | Normal | Male | 59 | |
| 60 | Necrotizing pancreatitis | Normal | Male | 7 | |
| 45 | Pulmonary failure, liver transplantation rejection | Normal | Male | 63 | |
| 51 | Metastatic melanoma | Normal | Male | 24 | |
| 71 | Adenocarcinoma colon | Normal | Male | 30 | |
| 67 | Gastrointestinal hemorrhage | Normal | Male | 43 | |
| Mean | 58 | 35 | |||
| SD | 20 | 19 | |||
| Fuchs Dystrophy Corneas | ||||
|---|---|---|---|---|
| Age (yrs) | Clinical Diagnosis | Pathologic Diagnosis | Gender | |
| 38 | Fuchs dystrophy | Fuchs dystrophy | Female | |
| 60 | Fuchs dystrophy | Fuchs dystrophy | Male | |
| 89 | Fuchs dystrophy | Fuchs dystrophy | Female | |
| 68 | Fuchs dystrophy | Fuchs dystrophy | Male | |
| 34 | Pseudophakic bullous keratopathy | Fuchs dystrophy | Male | |
| 68 | Fuchs dystrophy | Fuchs dystrophy | Male | |
| 74 | Fuchs dystrophy | Fuchs dystrophy | Female | |
| 67 | Fuchs dystrophy | Fuchs dystrophy | Male | |
| Mean | 62 | |||
| SD | 18 | |||
| Keratoconus Corneas | ||||
|---|---|---|---|---|
| Age (yrs) | Clinical Diagnosis | Pathologic Diagnosis | Gender | |
| 16 | Keratoconus | Keratoconus | Male | |
| 34 | Keratoconus | Keratoconus | Female | |
| 18 | Keratoconus | Keratoconus | Female | |
| 15 | Keratoconus | Keratoconus | Male | |
| 14 | Keratoconus | Keratoconus | Male | |
| Mean | 19 | |||
| SD | 8 | |||
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


