Purpose
To evaluate the feasibility, safety, and utility of intraoperative optical coherence tomography (OCT) for use during ophthalmic surgery.
Design
Prospective, consecutive case series.
Methods
A prospective, single-center, consecutive case series was initiated to assess intraoperative OCT in ophthalmic surgery. Intraoperative scanning was performed with a microscope-mounted spectral-domain OCT system. Disease-specific or procedure-specific imaging protocols (eg, scan type, pattern, size, orientation, density) were used for anterior and posterior segment applications. A surgeon feedback form was recorded as part of the study protocol to answer specific questions regarding intraoperative OCT utility immediately after the surgical procedure was completed.
Results
During the first 24 months of the PIONEER study, 531 eyes were enrolled (275 anterior segment cases and 256 posterior segment surgical cases). Intraoperative OCT imaging was obtained in 518 of 531 eyes (98%). Surgeon feedback indicated that intraoperative OCT informed surgical decision making and altered surgeon understanding of underlying tissue configurations in 69 of 144 lamellar keratoplasty cases (48%) and 63 of 146 membrane peeling procedures (43%). The most common anterior segment surgical procedure was Descemet stripping automated endothelial keratoplasty (DSAEK, n = 135). Vitrectomy with membrane peeling was the most common procedure for posterior segment surgery (n = 154). The median time that surgery was paused to perform intraoperative OCT was 4.9 minutes per scan session. No adverse events were specifically attributed to intraoperative OCT scanning during the procedure.
Conclusions
Intraoperative OCT is feasible for numerous anterior and posterior segment ophthalmic surgical procedures. A microscope-mounted intraoperative OCT system provided efficient imaging during operative procedures. The information gained from intraoperative OCT may impact surgical decision making in a high frequency of both anterior and posterior segment cases.
Optical coherence tomography (OCT) has revolutionized the clinical management of ophthalmic diseases. The outstanding cross-sectional anatomic information afforded physicians with OCT imaging has transformed the diagnosis, surveillance, and treatment paradigms for both anterior and posterior segment diseases. The translation of this technology to the operating room theater may have profound implications for the optimal surgical management of ophthalmic diseases.
Intraoperative OCT remains a young and emerging field. The description of intraoperative OCT has been limited to case reports and retrospective case series. These reports have highlighted the potential for intraoperative OCT to impact our understanding of the pathophysiology of surgical diseases, to facilitate optimal decision making regarding achievement of surgical objectives, and to improve our understanding of the tissue alterations that occur during surgical manipulations. The use of intraoperative OCT has been described for a wide variety of conditions and procedures, including optic pit–related maculopathy, epiretinal membranes, vitreomacular traction syndrome, macular hole, retinal detachments, lamellar keratoplasty, retinopathy of prematurity, and cataract surgery. These studies were in general small (<30 patients) and retrospective but offered early insight into intraoperative OCT feasibility and utility, while providing an important foundation for future research.
To better understand the feasibility and potential utility of intraoperative OCT, the Prospective Intraoperative and Perioperative Ophthalmic ImagiNg with Optical CoherEncE TomogRaphy: PIONEER Study was initiated. The focus of this prospective study is examining the feasibility, utility, and safety of performing intraoperative OCT and perioperative OCT for a wide variety of surgeries to better delineate the role for intraoperative OCT in a large number ophthalmic conditions. This report highlights the 2-year results of the feasibility, utility, and safety of the intraoperative OCT portion of this large prospective study.
Methods
PIONEER is a single-site, multi-surgeon, prospective clinical study examining the feasibility, safety, and utility of intraoperative OCT in the surgical management of ophthalmic disease. The study was approved by the Cleveland Clinic Institutional Review Board and adhered to all the tenets of the Declaration of Helsinki and HIPAA policies. All patients gave written informed consent before enrollment in the study. The primary inclusion criterion was any patient undergoing an ophthalmic incisional surgical procedure willing to undergo intraoperative OCT imaging. Exclusion criteria included any media opacity that precluded OCT scanning of the area of interest and inability to provide written informed consent. Patients were allowed to re-enroll in PIONEER (eg, fellow eye surgery, same eye reoperation). Additionally, bilateral OCT imaging was allowed for bilateral surgical procedures (eg, refractive surgery).
Clinical and surgical variables were collected including past ocular history, procedure type, preoperative diagnosis, pertinent surgical maneuvers/techniques (eg, instrumentation, unique manipulations), number of imaging sessions during surgery, duration of imaging sessions, interruption of surgical procedure time for imaging, and adverse events. Patients were also evaluated on the first postoperative day to assess for clinical variables, perioperative images in select cases, and adverse events. The structured study follow-up ended on postoperative day 1; however, patients were consented for up to 2 years of surveillance of medical records (eg, best-corrected Snellen visual acuity, OCT outcomes). Any visits beyond the first postoperative visit were based on standard-of-care visits per the individual surgeon.
A dedicated intraoperative OCT research coordinator assisted with data acquisition in the operating room. Intraoperative scanning was performed with the Bioptigen SDOIS portable spectral-domain OCT (SD OCT) system (Bioptigen, Research Triangle Park, North Carolina, USA). The portable SD OCT probe was stabilized with a custom-designed, microscope-mount system ( Figure 1 ) or with handheld scanning. The microscope mount system was developed for use with both Leica and Zeiss microscopes. It also was compatible with the BIOM indirect viewing system as well as contact lens wide-angle viewing systems. When using the microscope mount, the surgeon used the microscope pedal to control the X-Y-Z translation of the probe to image the area of interest.
Disease-specific or procedure-specific imaging protocols (eg, scan type, pattern, size, orientation, density) were outlined for anterior and posterior segment applications. Anterior segment imaging included a 12 mm × 12 mm volumetric cube scan at 0 degree orientation. Posterior segment imaging included a 10 mm × 10 mm volumetric cube with 100 B-scans per volume at both 0 and 90 degrees, a 5 × 10 mm volumetric cube with 7 B-scans per frame for averaging and a total of 175 B-scans per volume at both 0 and 90 degrees, and a 10 mm radial scan with 100 B-scans per volume. Surgeons were provided with guidance for surgical milestones to consider for imaging (eg, pre-incision, following membrane peeling, following graft placement), but the specific imaging sessions were at the surgeon’s discretion. When possible in vitreoretinal cases, scan sequences were obtained in the operating room prior to any surgical manipulation of the area of interest. Following various surgical milestones, scan sequences were performed to allow for comparative data and change analysis. A “scan” was defined as a single set of images (eg, volume scan). A “scan session” was defined as a series of scans obtained during a single pause during surgery. Typically, multiple scan sessions were often performed during the surgical procedures.
A specific surgical procedure (ie, membrane peeling, lamellar keratoplasty) intraoperative OCT feedback form was included as part of the study protocol. This form included questions regarding the impact of the intraoperative OCT on surgical decision making and on how intraoperative OCT may have impacted the understanding of the surgical disease of interest.
Results
Clinical Demographics
During the first 24 months, 531 eyes were enrolled in the PIONEER study. This included 275 eyes in the anterior segment arm and 256 eyes in the posterior segment arm ( Table 1 ). Overall, 285 right eyes and 246 left eyes were included in the analysis. Successful imaging was obtained in 518 of 531 eyes (98%) at some point during the surgical procedure, with variable image quality. Reasons for lack of imaging included poor media clarity and surgeon choice based on logistics (eg, delay to surgery, timing).
| Anterior | Posterior | |||
|---|---|---|---|---|
| Enrollment | 275 | 256 | ||
| Eye | ||||
| Right | 150 | 135 | ||
| Left | 125 | 121 | ||
| Lens status | ||||
| Pseudophakic | 124 | 87 | ||
| Phakic | 147 | 47 | ||
| Aphakic | 4 | 13 | ||
| Concurrent CE/IOL with PPV | 109 | |||
| Preoperative diagnosis | Fuchs dystrophy | 82 (30%) | Epiretinal membrane | 90 (35%) |
| Cataract | 66 (24%) | Full-thickness macular hole | 58 (23%) | |
| Bullous keratopathy | 32 (12%) | Retinal detachment | 44 (17%) | |
| Failed DSAEK | 23 (8%) | Proliferative diabetic retinopathy | 32 (13%) | |
| Keratoconus | 16 (6%) | Vitreous hemorrhage | 8 (3%) | |
| Refractive error | 12 (4%) | Vitreomacular traction | 6 (2%) | |
| Failed PK | 11 (4%) | Other | 18 (7%) | |
| Other | 33 (12%) | |||
| Procedures | DSAEK | 135 (49%) | Vitrectomy | 253 |
| CE/IOL | 29 (11%) | Vitrectomy and scleral buckle | 15 | |
| Femto CE/IOL | 37 (13%) | Other | 3 | |
| DALK | 19 (7%) | |||
| LASIK | 11 (4%) | |||
| DSAEK rebubble | 8 (3%) | |||
Anterior Segment Intraoperative Optical Coherence Tomography Summary
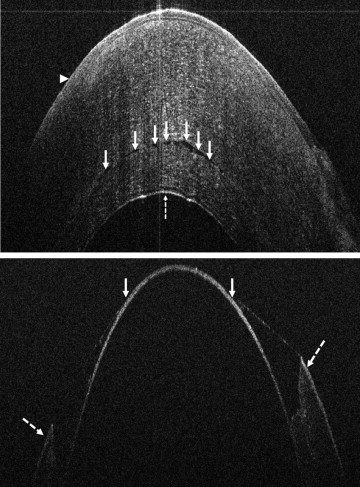
In the anterior segment group, 275 eyes were enrolled. Of these 275 eyes, 124 were pseudophakic, 147 eyes were phakic, and 4 eyes were aphakic. The most common indications for anterior segment surgery were Fuchs endothelial cell dystrophy (30%), cataract (24%), bullous keratopathy (12%), previous failed DSAEK (8%), and keratoconus (6%) ( Table 1 ). The most common surgical procedures performed were Descemet stripping automated endothelial keratoplasty (DSAEK), standard phacoemulsification cataract surgery, femtosecond-assisted phacoemulsification cataract surgery, and deep anterior lamellar keratoplasty (DALK) ( Table 1 ). For DSAEK, intraoperative OCT provided excellent visualization of fluid in the graft/host interface ( Figure 2 ). Graft apposition was monitored intraoperatively with intraoperative OCT and the procedure was continued until optimal fluid removal was achieved. The graft dislocation rate on postoperative day 1 was 3% (5/143). In DALK, intraoperative OCT images provided depth information regarding extent of trephination and residual bed information ( Figure 2 ). Intraoperative OCT was also used for intracorneal ring segments placement and cataract surgery. Implant location was easily identified with intraoperative OCT and was achieved for both intracorneal ring segments and intraocular lens placement ( Figure 3 ). Wound architecture was visualized with intraoperative OCT during cataract surgery ( Figure 3 ).
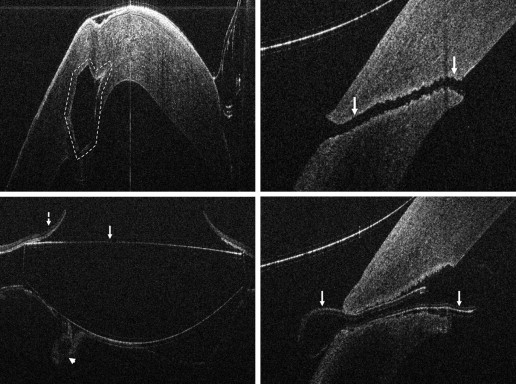
Impact of Intraoperative Optical Coherence Tomography on Surgical Decision Making: Lamellar Keratoplasty
In lamellar keratoplasty procedures, 144 cases included completed surgeon feedback forms. For DSAEK procedures, following graft placement and prior to intraoperative OCT imaging, the surgeon believed the graft was completely apposed in 31 of 135 cases (23%), believed the graft was not apposed in 17 of 135 (13%), and was unsure of apposition in 87 of 135 cases (64%). Additional manipulations were performed (eg, corneal sweep, varied infusion pressure) based on intraoperative OCT imaging findings of incomplete apposition and persistent interface fluid. In 9 of 31 cases (29%) in which the surgeon believed the graft was entirely apposed, intraoperative OCT revealed persistent fluid that resulted in additional manipulations. In 3 of 17 cases (18%) in which the surgeon thought that the graft was not apposed, the intraoperative OCT revealed complete apposition confirming completion of the surgical objectives. Finally, in cases where the surgeon was unsure of apposition, 42 of 87 eyes (48%) revealed persistent fluid requiring additional manipulations. Overall following intraoperative OCT, 65 of 135 eyes (48%) revealed persistent interface fluid requiring additional manipulations. In DALK procedures, the initial trephination was not believed to be deep enough in 3 of 9 cases and the surgeon was unsure of depth in 6 of 9 cases prior to intraoperative OCT imaging. In 1 of 3 cases where the surgeon did not believe the trephination was deep enough, intraoperative OCT revealed the depth was optimal and did not require further deepening. In 5 of 9 cases intraoperative OCT prompted further manual dissection to deepen the initial trephination.
Posterior Segment Intraoperative Optical Coherence Tomography Summary
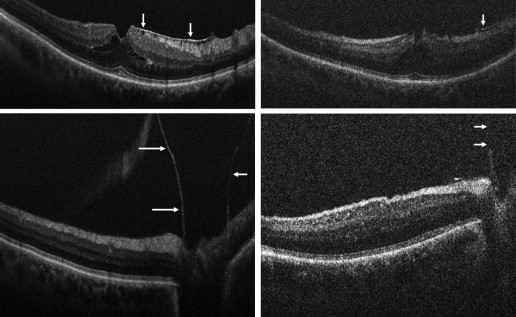
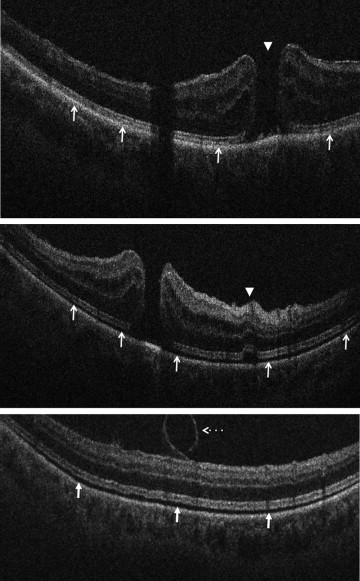
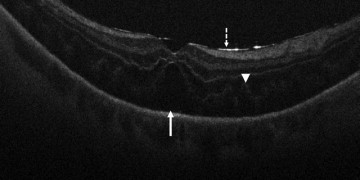
In the posterior segment group, 256 eyes were included. Of these eyes, 87 were pseudophakic, 47 were phakic, 109 underwent concurrent phacoemulsification, and 13 were aphakic. The most common indications for posterior segment surgery were epiretinal membrane (ERM, 35%), full-thickness macular hole (FTMH, 23%), rhegmatogenous retinal detachment (RD, 17%), proliferative diabetic retinopathy (PDR, 13%), and vitreous hemorrhage (VH, 3%) ( Table 1 ). Nearly all eyes (253 of 256) underwent vitrectomy. Fifteen of 256 eyes underwent combined scleral buckle and vitrectomy procedures. For epiretinal membrane cases, intraoperative OCT was able to visualize areas of successful membrane peeling and reveal any residual membranes ( Figure 4 ). Additionally, alterations in the outer retinal architecture were noted with observation of the expansion between the ellipsoid zone and the retinal pigment epithelium (RPE) after peeling. Similarly, in FTMH cases, changes in hole architecture were frequently seen and identification of residual internal limiting membrane as well as expansion of the ellipsoid zone to RPE height was noted after membrane peeling ( Figure 5 ). Intraoperative OCT imaging in retinal detachment surgery identified variable amounts of residual subretinal fluid following perfluorocarbon liquid placement ( Figure 6 ). Using intraoperative OCT imaging during surgical intervention for vitreomacular traction, confirmation of release of traction was feasible and identification of newly formed FTMH occurred in select cases ( Figure 7 ).

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


