Purpose
To test if the drug FK962 ( N -(1-acetylpiperidin-4-yl)-4-fluorobenzamide) facilitates axonal elongation and recovery of corneal sensitivity after creation of a corneal flap in rabbits.
Design
Animal study.
Methods
Primary cultures of rabbit trigeminal ganglion cells were used to test if FK962 promoted nerve elongation in vitro. A 130 μm-thick × 8.6 mm-diameter flap was created in rabbit corneas where topical 10 −6 M FK962 was administered 4 times daily. After treatment of 7 days, corneal mechanical sensitivity was measured using a Cochet-Bonnet esthesiometer. Whole-mount corneal sections were prepared, sensory nerve axons were stained with antibody for neurofilament, and axonal elongation from transected nerve termini were scored using standardized criteria. Ocular pharmacokinetics modeling was used to predict permeation of topical FK962 into cornea.
Results
FK962 accelerated sprouting and elongation of neurites in cultured neuronal cells from rabbit trigeminal ganglia. In the in vivo rabbit model, distal axons from transected nerve termini in corneas disappeared soon after flap surgery; but with time, axons regenerated and elongated. Topical application of 10 −6 M FK962 for 7 days significantly enhanced axonal elongation and increased corneal sensitivity. Increased corneal sensitivity was directly and significantly correlated with axonal elongation, suggesting functional enhancement of re-innervation by FK962.
Conclusions
Results from a rabbit model of laser in situ keratomileusis (LASIK) surgery showed that topical FK962 facilitated corneal re-innervation leading to recovery of sensitivity. Results suggested that topical application of FK962 might decrease complications in patients after LASIK surgery.
Laser in situ keratomileusis (LASIK) is a well-accepted surgical procedure for correcting myopia. However, sensory nerves in the sub-basal (Bowman’s layer) and superficial stromal regions of the cornea are amputated during flap creation, leading to nerve degeneration and decreased corneal sensitivity. The density of sub-basal nerves is significantly decreased 1 month after LASIK surgery, then gradually returns; but the nerves were still abnormal after 6 months. These corneal nerves are involved in feedback loop stimulation of tear secretion via the sympathetic and parasympathetic nerves innervating the lacrimal gland. Decreased corneal sensitivity can thus lead to complications such as dry eye and neurotrophic epitheliopathy.
The cornea/lacrimal gland feedback loop suggests that regeneration of corneal nerves by growth factors would help restore corneal sensitivity and be useful in therapy for dry eye. Indeed, pituitary adenylate cyclase–activating polypeptide (PACAP) induced outgrowth of neuronal processes in trigeminal ganglion cells and accelerated recovery of corneal sensitivity in a rabbit model of LASIK surgery. Nerve growth factor (NGF) has been well studied and causes neuronal outgrowth in several cell types, including trigeminal neurons. NGF is also known to be released following LASIK surgery and promotes corneal wound healing. FK962 ( N -[1-acetylpiperidin-4-yl]- 4-fluorobenzamide) enhanced secretion of somatostatin from rat hippocampal slices, and somatostatin induced neurite outgrowth in rat cerebellar granule cells. In cultured rat astrocytes, an analog of FK962 caused production of glial cell line–derived neurotrophic factor (GDNF), a promoter of cell survival and axonal regeneration in a wide variety of neuronal populations. The purpose of the present experiments was therefore to determine if FK962 facilitates axonal regeneration and recovery of corneal sensitivity in an in vivo model of LASIK surgery in rabbits.
Methods
Experimental Animals
Male Japanese white rabbits (Kitayama Labes, Nagano, Japan) at 4 days of age were the source of trigeminal ganglia for cell culture experiments, 9- to 10-week-old male rabbits provided corneas for an in vitro trans-corneal study, and 11- to 12-week-old male rabbits were used in an in vivo model of flap creation as described in a previous report.
Culture of Trigeminal Ganglion Cells
Trigeminal ganglia were isolated using a modification of a method previously reported. Minced tissues from isolated trigeminal nerves were digested for 30 minutes with 3 mg/mL collagenase A (Roche Diagnostics, Basel, Switzerland) followed by incubation for 40 minutes at 37 C in Nerve-Cell Culture System/Dissociation Solutions (Sumitomo Bakelite, Tokyo, Japan). Following filtration through a 40-μm cell strainer (Becton-Dickinson, Franklin Lakes, New Jersey, USA), cells were prepared according to the manufacturer’s protocol. Cells (1 × 10 3 ) were cultured in 8-well culture dishes coated with poly-D-lysine and laminin (Becton-Dickinson) in neurobasal medium (Invitrogen/Life Technologies, Carlsbad, California, USA) supplemented with 1 mM L-glutamine (Invitrogen/Life Technologies), 0.2% primocin (Invitrogen), and 10 μM cytosine β-D-arabinofuranoside (Wako, Osaka, Japan). FK962, kindly provided by Astellas Pharma Inc (Tokyo, Japan), was used at 10 −8 to 10 −10 M.
Immunocytochemistry and Quantification of Elongation in Neuronal Cells
Cultured cells were fixed for 20 minutes in 2% paraformaldehyde, washed, incubated for 30 minutes in blocking solution containing 5% goat serum (Cosmo bio, Tokyo, Japan) and 0.1% Triton X-100, and incubated for 1 hour with neurofilament 200 mouse monoclonal antibody (clone N52; Sigma, St. Louis, Missouri, USA) at 1:1000 dilution. Cells were visualized by incubation for 1 hour with goat anti-mouse IgG conjugated to Alexa Fluor-568 (Invitrogen) at 1:1000 dilution. The digital images of cells were captured with a fluorescence microscope (IX71; Olympus, Tokyo, Japan). Quantitative assessment of neurite elongation was performed by cell counting using Image-Pro Plus ver. 4.5 (Media Cybernetics, Bethesda, Maryland, USA). A cell with an elongated neurite was defined as a cell body immunopositive for neurofilament and with a process extending to a length ≥ twice the diameter of the cell body. Neurofilament-positive cells in each well were counted in twelve 883 × 665-μm steady fields and totaled. Data were expressed as percentage of cells with elongated neurites compared to total neurofilament-positive cells.
Transcorneal Delivery of FK962 and Simulation of FK962 Uptake Into Cornea
For in vitro transcorneal permeation studies, the corneal epithelium from 1 eye of each pair with flap surgery was removed with a surgical knife, and intact and de-epithelialized corneas were mounted side-by-side in an Ussing diffusion chamber maintained at 34 C. Testing solution containing 0.1% FK962 was added to 1 side of the chamber (donor cell), while buffer solution was added to the other side of the chamber (receptor cell), separated by the cornea. At predetermined time intervals, 200 μL of receptor solution was collected and replaced with fresh buffer solution. FK962 in the receptor solution was measured at 225 nm after separation by high-performance liquid chromatography (HPLC) using a 4.6-mm-inner-diameter × 250-mm-long Inertsil ODS-3 column (GL Sciences, Tokyo, Japan) with 35% methanol in 5 mM disodium hydrogen phosphate/12-water (pH 6.0). Permeation profiles for FK962 across corneas with flaps were constructed for both intact and de-epithelialized preparations by plotting the accumulation of permeated FK962 with time. Permeation rates and time lags were calculated by using a pharmacokinetics model based on Fick’s second law of diffusion. Diffusion and partition coefficients for corneal epithelium and stroma were calculated based on the bilayer membrane model. These constants were used in ocular pharmacokinetics modeling to predict FK962 permeation into corneal epithelium and stroma, in preparation for topical application of FK962 in the in vivo experiments below.
Rabbit Model of Flap Surgery
A corneal flap was created in the right eye of anesthetized rabbits from the nasal side at the center of the cornea using an automated corneal shaper microkeratome (Nidek, Aichi, Japan) with a planning thickness of 130 μm and a diameter of 8.6 mm. The corneal flap was returned in place without sutures under a stereoscopic microscope. Gatifloxacin antibiotic eye drops (0.3%, GATIFLO; Senju, Osaka, Japan) were administered 4 times a day for 6 days after surgery. Rabbits with initial corneal sensitivity of more than 15 mm in the right eye were selected 1 day before surgery. One day after surgery, rabbits with sensitivity less than 5 mm of filament were selected (n = 31). Based on corneal sensitivity before surgery, the rabbits were then divided into 2 groups using stratified randomization, resulting in mean initial sensitivities shown in Table 1 . The treated group received one topical 50-μL drop containing 10 −6 M FK962 in 6.4 mM phosphate-buffered saline (PBS, pH 7.0) in the right eye 4 times a day for 7 days, while the vehicle group received PBS.
| Sensitivity (mm Filament) | ||||
|---|---|---|---|---|
| Group | N | Day 0 (Before LASIK) | Day 1 (Start Treatment) | Day 8 |
| Vehicle (mean ± SD) | 15 | 28.3 ± 6.7 | 0 | 2.0 ± 5.0 |
| FK962 (mean ± SD) | 16 | 28.1 ± 7.3 | 0 | 4.7 ± 3.7 |
| P a | >.1 | .050 | ||
Corneal Sensitivity Measurements
Corneal sensitivity was measured at the center of the cornea with a Cochet-Bonnet esthesiometer (Luneau, Paris, France) under a double-masked procedure 1 day before and 1 and 8 days after surgery. The diameter of the nylon filament was 0.12 mm, and the length could be varied from 60 mm (maximum sensitivity) to 5 mm (minimum); lower than 5 mm was recorded as 0 sensitivity.
Since use of the esthesiometer is limited because rabbits are poor blinkers and generally not cooperative, the following procedures were used to eliminate false positives attributable to excited animals blinking simply because of an approaching instrument. 1) Each animal was gently held in a custom-made restraint box and placed in a quiet room (noise of 30-40 dB) during measurement. 2) If an excited animal moved his head during measurement, the measurements were restarted after sedation. 3) Rabbits with half-closed eyes were eliminated. 4) The upper and lower cilia were trimmed off, and hair debris on the corneal surface and nictitating membrane were gently removed using forceps. 5) A positive response was recorded if the eyelid reflex closed immediately after touching the nylon filament perpendicularly to the center of the corneal surface and if ≥5 reflexes occurred in 10 consecutive touches. Each sensitivity test was repeated 3 times, advancing in 5-mm steps. Two positive responses in the 3 attempts at each filament length were regarded as a positive result. The longest filament length causing a positive result was considered the threshold of sensitivity for that cornea. These procedures produced reliable data, but were so labor-intensive that experiments were limited to 7 days.
Immunohistochemical Staining of Corneal Nerves
Corneoscleral buttons were obtained from globes after treatment with FK962 or vehicle for 7 days. To allow penetration of antibodies into cornea, previous methods were modified. Corneal endothelium was scraped off, and the button was immersed in ice-cold saline containing 5 mM EDTA for 1 hour to hydrate the stromal layer. The button was then fixed overnight in Zamboni’s solution containing 4% paraformaldehyde and 0.1% picric acid in 0.1 M phosphate buffer, pH 7.4. The button received four 30-minute rinses in 70% ethanol at 4 C, followed by incubation overnight at −30 C in dimethyl sulfoxide (DMSO)/methanol (1:4). After rinsing 2 times for 30 minutes each with 100% methanol, the button was subjected to 4 cycles of freezing and thawing between −80 C and room temperature to break up the plasma membrane. The button was rehydrated in 70%, 50%, and 15% methanol and phosphate buffer for 30 minutes each. The sclera was removed from the button and 4 radial incisions were made in the remaining cornea. Incised tissues were immersed for 30 minutes in phosphate buffer containing 30% sucrose, followed by OCT compound (Sakura Finetech, Tokyo, Japan) for 3 minutes and flat-mounting by freezing. The Descemet membrane was removed with a freezing microtome, and the corneal tissue was thawed in PBS.
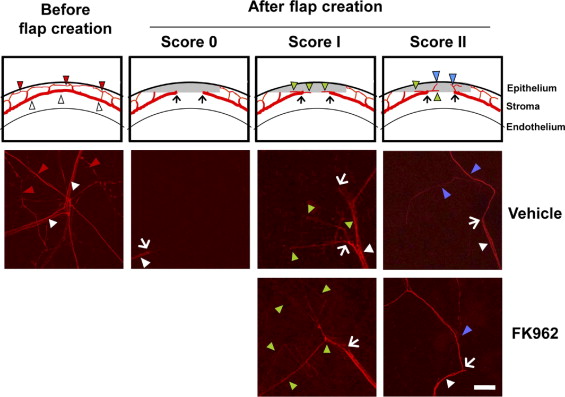
The cornea was free-rotated overnight at 4 C in PBS blocking solution containing 2% skim milk, 5% DMSO, and 0.1% Triton X-100 and then incubated for 2 days with primary monoclonal antibody for neurofilament 200 at 1:1000 dilution. After three 90-minute washes in blocking solution, the tissue was incubated overnight with goat anti-mouse IgG conjugated to Alexa Fluor 488 or 568 (Invitrogen) at 1:200 dilution. After three 90-minute washes in PBS containing 5% DMSO, the tissue was immersed in Vectashield (Vector, Burlingame, California, USA) and flat-mounted with the epithelium side down on glass-bottom culture dishes. Images of nerve axons were observed using a laser scanning confocal microscope (LSM710; Carl Zeiss, Hallbergmoos, Germany) and digitized with ZEN 2000 software. Axonal regeneration was assessed in a 2-mm-diameter circular area at the center of the cornea, where corneal sensitivity had been measured, and was scored using defined criteria as follows ( Figure 1 ): score 0, no nerve fibers or only transected nerve termini; score I, horizontal axonal elongation along the flap-bed interface (green arrowheads); score II, horizontal and vertical axonal elongation into the flap (blue arrowheads).
Normal corneas were also stained for nerves and Schwann cells. Anti–substance P (SP) primary monoclonal antibody (1:200 dilution, clone NC1; Chemicon, Temecula, California, USA) and goat anti-rat IgG conjugated to Alexa Fluor 568 (1:200 dilution, Invitrogen) were used to visualize Aδ and C fibers. Primary monoclonal antibody conjugated to Cy3 (1:200 dilution, clone G-A-5; Sigma) without Zamboni’s fixative stained for glial fibrillary acidic protein (GFAP) in Schwann cells. Washing buffer containing 5 μg/mL DAPI (Dojindo, Tokyo, Japan) was then used to stain for nuclei before flat-mounting the immunostained tissues.
Statistical Analysis
Statistical analyses were performed using software (SAS ver. 9.1 or JMP ver. 8.0; SAS Institute, Cary, North Carolina, USA). The normality of the data distribution was tested using the Shapiro-Wilk test, with the cut-off for non-normality set at P < .05, and then in the case of normal distribution, the Dunnett test was performed for analyzing cell numbers/neurite elongation. Corneal sensitivity and nerve morphologic scoring were analyzed using a 1-tailed nonparametric Wilcoxon rank sum test. Spearman rank correlation coefficient was used to determine direct correlation between morphologic scores and corneal sensitivity.
Results
Sensory Nerve Fibers in Rabbit Cornea
Trigeminal ganglion contains sensory neurons classified as myelinated Aα, Aβ, and Aδ fibers; and as unmyelinated C fibers. The innervation of cornea in rabbits by Aδ and C neurons has been demonstrated by electrophysiology, but not by immunohistochemistry, and the fibers originate from the ophthalmic division of the trigeminal ganglion. Further, almost all of the A-type neurons were found to stain by anti-NF200 antibody and some Aδ and C neurons were stained by anti-SP antibody located in the dorsal root ganglion. To assess axonal elongation after flap surgery in our rabbit study, we therefore first established the immunohistochemical staining for corneal Aδ and C fibers in normal cornea. We found that nerve fibers in normal corneas from rabbits showed 3 staining patterns: NF200-positive/SP-negative Aδ fibers (green, after image merging, Figure 2 , Top); NF200-positive/SP-positive, possible Aδ fibers (yellow after merging); and NF200-negative/SP-positive fibers, possible C fibers (red after merging). NF200-positive nerves were also found to be ensheathed by myelinating GFAP-positive Schwann cells (red, Figure 2 , Bottom). These data suggested that NF200 was the better marker for detection of Aδ fibers, which are believed to be a main mechano-sensory fiber in cornea.
FK962 Enhanced Neurite Elongation In Vitro
Primary rabbit trigeminal ganglion cells staining positive for neurofilament (red) were round and without sprouting after 10 hours of culture, in both control ( Figure 3 , Top, control row) and FK962-treated groups ( Figure 3 , Top, 10 −9 M FK962 row). At 12 hours, control cells started sprouting neurites ( Figure 3 , Top, control row, arrowhead), and treatment with FK962 enhanced neurite elongation ( Figure 3 , Top, 10 −9 M FK962 row, arrow). At 17 hours, the number of cells with elongated neurites was further increased by FK962. Cell counting produced a bell-shaped dose response ( Figure 3 , Bottom), showing that FK962 at 10 −10 and 10 −9 M significantly increased the percentage of cells with elongated neurites compared to controls. The higher dose at 10 −8 M did not enhance elongation. These histologic observations and cell quantification data established that FK962 stimulated neurite elongation in vitro. They also suggested that effective dosing levels and protocols used for in vivo treatment would need to cause accumulation of 10 −10 to 10 −9 M levels of FK962 within the intact corneal layers.

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


