Purpose
To determine treatment outcomes of intravitreal ocriplasmin in symptomatic vitreomacular adhesion.
Design
Retrospective interventional case series.
Methods
setting : Private practice. study population : Thirty-five eyes (35 subjects) with symptomatic vitreomacular adhesion. intervention/observation : Intravitreal ocriplasmin injection from February to November 2013. main outcome measure : Vitreomacular adhesion resolution rate. Secondary endpoints included postinjection visual acuity and rates of outer retinal attenuation and full-thickness macular hole (MH) closure.
Results
The 35 subjects included were of mean age 69 years, 66% female, and 71% phakic. Eleven subjects (31%) had macular comorbidities. Average adhesion diameter was 571 μm, with mean 7.9 months duration of symptoms. Nine subjects (26%) had epiretinal membrane and 6 (17%) had MH (mean diameter 186 μm). Mean preinjection logarithm of the minimal angle of resolution visual acuity was 0.46, and improved to 0.33 at final follow-up. Fifteen eyes (43%) achieved adhesion release at mean 10 days post injection. One of 6 MH (17%) closed. Transient outer retinal attenuation occurred in 10 of 35 cases (29%), with 8 of 10 (80%) achieving adhesion release. One subject (3%) developed a retinal detachment. Adhesion resolution was more likely in patients with younger age ( P = .04), absence of comorbidities ( P = .02), small adhesion diameter ( P = .005), short adhesion duration ( P = .03), and transient outer retinal attenuation ( P = .008).
Conclusions
Intravitreal ocriplasmin efficacy in the private practice setting, while including patients with macular comorbidities, is similar to that of previous studies. Transient toxicity to the outer retina occurs frequently—typically with adhesion resolution—necessitating careful postinjection spectral-domain optical coherence tomography monitoring.
Anomalous posterior vitreous detachment (PVD) is a pathologic variant of typical PVD that occurs when the vitreoretinal interface does not adequately weaken, particularly at sites of strong adhesion, including the optic nerve, macula, and vitreous base. Vitreomacular adhesion is defined as the persistence of cortical vitreous macular attachment within 3 millimeters of the foveal center during surrounding perifoveal vitreous cortex separation from the retinal surface without a detectable alteration in foveal contour or the underlying retina clinically and on optical coherence tomography (OCT). Vitreomacular adhesion may be asymptomatic or symptomatic if associated with a functional or visual disturbance. Associated distortion of the foveal surface or intraretinal structure or elevation of the fovea above the retinal pigment epithelium (RPE) distinguishes vitreomacular traction (VMT) from vitreomacular adhesion. VMT may have features of an impending or evolved full-thickness macular hole (MH). The International Vitreomacular Traction Study (IVTS) Group further defined vitreomacular adhesion and VMT as focal (<1500 μm in diameter) or broad (≥1500 μm in diameter), and as isolated or concurrent (with coexisting macular comorbidities such as age-related macular degeneration [AMD] or diabetic retinopathy [DR]). Until now, surgical management with pars plana vitrectomy (PPV) and observation have traditionally been the only management options for vitreomacular interface disorders. Currently, symptomatic vitreomacular adhesion and VMT are indications for a new nonsurgical treatment modality to induce PVD—pharmacologic vitreolysis—and avoid the potential complications and limitations of vitrectomy.
Ocriplasmin (Jetrea; ThromboGenics, Inc, Iselin, New Jersey, USA) is a recombinant truncated protease subunit of human plasmin that can induce vitreous liquefaction and has proteolytic activity at the vitreoretinal interface. More specifically, it hydrolyzes the peptide bonds of laminin, fibronectin, and collagen—molecules that maintain the extracellular matrix adhesion between the posterior hyaloid and the internal limiting membrane (ILM). The Microplasmin for Intravitreous Injection—Traction Release without Surgical Treatment (MIVI-TRUST) study groups, composed of 2 large randomized, controlled, double-masked, multicenter, phase III clinical trials (TG-MV-006 and TG-MV-007), evaluated the safety and efficacy of ocriplasmin in the setting of symptomatic vitreomacular adhesion. These data subsequently led to approval by the US Food and Drug Administration (FDA) in October 2012 for management of symptomatic vitreomacular adhesion, and in Europe for vitreomacular traction with or without associated full-thickness MH of ≤400 μm. The phase III studies found that a single 125 μg intravitreal dose of ocriplasmin induced vitreomacular separation at a statistically higher rate than in placebo-injected eyes (26.5% vs 10.1%) and also induced full-thickness MH closure in 40.6% of treatment eyes compared to 10.6% of placebo.
Both the structural and functional outcomes of drugs in postmarketing clinical use sometimes differ from results obtained during carefully controlled clinical trials. Therefore, it is important to examine the behavior of eyes treated with ocriplasmin in a nontrial setting. The MIVI-TRUST studies used time-domain OCT and excluded patients with concurrent vitreomacular adhesion. In this report, we describe our experience with intravitreal ocriplasmin in 35 cases of both isolated and concurrent symptomatic vitreomacular adhesion in a private practice setting, report adhesion release rate—our primary outcome, and offer comprehensive analysis of factors predictive of adhesion release. Secondarily, we report our visual outcomes and additional details related to the nature of adhesion release. Structural analysis using spectral-domain optical coherence tomography (SD OCT) was performed for all cases.
Methods
This investigation is an Inova Health System institutional review board–approved retrospective case review adherent to the tenets of the Declaration of Helsinki and compliant with the Health Insurance Portability and Accountability Act. Thirty-five consecutive patients were included in this study, each of whom underwent comprehensive evaluation, including extensive history, ocular examination, and SD OCT, and received a single intravitreal injection of 125 μg ocriplasmin by 1 of 15 participating vitreoretinal specialists at the Retina Group of Washington, a multicenter tertiary referral vitreoretinal clinical practice, between February 22, 2013 and November 22, 2013. All patients with symptomatic vitreomacular adhesion or VMT who underwent ocriplasmin injection, at the judgment of each retinal specialist, were included; no injected patients were excluded. These subjects were identified from an electronic database.
Data extracted from pre- and postinjection visits included patient demographic information, lens status, medical and ocular comorbidities, Snellen best-corrected visual acuity (BCVA), vitreomacular adhesion duration prior to injection, presence of PVD, time to vitreomacular adhesion resolution, postinjection adverse events, surgical intervention, and total follow-up. Snellen BCVA was converted to logarithm of the minimal angle of resolution (logMAR) to evaluate vision changes following injection. All patients underwent evaluation with SD OCT at each visit with either the Cirrus (Carl Zeiss Meditec, Dublin, California, USA) or Spectralis (Heidelberg Engineering, Heidelberg, Germany) system. Information gathered from these scans, via each OCT system’s proprietary software, included vitreomacular adhesion size, presence and size of MH, epiretinal membrane (ERM), cystoid macular edema (CME), vitreomacular adhesion release, and changes in the photoreceptor outer segments, known as the ellipsoid zone, after injection. All patients with Cirrus OCT imaging underwent both 5-line Raster scan and Macular Cube scan, and both scans were used for careful pre- and postinjection structural analysis. Specifically, the Macular Cube scans, with a radius of 3 mm from the foveal center, were used to identify and measure the diameter of the vitreomacular adhesion at its broadest point. Patients who underwent Spectralis OCT had a 15-slice horizontal scan. Vitreomacular adhesion maximal diameter within 3 mm of the foveal center was independently found and measured in all cases using the OCT caliper function in an unmasked fashion by 2 examiners (D.J.W. and J.R.) per the IVTS Group, subsequently averaged and used for statistical analysis. Analogously, careful assessment of each postinjection Macular Cube scan was performed to search for ellipsoid zone changes. MH diameter was measured at the aperture—the narrowest hole point or minimum hole width in the mid-retina—as described by the IVTS Group.
The study’s primary result was the rate of vitreomacular adhesion release. Secondary outcomes included timing of adhesion resolution and ellipsoid zone attenuation, BCVA at final follow-up visit, and rates of complete PVD induction, MH closure, ellipsoid zone attenuation, other adverse events, and surgical intervention.
Statistical analysis was performed using Statistical Analysis Software (SAS, Cary, North Carolina, USA). Fisher exact test 2-sided probability was used to compare individual categorical independent variables, and Satterthwaite t test probability of unequal variances compared individual continuous independent variables. Statistical significance was set at P < .05 for all analyses.
Results
Thirty-five consecutive patients were injected with intravitreal ocriplasmin by 1 of 15 different retina specialists within the Retina Group of Washington at 12 different offices. Baseline characteristics of the subject population are summarized in Table 1 . Regarding specific details of patients with concurrent VMA, 4 subjects had baseline intermediate dry AMD and 1 patient had controlled wet AMD on a monthly aflibercept regimen. Five subjects had mild to moderate nonproliferative diabetic retinopathy (NPDR). Three patients had diabetic macular edema (DME); all 3 had prior focal laser treatment in the eye that received ocriplasmin injection; 2 patients had prior anti–vascular endothelial growth factor (anti-VEGF) injection—1 patient nearly a year prior, and the second patient had monthly ranibizumab injections up until 4 weeks prior to ocriplasmin. One patient had a history of branch retinal vein occlusion (BRVO), and 1 patient had adult-onset vitelliform foveomacular dystrophy.
| Mean age (y) | 69.4 |
| No. of female subjects (%) | 23 (65.7) |
| Phakic (%) | 25 (71.4) |
| Retinal comorbidities present (%) | 11 (31.4) |
| Mean logMAR BCVA (Snellen equivalent) | 0.46 (20/57) |
| Mean vitreomacular adhesion duration (mo) | 7.9 |
| Mean vitreomacular adhesion diameter (μm) | 571 |
| MH present (%) | 6 (17.1) |
| Mean MH aperture diameter (μm) | 186 |
| CME present (%) | 25 (71.4) |
| ERM present (%) | 6 (17.1) |
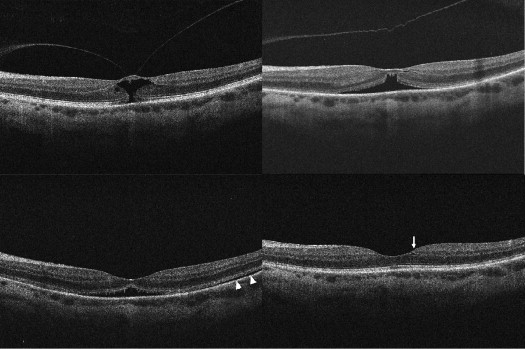
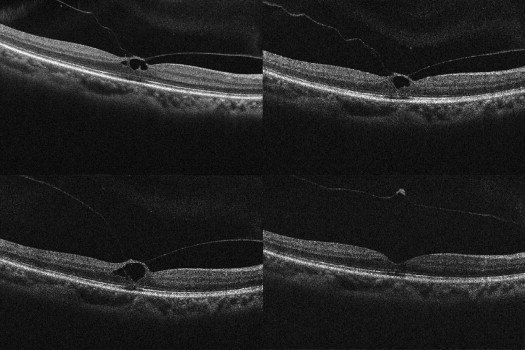
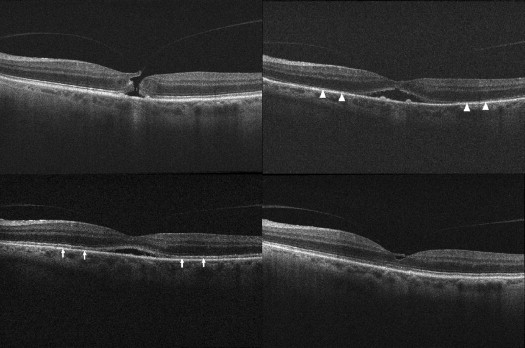
Complete resolution of vitreomacular adhesion ( Figures 1 and 2 ) was noted in 15 subjects (42.9%), with associated complete PVD induction in 12 (34.3%). Mean time to adhesion resolution was 10.2 days (range 1–46). Full-thickness MH closure occurred in 1 of 6 cases (16.7%), at postinjection day 6; while this subject’s MH closed, the vitreomacular adhesion persisted ( Figure 3 ). Conversely, 3 patients with full-thickness MH (50%) had adhesion release, but each had a persistent MH ( Figure 4 ).
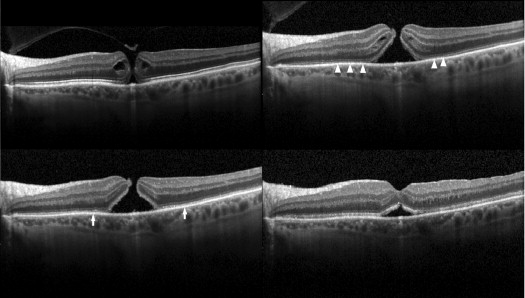
Baseline clinical characteristics of subjects who experienced complete resolution of vitreomacular adhesion, and of those who had persistent vitreomacular adhesion after ocriplasmin injection, are summarized in Table 2 and Table 3 , respectively. Statistical analysis of these baseline characteristics is depicted in Table 4 . Vitreomacular adhesion resolution was more likely to occur in eyes with smaller adhesion diameter ( P < .005), shorter adhesion duration ( P < .05), younger age ( P < .05), and absence of retinal comorbidities ( P < .05) ( Figures 5 and 6 ). Better preinjection BCVA, female sex, and absence of ERM also favored vitreomacular adhesion resolution, although the difference did not reach statistical significance ( Table 4 ) using Fisher exact test 2-sided probability and Satterthwaite t test probability of unequal variances for categorical and continuous analysis, respectively.
| Age | Sex | Lens Status | Initial BCVA | LogMAR | MH | CME | ERM | Retinal Comorbidity | Vitreomacular Adhesion | |
|---|---|---|---|---|---|---|---|---|---|---|
| Size (μm) | Duration (mo) | |||||||||
| 66 | F | Phakic | 20/30 | 0.176 | + | 378 | 1 | |||
| 60 | F | Phakic | 20/20 | 0 | + | 702 | 1.5 | |||
| 59 | F | Phakic | 20/200 | 1 | + | + | + | 192 | 0.5 | |
| 58 | F | Phakic | 20/20 | 0 | + | 384 | 25 | |||
| 66 | M | Phakic | 20/30 | 0.176 | + | 483 | 5 | |||
| 80 | F | Phakic | 20/40 | 0.301 | + | 156 | 9 | |||
| 51 | F | Phakic | 20/40 | 0.301 | + | 261 | 1 | |||
| 67 | M | Phakic | 20/100 | 0.699 | + | + | 542 | 1 | ||
| 71 | F | Phakic | 20/70 | 0.544 | + | 366 | 6 | |||
| 68 | F | PCIOL | 20/80 | 0.602 | + | + | 296 | 1 | ||
| 69 | M | Phakic | 20/20 | 0 | 121 | 6 | ||||
| 59 | F | Phakic | 20/30 | 0.176 | + | 332 | 5 | |||
| 71 | F | PCIOL | 20/70 | 0.544 | + | + | 407 | 3 | ||
| 69 | F | Phakic | 20/100 | 0.699 | 242 | 1.5 | ||||
| 74 | F | PCIOL | 20/40 | 0.301 | + | 321 | 3 | |||
| Age (y) | Sex | Lens Status | Initial BCVA | LogMAR | MH | CME | ERM | Retinal Comorbidity | Vitreomacular Adhesion | |
|---|---|---|---|---|---|---|---|---|---|---|
| Size (μm) | Duration (mo) | |||||||||
| 64 | F | Phakic | 20/25 | 0.099 | + | + | 729 | 1.5 | ||
| 73 | F | Phakic | 20/50 | 0.398 | + | 713 | 4 | |||
| 84 | M | Phakic | 20/200 | 1 | + | 582 | 24 | |||
| 62 | F | Phakic | 20/20 | 0 | + | 446 | 16 | |||
| 53 | F | Phakic | 20/400 | 1.301 | + | 220 | 3 | |||
| 56 | F | Phakic | 20/50 | 0.398 | 515 | 8 | ||||
| 77 | M | PCIOL | 20/60 | 0.477 | + | + | 1712 | 1.5 | ||
| 77 | F | Phakic | 20/40 | 0.301 | + | + | 449 | 2 | ||
| 80 | M | PCIOL | 20/40 | 0.301 | + | + | 1316 | 29 | ||
| 80 | F | PCIOL | 20/70 | 0.544 | + | + | 895 | 1 | ||
| 65 | F | Phakic | 20/80 | 0.602 | + | + | 314 | 18 | ||
| 69 | F | Phakic | 20/100 | 0.699 | + | 656 | 2 | |||
| 76 | M | Phakic | 20/200 | 1 | + | 392 | 5.5 | |||
| 65 | F | PCIOL | 20/80 | 0.602 | + | + | 1355 | 7 | ||
| 70 | F | Phakic | 20/25 | 0.099 | + | + | + | 979 | 11 | |
| 85 | M | PCIOL | 20/40 | 0.301 | + | 74 | 20 | |||
| 90 | M | PCIOL | 20/50 | 0.398 | + | + | 416 | 17 | ||
| 77 | M | Phakic | 20/50 | 0.398 | + | 117 | 14 | |||
| 73 | M | PCIOL | 20/40 | 0.301 | + | + | 680 | 21 | ||
| 66 | M | Phakic | 20/400 | 1.301 | + | + | + | + | 2035 | 2 |
| Resolution | Persistence | P Value | |
|---|---|---|---|
| Mean age (y) | 65.9 | 72.1 | .04 a |
| No. of female subjects (%) | 12 (80) | 11 (55) | .16 |
| Phakic (%) | 12 (80) | 13 (65) | .46 |
| Mean logMAR BCVA (Snellen equivalent) | 0.368 (20/47) | 0.526 (20/67) | .17 |
| MH present (%) | 3 (20) | 3 (15) | 1.0 |
| CME present (%) | 12 (80) | 13 (65) | .46 |
| ERM present (%) | 2 (13) | 7 (35) | .24 |
| Retinal comorbidity present (%) | 1 (7) | 10 (50) | .02 a |
| Mean vitreomacular adhesion size (μm) | 346 | 730 | .005 b |
| Mean vitreomacular adhesion duration (mo) | 4.6 | 10.4 | .03 a |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


