Purpose
To determine the sensitivity of the Bielschowsky head-tilt test and other commonly used criteria in identifying patients with true bilateral superior oblique paresis.
Design
A retrospective chart review was performed to identify patients seen between 1978 and 2009 who were diagnosed with acquired bilateral superior oblique paresis.
Methods
All patients had a confirmed history of head trauma or brain surgery with altered consciousness followed by symptomatic diplopia. Bilateral superior oblique paresis was defined and diagnosed by the above history, including the presence of greater extorsion in downgaze than upgaze on Lancaster red-green testing, a V-pattern strabismus, and bilateral fundus extorsion. We analyzed findings of the Bielschowsky head-tilt test, the Parks 3-step test, and reversal of the hypertropia from straight-ahead gaze to the other 8 diagnostic positions of gaze to determine these tests’ sensitivity in identifying true bilateral superior oblique paresis.
Results
Twenty-five patients were identified with the diagnosis of true bilateral superior oblique paresis. The Bielschowsky head-tilt test had a 40% sensitivity, the Parks 3-step test had a sensitivity of 24%, and reversal of the hypertropia had a sensitivity of 60% in making the diagnosis of true bilateral superior oblique paresis.
Conclusions
What previously has been described as masked bilateral superior oblique paresis simply may be a reflection of inherent poor sensitivity of the Bielschowsky head-tilt test, the Parks 3-step test, and reversal of the hypertropia in diagnosing bilateral superior oblique paresis. Hence, none of these tests should be relied on exclusively to make this diagnosis.
In patients with hypertropia in straight-ahead gaze, Bielschowsky described characteristic changes in the hypertropia with head tilt as a means of diagnosing paresis of the superior oblique muscle. Parks later described his 3-step test that used this phenomenon in a broader sense to help the clinician identify which paretic cyclovertical muscle could cause such a hyperdeviation. Over time, however, limitations of the Bielschowsky head-tilt test as a diagnostic tool have become apparent. Kushner has explored various scenarios elegantly in which the 3-step test, when relied on exclusively, can suggest other forms of strabismus, thus indicating the 3-step test’s lack of specificity. The limitations of the 3-step test in differentiating bilateral superior oblique paresis from a unilateral paresis also have been described.
The concept of masked bilateral superior oblique palsy has been applied to patients having surgery for unilateral superior oblique paresis where postoperatively apparent paresis of the fellow superior oblique muscle develops, or is unmasked. Explanations for masked bilateral superior oblique palsy include asymmetry of the paresis before surgery, or, as described by Saunders and Roberts and by Ellis and associates, surgical overcorrection of the unilateral palsy can masquerade as an apparent contralateral superior oblique paresis.
In our practice, the senior author uses the Lancaster red-green (RG) test to evaluate all patients with vertical strabismus whenever possible. This test provides dissociated measurements of ocular misalignment in 9 standardized positions of gaze, providing more complete data for establishing the diagnosis of superior oblique paresis: the subjective horizontal, vertical, and torsional deviations of both eyes. The primary purpose of our study was to identify patients with acquired, bilateral superior oblique muscle paresis after a head injury, using the pattern of deviation on the Lancaster RG test as the standard for diagnosing bilateral superior oblique paresis in these patients, and to examine the sensitivity of the Bielschowsky head-tilt test in identifying these patients. We also explored the sensitivity of other previously described criteria for such diagnosis. We did not have magnetic resonance imaging scans of these patients, and therefore we were not able to investigate their usefulness in establishing or confirming bilateral disease via atrophy of the superior oblique muscles.
Methods
The Johns Hopkins Medicine Institutional Review Board approved the study protocol and agreed to a waiver of informed consent for use in this retrospective, single-center study. The study and data collection were in accordance with the Health Insurance Portability and Accountability Act of 1996. We performed a retrospective chart review of the medical records of all patients who were seen by the senior author at the Krieger Children’s Eye Center at the Wilmer Eye Institute from 1978 through 2009. Records of interest were identified by a search of the division’s clinical database, the Wilmer Information System.
Inclusion and Exclusion Criteria
Patients were included if they had a diagnosis of bilateral superior oblique palsy or paresis and had undergone at least 1 preoperative Lancaster RG test and a documented preoperative Bielschowsky head-tilt test. The diagnosis required (1) the presence of V-pattern esotropia or exotropia, (2) bilateral underaction of the superior oblique muscles on duction or version testing, (3) objective bilateral fundus extorsion, and (4) subjective extorsion that was greater in downgaze (in the field of action of the superior oblique muscles) than in upgaze on the Lancaster RG plot. The V pattern was based on either the pattern on the Lancaster RG plot or on measurements from prism and alternate cover testing. A difference of 5 prism diopters (PD) or more between upgaze and downgaze on the prism and alternate cover test, although not clinically significant, was considered significant by us if it was supported by the presence of a V pattern on the Lancaster RG plot.
The fifth criterion for inclusion was a documented history of significant head trauma (or surgery for intracranial malignancy) with altered conciousness. (The fourth cranial nerve has a long intracranial course, emerging from the dorsal aspect of the midbrain. It thus is highly susceptible to traumatic injury after head trauma or surgical intervention in the posterior fossa.) We included only patients who reported vertical and torsional diplopia that occurred immediately or within 2 weeks after recovery of consciousness from head trauma, because we believe that patients who have a gradual onset of vertical or torsional diplopia may represent a separate pathologic process.
Patients were excluded if they had previous eye muscle surgery or if their diplopia resulted from direct severe orbital trauma or blowout fractures. Patients also were excluded if the combined clinical signs on the Lancaster RG test suggested unilateral superior oblique paresis or skew deviation, or if they had other cranial nerve palsies. Disregarding the Bielschowsky head-tilt test findings, these inclusion and exclusion criteria aimed to ensure that the patients included in this study had both probable cause for, and the clinical signs and symptoms of, true bilateral superior oblique paresis.
All patients underwent an orthoptic evaluation including measurement of their deviations by prism and alternate cover testing in straight ahead, up, down, left, and right gazes, measured in prism diopters. Subjective torsional misalignment was assessed by the Lancaster RG plot in the 9 diagnostic positions of gaze. Degrees of torsion were measured directly from the Lancaster RG plot using a protractor. A horizontal line first was drawn connecting the lower dots on the Lancaster RG plot. For each position of gaze of interest, we drew a line through and parallel to the red steak (right eye) extending to this horizontal line. With the base of the protractor on the horizontal line, the angle of the red streak away from 90 degrees was taken as the angle of abnormal torsion (R degrees). This was repeated for the green streak (L degrees). Extorsion of the right eye was expressed in positive degrees and extorsion of the left eye was expressed in negative degrees. The combined extorsion of the 2 eyes is the difference between the 2 measurements: R degrees − L degrees. We then calculated net upgaze torsion as the average of the torsion in the 3 upgaze positions. Similar calculations were made for net straight-ahead gaze torsion and net downgaze torsion. Straight-ahead gaze extorsion of 10 degrees or more was considered supportive of the diagnosis of bilateral superior oblique paresis, as was an increase in extorsion of more than 10 degrees from upgaze to downgaze. The Lancaster RG plots were removed from the patient records and were analyzed independently of the case histories to reduce observer bias. We also used the Lancaster RG plot to confirm the V pattern in all patients.
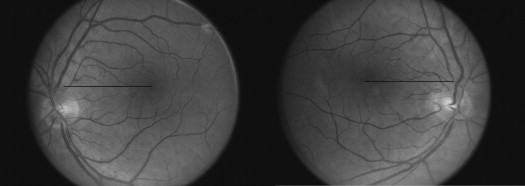
Objective fundus torsion was assessed by examination of the dilated fundus with indirect ophthalmoscopy. Torsion was graded by estimation using the method previously described by the senior author, using the indirect ophthalmoscopic view (rotated 180 degrees from the fundus camera view; Figure 1 ).
We investigated the findings for 6 previously described diagnostic tests in this population of patients with true bilateral superior oblique paresis to determine their sensitivity in identifying true bilateral superior oblique paresis: (1) the Bielschowsky head-tilt test, (2) the Parks 3-step test, (3) reversal of the hypertropia found in straight-ahead gaze to the opposite vertical deviation in any of the other 8 diagnostic positions of gaze, (4) vertical incomitance of more than 20 PD from right to left gaze, (5) the difference in magnitude of the hyperdeviation between right and left head tilt, and (6) the net subjective extorsion in straight-ahead gaze, measured by the Lancaster RG test.
The Bielschowsky head-tilt test was performed using a distance fixation target. The criteria for the diagnosis of bilateral superior oblique palsy by the Bielschowsky head-tilt test alone were a right hypertropia with right head tilt and a left hypertropia with left head tilt.
The Parks 3-step test originally was not described to make the diagnosis of bilateral paresis, but subsequently has been described using the following criteria: step 1, presence or absence of a hypertropia in straight ahead gaze; step 2, a right hypertropia on left gaze and a left hypertropia on right gaze; and step 3, the Bielschowsky head-tilt test showing alternating hyperdeviation with head tilt toward either side as described above.
Reversal of the hypertropia from straight-ahead gaze to any of the other 8 diagnostic positions of gaze has been said to be diagnostic of bilateral superior oblique paresis. We evaluated the prism and alternate cover test measurements in lateral gaze and the Lancaster RG plots to identify signs of reversal of the hypertropia in any of the other 8 diagnostic directions of gaze.
It has been suggested that a vertical incomitance between side gazes of more than 20 PD may indicate bilateral disease. We measured the incomitance of the vertical deviation on prism and alternate cover test between right and left horizontal gaze positions: hypertropia in right gaze − hypertropia in left gaze = vertical incomitance between side gazes. We assigned right hypertropia a positive value and left hypertropia a negative value and expressed the difference in absolute number of prism diopters.
Similarly, we calculated the difference in the vertical deviation, in prism diopters, between right head tilt and left head tilt in these patients: hypertropia in right head tilt − hypertropia in left head tilt = difference between right and left head tilt. Again, we assigned right hypertropia a positive value and left hypertropia a negative value and expressed the difference in absolute number of prism diopters. Published data suggest that a relatively small difference in the hypertropia on right head tilt versus left head tilt is suggestive of bilateral paresis.
Statistical Analysis
The Mann–Whitney U test was used to test the null hypothesis against non-normally distributed values, and a P value of less than .05 was considered significant.
Literature Search
A literature search was performed of the MEDLINE database using a combination of the keywords: bilateral fourth nerve , bilateral trochlear nerve , bilateral superior oblique , cyclovertical muscle , masked bilateral , palsy , paresis , ophthalmoplegia , and Lancaster red-green , covering the years 1949 to the present.
Illustrative Case History
A 25-year-old man, Patient 10, sustained a closed-head injury after a mountain biking accident. On regaining consciousness, he experienced vertical binocular diplopia that was most troublesome in downgaze. He was diagnosed as having a bilateral superior oblique paresis by the senior author 10 months after his injury. Bilateral Harada-Ito surgical procedures were recommended. The patient sought a second opinion and was told he had unilateral paresis, and unilateral superior oblique surgery was recommended. He did not undergo any surgery because of the discrepancy in recommendations and returned to our clinic 3 years later. His uncorrected visual acuity was 20/15 in both eyes. He was able to fuse at distance and near, but used his dominant right eye in downgaze. He had near stereoacuity of 40 seconds of arc in straight-ahead gaze. Ductions showed −1 limitation of depression in adduction of the right eye, and versions reflected this via −2 apparent underaction of the right superior oblique muscle. He was essentially orthophoric in straight-ahead gaze and demonstrated a V pattern with no misalignment in upgaze and an esotropia of 10 PD and right hypertropia of 16 PD in downgaze. He had an esotropia of 4 PD and right hypertropia of 2 PD on right gaze, and a right hypertropia of 6 PD on left gaze.
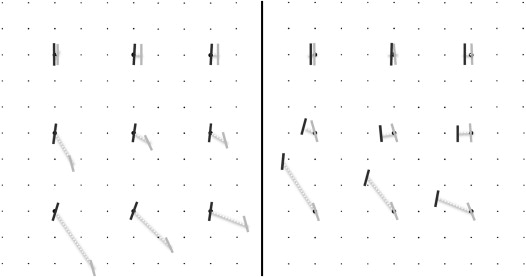
His Lancaster RG plot is shown in Figure 2 . It shows a typical pattern of asymmetric, bilateral superior oblique muscle paresis, greater on the right than the left, with increasing right hyperdeviation in downgaze. The V-pattern esotropia is nicely demonstrated here. There is significantly increased bilateral extorsion in downgaze, greatest in the field of action of the right superior oblique muscle, presumably because of the greater weakness of that muscle. Examination of the fundus showed bilateral trace extorsion. He underwent bilateral Harada-Ito surgery under general anesthesia, using adjustable sutures, a small right medial rectus muscle recession of 1.5 mm, and a left inferior rectus muscle recession of 1.5 mm, both after adjustment. Fundus torsion was assessed under anesthesia, before surgery by indirect ophthalmoscopy, and both fundi appeared to be approximately 1+ extorted. The sutures were adjusted (tightened) under anesthesia to create consecutive 1+ to 2+ intorsion bilaterally before the patient was awakened. The patient was evaluated 4 hours later and showed a left hypertropia and mild overcorrection of the extorsion in both eyes. The Lancaster RG test was repeated, and the sutures were adjusted until there was no subjective torsion in downgaze and in straight-ahead gaze and there was mild intorsion in upgaze. At the 6-week postoperative assessment, he was able to look farther into downgaze while still fusing, but still experienced some vertical misalignment in far downgaze. He was not troubled by diplopia in upgaze, and his visual acuity and near stereoacuity remained stable. He remained orthophoric in straight-ahead gaze with no significant A or V pattern and only 1 PD of esotropia in downgaze. His Lancaster RG test did demonstrate bilateral intorsion in upgaze and a small consecutive left hypertropia in extreme downgaze to the right. The patient was happy with the outcome.
Results
Thirty-four patients were identified, of whom 25 met our inclusion and exclusion criteria. The patients’ demographic profiles showed a male-to-female ratio of 1.78, with an average age of 31 years (standard deviation [SD], 11.3 years). Twenty-two patients had sustained head trauma after a motor vehicle accident, 1 patient sustained a head injury after a fall, and 2 patients had undergone surgery for resection of a brain tumor. All patients reported vertical diplopia immediately or within 2 weeks of regaining consciousness after their head injury. The average time between the injury and review in our clinic was 4.75 years (SD, 4.95 years). Nine (36%) of our 25 patients had a chin-down head posture, typical of bilateral superior oblique paresis when fusion can be obtained in upgaze.
The Lancaster RG test confirmed that all 25 patients had greater extorsion in downgaze than in upgaze. The net fundus extorsion ± SD in upgaze was 6.6 ± 6.9 degrees and the net fundus extorsion in downgaze ± SD was 25.2 ± 7.3 degrees ( P < .001). All 25 patients showed bilateral fundus extorsion in primary gaze on indirect ophthalmoscopy. Twenty-one patients had between trace and +1 fundus extorsion in each eye, and 4 patients showed between +1.5 and +4 extorsion.
Orthoptic measurements in right and left head tilt were available for all 25 patients ( Table 1 ), but the lateral gaze measurements were available only for 23 patients. For the 2 patients who did not have these measurements recorded in the case notes (Patients 15 and 24), we directly measured the misalignment in lateral gazes from the Lancaster RG plot ( Table 2 ).
| Patient No. | Superior Oblique Paresis as Diagnosed by Bielschowsky Head-Tilt Test | Head-Tilt Test Results to the Right | Head-Tilt Test Results to the Left |
|---|---|---|---|
| 1 | Right | RHT 16 | RHT 8 |
| 2 | Right | RHT 10 | No HT |
| 3 | Right | RHT 9 | RHT 1-2 |
| 4 | Right | RHT 3 | No HT |
| 5 | Right | RHT 12 | No HT |
| 6 | Right | RHT 22 | RHT 2 |
| 7 | Left | No HT | LHT 5 |
| 8 | Left | LHT 1 | LHT 3 |
| 9 | Left | No HT | LHT 5 |
| 10 | Left | LHT 3 | LHT 10 |
| 11 | Left | No HT | LHT 16 |
| 12 | Left | No HT | LHT 2 |
| 13 | Left | No HT | LHT 16 |
| 14 | Left | No HT | LHT 3 |
| 15 | Left | LHT 4 | LHT 30 |
| 16 | Bilateral | RHT 12 | LHT 1 |
| 17 | Bilateral | RHT 5 | LHT 2 |
| 18 | Bilateral | RHT 3 | LHT 3 |
| 19 | Bilateral | RHT 8 | LHT 20 |
| 20 | Bilateral | RHT 5 | LHT 2-3 |
| 21 | Bilateral | RHT 3 | LHT 9 |
| 22 | Bilateral | RHT 9 | LHT 6 |
| 23 | Bilateral | RHT 25 | LHT 14 |
| 24 | Bilateral | RHT 6 | LHT 1 |
| 25 | Bilateral | RHT 28 | LHT 8 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


