Purpose
To evaluate the therapeutic benefits of tear-exchangeable, limbal, rigid contact lenses (limbal CLs) in patients with Stevens-Johnson syndrome- or toxic epidermal necrolysis-associated ocular sequelae.
Design
Noncomparative, retrospective, interventional case series.
Methods
We enrolled 53 eyes of 42 patients (mean age, 51.8 ± 13.9 years; mean follow-up, 25.7 ± 15.7 months) with Stevens-Johnson syndrome- or toxic epidermal necrolysis-associated ocular sequelae and divided them into 3 groups according to the best-corrected visual acuity (BCVA) before limbal CL fitting: (1) BCVA worse than 20/2000 (11 eyes), (2) BCVA ranging from 20/200 to 20/2000 (31 eyes), and (3) BCVA of 20/200 or better (11 eyes). The BCVA and the 25-item National Eye Institute Visual Function Questionnaire (NEI VFQ-25) composite score before fitting and after 3 months of limbal CL use were evaluated. The change in BCVA (in logarithm of the minimal angle of resolution [logMAR] units) and 25-item National Eye Institute Visual Function Questionnaire composite score change were compared among the 3 groups.
Results
Best-corrected visual acuity improved from 1.61 to 0.86 logMAR at 3 months after fitting CL use. Improvement in BCVA in groups 1, 2, and 3 was 0.95 logMAR, 0.82 logMAR, and 0.37 logMAR, respectively. The mean 25-item National Eye Institute Visual Function Questionnaire composite score for the 11 subscales improved from 37.6 ± 16.0 to 58.4 ± 17.4 ( P = .000001). All 11 subscores, except that for driving ability, improved significantly. The general vision subscore improved most in group 3, yet the general health subscore improved most in group 1. No serious adverse events attributable to limbal CL use occurred.
Conclusions
The tear-exchangeable limbal CL is safe and effective for the improvement of vision and quality of life in Stevens-Johnson syndrome or toxic epidermal necrolysis patients with severe ocular sequelae.
Stevens-Johnson syndrome (SJS), and its severe variant, toxic epidermal necrolysis (TEN), are acute, life-threatening diseases of the skin and mucous membranes. Although the incidence of SJS and TEN is very low, that is, approximately 0.4 to 1 and 1 to 6 cases per 1 million persons, respectively, both can affect anyone at any age, usually as a consequence of adverse drug reactions. In more than 50% of SJS or TEN patients, ocular complications are involved at the acute stage of the disease. In mild cases, bilateral acute conjunctivitis occurs before, or simultaneously with, acute fever and skin eruption. In severe cases, ocular surface epithelial defect, pseudomembranous conjunctivitis, or both are observed, and extensive ocular surface inflammation persists for more than several weeks after disease onset. Coinciding with the remission of each disease, ocular sequelae associated with the loss of corneal epithelial stem cells such as conjunctivalization, neovascularization, opacification, and keratinization of the cornea appear. All of these cornea-related ocular sequelae disrupt visual function. Cicatrization resulting from fibrosis, symblepharon, trichiasis, and xerophthalmia cause ocular discomfort such as eye pain, dry eye, or foreign body sensation, and visual impairment and ocular discomfort continue throughout the life of the patient. In more than 50% of eyes with SJS- or TEN-associated ocular sequelae, best-corrected visual acuity (BCVA) reportedly is worse than 20/200. Therefore, it is vital to improve the visual acuity (VA) of SJS or TEN patients with ocular sequelae.
The reported long-term outcomes of penetrating keratoplasty, lamellar keratoplasty, or limbal transplantation for SJS or TEN patients is poor. Graft rejection or the loss of donor epithelial cells occurs easily, ultimately resulting in progressive conjunctivalization and scarring of the ocular surface, which once again causes severe vision loss. Amniotic membrane transplantation alone or in combination with limbal transplantation can be used for epithelialization, reducing pain, and reconstructing the conjunctival fornix in SJS or TEN patients. Cultivated limbal epithelial transplantation or cultivated oral mucosal epithelial transplantation (COMET) are 2 methods that improve visual function in eyes with SJS- or TEN-associated ocular sequelae. However, it is impossible to restore the patient’s ocular surface to its previous normal and healthy state.
Scleral contact lenses (CLs) originally were introduced in the 1880s in the form of a rigid contact lens made from glass. Well-designed scleral CLs with minimal clearance reportedly have been used for the treatment of severe dry eye of various causes. However, nonmanageable sequelae such as corneal vascularization and ulceration were serious problems associated with the use of those lenses. The introduction of rigid gas-permeable materials to the construction of scleral CLs increased corneal oxygenation and improved the therapeutic efficacy of using scleral CLs for the treatment of ocular surface disorders. At present, scleral CLs with high oxygen permeability are fitted for the treatment of a variety of diseases manifesting an irregularly shaped cornea, such as keratoconus or corneal ectasia, or in patients who have undergone penetrating keratoplasty. In the use of scleral CLs, artificial tears are captured under the CL and remain trapped between the CL and the cornea until the CL is removed. Because artificial tears usually remain between the CL and the cornea for an extended period, an unhealthy physiologic status can arise.
Recent reports have demonstrated the therapeutic benefits of scleral CLs in the management of severe ocular surface diseases such as SJS and TEN. In most of the patients in those studies, the initial VA before CL fitting was 20/200 or better. However, the findings of our previous study revealed that in more than 50% of eyes with ocular sequelae, VA is worse than 20/200. Because the size of scleral CLs ranges from 16 to 23 mm, they are too large to be used on severely cicatrized eyes with conjunctival fornix shortening. Even in normal eyes, the depth of the fornix in Asians is smaller than that in white persons.
Recently, we developed a new type of rigid contact lens with a 13.0- or 14.0-mm diameter size (smaller and larger diameter sizes also can be made) that can be used in eyes with a short fornix, symblepharon, or both. Because severe dry eye is a frequent, long-term ocular sequelae caused by SJS or TEN, the tear film plays a critical role in the homeostasis of the ocular surface in those patients. Thus, we designed this new CL to enable tear exchange under the CL during the blinking process. We named this new tear-exchangeable CL the limbal rigid contact lens (or limbal CL). The aim of this present study was to evaluate the safety and therapeutic benefits of the limbal CL in patients with ocular sequelae caused by SJS or TEN.
Methods
The study was approved by the Ethics Committee and Institutional Review Board of Kyoto Prefectural University of Medicine, Kyoto, Japan, and was carried out in accordance with the tenets set forth in the Declaration of Helsinki. Written informed consent was obtained from each patient before the initiation of the study. The study protocol was designed to evaluate the efficacy and safety of the limbal CL. The SJS or TEN patients with ocular sequelae seen at the SJS outpatient service at Kyoto Prefectural University Hospital were enrolled. The diagnosis of SJS was based on a confirmed history of the acute onset of high fever, serious mucocutaneous illness with skin eruptions, and the involvement of at least 2 mucosal sites, including the ocular surface. Patient inclusion criteria were as follows: (1) visual disturbance, severe dry eye, or both caused by SJS or TEN; (2) patients who can handle the CL by themselves, or with the support of family; and (3) age older than 7 years. Exclusion criteria were as follows: (1) patients with systemic or ocular infections, (2) patients with colonization of methicillin-resistant Staphylococcus aureus or methicillin-resistant Staphylococcus epidermidis on the ocular surface, (3) lost vision resulting from other reasons such as glaucoma or retinal diseases, (4) patients who are unable to come to the hospital at regular intervals, and (5) patients with dementia. Only the patients who met all of the inclusion criteria were enrolled in this study.
Materials and Design of the Limbal Contact Lens
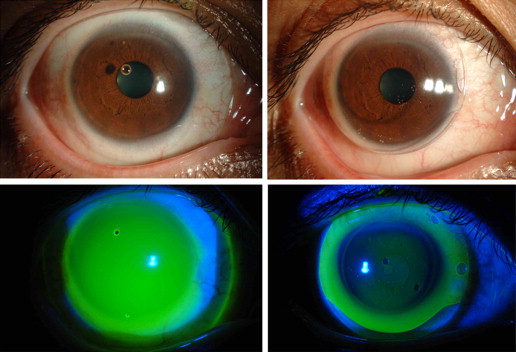
The limbal CL is lathed from Hexafocon A (Bausch & Lomb, Inc, Rochester, New York, USA), a special polymer with an oxygen permeability value of 100 Dk. The curvature of the lens was designed with an 8.5- or 9.0-mm diameter optical zone at the center of the lens and a peripheral zone at the peripheral area of the lens ( Figure 1 ). The peripheral zone is designed in the shape of a projecting edge, for instance, like the brim of a hat, which consists of a quad-curve design that enables the inflow of tears under the CL at the time of blinking. In the use of scleral CLs, the edge of the CL touches the sclera and the thick fluid coverage beneath the CL remains trapped and does not exchange ( Figure 2 , Top left and Bottom left). In contrast, in the use of limbal CLs, a thin fluid layer exists under the CL with a fluid reservoir at the peripheral zone of the CL ( Figure 2 , Top right and Bottom right), thus allowing tear exchange beneath the CL at every blink.
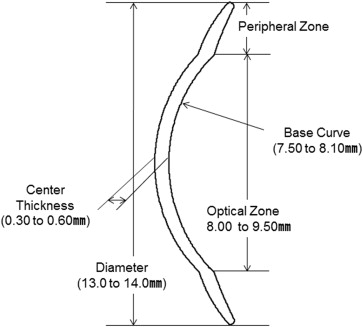
Although patients using scleral CLs must remove and clean the CL several times per day, patients using the limbal CL do not need to remove and clean the lens because of the continual tear exchange. The diameter of the lens (13 or 14 mm; larger and smaller diameter sizes also can be made), the base curve (750 to 810 mm), the size of the optical zone (8.5 or 9 mm), and the peripheral-area design (2 available patterns: flat or tight) are selections that are determined by the physician at the time of the patient’s trial fitting.
Tear Exchange Under the Contact Lens
Of the 53 total eyes involved in this study, 4 eyes of 3 patients who wear scleral CLs participated in the preliminary experiment to elucidate tear exchange via fluorescein staining patterns under the CL, that is, between the CL and the cornea. All 3 of those patients used a Boston Scleral Lens Prosthetic Device (The Boston Foundation for Sight, Inc, Needham, Massachusetts, USA) prescribed at other clinics. Those 4 eyes were examined and selected to wear either a scleral CL or the limbal CL. In the experiment, 50 μL of 0.001% sodium fluorescein dye (Fluorescite; Alcon Japan Ltd, Tokyo, Japan) was put into the concave side of the respective CLs before CL wear to elucidate the change in fluorescein pattern and concentration between the CL and the cornea associated with each type of CL. Each type of CL first was filled with fluorescein dye, and then placed on the patient’s eye. The patient then was instructed to blink normally for 10 minutes. Artificial tears then were instilled in the eye at between 5 and 10 minutes after insertion of the CL, and the change in fluorescein staining pattern and concentration then was calculated by a fluorophotometer (Anterior Fluorometer FL-500; Kowa Company, Ltd, Nagoya, Japan) at the following 3 time points: (1) immediately after insertion of the CL, (2) after 5 minutes of CL wear, and (3) after 10 minutes of CL wear.
Ocular Surface Grading Score
All enrolled patients underwent slit-lamp examinations to observe 12 components of the following 3 categories of ocular sequelae: (1) corneal sequelae consisting of epithelial defect, loss of the palisades of Vogt, conjunctivalization, neovascularization, opacification, and keratinization; (2) conjunctival sequelae consisting of hyperemia and symblepharon formation; and (3) eyelid sequelae consisting of trichiasis, mucocutaneous junction involvement, meibomian gland involvement, and punctal damage in accordance with our previously reported grading system. Each component was graded on a scale from 0 to 3, depending on the severity of involvement. The sum of each grading score was defined as the ocular surface grading score (maximum score, 36).
Outcomes
The main outcome measure was the BCVA before CL fitting and after 3 months use of the limbal CL after fitting. The 25-item National Eye Institute Visual Function Questionnaire (NEI VFQ-25) composite score was defined as the secondary outcome. The SJS- or TEN-related ocular surface sequelae were graded on a scale from 0 to 3 according to their severity as described above. The patients were divided into 3 groups according to the BCVA before CL fitting: group 1, VA worse than 20/2000 (the logarithm of the minimal angle of resolution [logMAR] > 2); group 2, VA from 20/200 to 20/2000 (2 ≥ logMAR > 1); and group 3, VA 20/200 or better (1 ≥ logMAR). The ocular surface grading score, the change of VA, and the change of the NEI VFQ-25 composite score then were compared among the 3 groups.
Visual Acuity
Best-corrected visual acuity was measured at baseline (before fitting of the limbal CL) and after 3 months use of the limbal CL. Best-corrected visual acuity at baseline was best-corrected spectacle VA, and BCVA at after 3 months of CL use was measured with the CL fitted, and if needed, with spectacles. Improvements in VA were analyzed in relation to changes in logMAR BCVA. Counting fingers, hand movements, and light perception were determined to be 0.004, 0.002, and 0.001, respectively.
Visual Function Questionnaire
Each patient completed a Japanese language version of the NEI VFQ-25 at the baseline examination and after 3 months of limbal CL use to evaluate their vision-related quality of life. The questionnaire was administered via a direct interview by a trained interviewer (S.M.) who was otherwise not involved in the patient’s care.
Statistical Analysis
The Wilcoxon signed-rank test was used to compare the NEI VFQ-25 data at baseline and after 3 months of limbal CL use. P values of less than .05 were considered statistically significant.
Additional Collected Data
For each patient, additional collected data included the diameter and the peripheral design of the CL, the base curve and the size of the optical zone, the length in time of CL wear, patient reports of symptoms, if any, during CL wear, and all adverse events that occurred during the period of CL use.
Results
Patient Characteristics
This retrospective study involved a total of 53 eyes of 42 SJS or TEN patients (15 men and 27 women) ranging in age from 28 to 78 years (mean age ± standard deviation [SD], 51.8 ± 13.9 years). At disease onset, patient age ranged from 2 to 64 years (mean age ± SD, 22.4 ± 16.7 years), and 23 (53.5%) of the 41 patients were younger than 20 years. The duration of the illness ranged from 2 to 68 years (mean duration ± SD, 29.3 ± 15.7 years), and the mean ± SD patient follow-up period was 25.7 ± 15.7 months. The causative drugs were cold medicines in 13 patients, nonsteroidal anti-inflammatory drugs in 11 patients, antibiotics in 6 patients, anticonvulsants in 2 patients, and others in 6 patients.
Prior ocular surgeries had been performed in 37 (70.0%) of the 53 eyes at Kyoto Prefectural University of Medicine or at a different medical facility. Corneal reconstruction was performed in 21 eyes: COMET in 9 eyes, cultivated limbal epithelial transplantation in 5 eyes, limbal transplantation or keratoepithelioplasty in 5 eyes, and penetrating keratoplasty or lamellar keratoplasty in 6 eyes. Conjunctival reconstruction was performed in 17 eyes: amniotic membrane transplantation in 11 eyes, COMET in 2 eyes, oral mucosal transplantation in 2 eyes, and other surgeries in 2 eyes. Cataract surgery was performed in 13 eyes. Entropion surgeries were performed in 12 eyes, and ptosis surgeries were performed in 4 eyes. Fitting and use of the limbal CL was initiated only when the ocular surface had been stable for at least 1 month after cataract or eyelid surgery and for at least 3 months after ocular surface reconstruction or keratoplasty.
Ocular Surface Grading Score
As reported previously, the ocular surface grading score reflects the severity of sequelae caused by SJS or TEN. The loss of the palisades of Vogt and meibomian gland involvement were grade 3 in 50 eyes (94.3%) and 51 eyes (96.2%), respectively. That is, more than 95% of eyes were limbal stem cell deficient and also had severe meibomian gland dysfunction. Mild to moderate neovascularization and opacification of the cornea existed ( Supplemental Table , available at AJO.com ).
There were 11 eyes in group 1, 31 eyes in group 2, and 11 eyes in group 3. All ocular surface grading scores (except those for hyperemia and punctal damage), as well as the total score, were highest in group 1 and lowest in group 3 ( Table 1 ).
| Total | BCVA before Limbal Contact Lens Fitting | |||
|---|---|---|---|---|
| Group 1: BCVA worse than 20/2000 (logMAR > 2), Average Grade | Group 2: BCVA 20/200 to 20/2000 (2 ≥ logMAR >1), Average Grade | Group 3: BCVA 20/200 or Better (1 ≥ logMAR), Average Grade | ||
| No. of eyes | 53 | 11 | 31 | 11 |
| Ocular surface grading score | ||||
| Corneal complications | ||||
| Epithelial defect | 0.00 | 0.00 | 0.00 | 0.00 |
| Loss of POV | 2.89 | 2.91 | 2.94 | 2.73 |
| Conjunctivalization | 2.70 | 2.82 | 2.71 | 2.55 |
| Neovascularization | 2.02 | 2.64 | 1.94 | 1.64 |
| Opacification | 1.42 | 2.27 | 1.23 | 1.09 |
| Keratinization | 0.83 | 1.82 | 0.77 | 0.00 |
| Conjunctival complications | ||||
| Hyperemia | 1.17 | 1.00 | 1.16 | 1.36 |
| Symblepharon formation | 1.25 | 1.73 | 1.23 | 0.82 |
| Eyelid complications | ||||
| Trichiasis | 1.77 | 2.09 | 1.90 | 1.09 |
| Mucocutaneous junction involvement | 2.04 | 2.36 | 2.00 | 1.82 |
| Meibomian gland involvement | 2.94 | 3.00 | 3.00 | 2.73 |
| Punctal damage | 2.51 | 2.64 | 2.45 | 2.55 |
| Total | 21.7 | 25.3 | 21.5 | 18.5 |
Tear Exchange under the Contact Lens
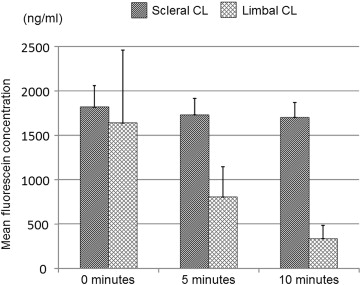
The results of the tear-exchange experiment revealed substantial differences in fluorescein staining patterns and concentrations between the scleral CL and the limbal CL. Fluorescein patterns showed no change of fluorescein concentration over a 10-minute period in the eyes with the scleral CL, but did reveal a gradual decrease of fluorescein concentration in the eyes with the limbal CL ( Figure 3 ).
Limbal Contact Lens Wear in Eyes with Ocular Surface Scarring
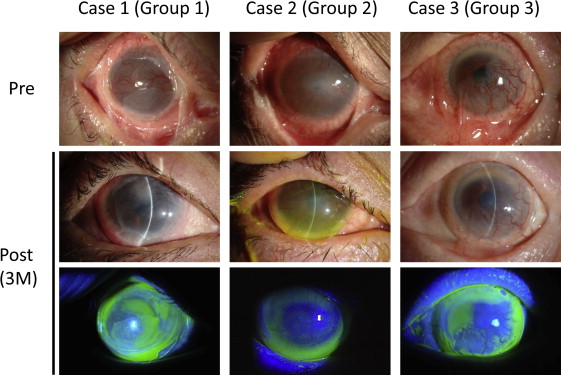
The limbal CLs were able to be used for eyes with fornix shortening, conjunctivalization, and neovascularization of the cornea, and there was no need to fill the CL with saline or artificial tears. During limbal CL wear, a thin fluid layer existed beneath the CL ( Figure 4 ) and the precorneal fluid layer exchanged at every blink ( Supplemental Video , available at AJO.com ).
Visual Acuity
Best-corrected visual acuity improved from 1.61 to 0.86 logMAR after 3 months of limbal CL use, and in 43 eyes (81.1%), the BCVA improvement was more than 0.2 logMAR ( Figure 5 ). The BCVA improvement in groups 1, 2, and 3 was 0.95 logMAR, 0.82 logMAR, and 0.37 logMAR, respectively ( Table 2 ). Mean BCVA after 3 months of CL use was 1.55 logMAR in group 1, the worst among the 3 groups. However, BCVA improvement was best in group 1. Of 29 cases in which the CL was fitted unilaterally, the CL was fitted in the eye with better VA in 26 cases and in the eye with worse VA in 3 cases. At the final examination, the eye fitted with the limbal CL was the eye with better VA in all 29 unilateral cases.

| Total | BCVA Before Limbal CL Fitting | |||
|---|---|---|---|---|
| Group 1: BCVA Worse Than 20/2000 (logMAR >2), Average Grade | Group 2: BCVA 20/200 to 20/2000 (2 ≥ logMAR > 1), Average Grade | Group 3: BCVA 20/200 or Better (1 ≥ logMAR), Average Grade | ||
| No. of eyes | 53 | 11 | 31 | 11 |
| BCVA before limbal CL fitting (logMAR) | 1.61 | 2.51 | 1.67 | 0.57 |
| BCVA after 3 months of limbal CL use (logMAR) | 0.86 | 1.55 | 0.85 | 0.20 |
| BCVA improvement (logMAR) | 0.75 | 0.95 | 0.82 | 0.37 |
Visual Function Questionnaire
The mean NEI VFQ-25 composite score of the 11 subscores improved from 37.6 ± 16.0 to 58.4 ± 17.4 ( P = .000001). Significant improvement was found in all 11 subscores, except for the score for driving ability ( Table 3 ). Significant improvement was found not only in the vision-related subscores, but also in the behavioral subscores. It should be emphasized that the subscore for mental health (ie, patient well-being) was very low in group 1, but greatly improved after using the limbal CL.
| Before CL Fitting | After CL Fitting | Mean Change | P Value a | |
|---|---|---|---|---|
| Composite score 11 | ||||
| Mean ± SD | 37.6 ± 16.0 | 58.5 ± 17.4 | 20.8 ± 15.8 | .000001 |
| Median (range) | 35.2 (8.4 to 69.4) | 58.4 (23.2 to 92.7) | 21.6 (–17.2 to 59.7) | |
| Composite score 7 | ||||
| Mean ± SD | 35.7 ± 16.0 | 58.4 ± 17.6 | 22.7 ± 17.6 | .000001 |
| Median (range) | 35.1 (4.3 to 67) | 57.2 (17.0 to 93.9) | 20.7 (–17.4 to 67) | |
| Subscale scores | ||||
| General health | ||||
| Mean ± SD | 47.4 ± 21.2 | 58.8 ± 15.1 | 11.4 ± 21.6 | .006265 |
| Median (range) | 50 (0 to 100) | 60 (5 to 85) | 0 (–27.5 to 65) | |
| Vision-related subscales | ||||
| General vision | ||||
| Mean ± SD | 32.6 ± 15.8 | 65.1 ± 20.3 | 32.6 ± 22.6 | .000001 |
| Median (range) | 30 (0 to 75) | 70 (0 to 100) | 35 (–15 to 75) | |
| Near vision | ||||
| Mean ± SD | 31.1 ± 17.0 | 53.1 ± 21.7 | 22 ± 19.9 | .000007 |
| Median (range) | 29.2 (0 to 66.7) | 54.2 (8.3 to 100) | 22.5 (–16.7 to 66.7) | |
| Distance vision | ||||
| Mean ± SD | 29.9 ± 17.2 | 53.4 ± 18.3 | 23.5 ± 18.7 | .000003 |
| Median (range) | 33.3 (0 to 62.5) | 50 (12.5 to 95) | 25 (–12.5 to 60) | |
| Color vision | ||||
| Mean ± SD | 62.9 ± 24.2 | 77.1 ± 21.0 | 14.3 ± 24.1 | .003496 |
| Median (range) | 75 (0 to 100) | 75 (0 to 100) | 0 (–50 to 75) | |
| Peripheral vision | ||||
| Mean ± SD | 33.1 ± 24.0 | 50.0 ± 23.9 | 16.9 ± 25.5 | .000447 |
| Median (range) | 25 (0 to 100) | 50 (0 to 100) | 25 (–75 to 75) | |
| Ocular pain | ||||
| Mean ± SD | 43.9 ± 29.9 | 65.7 ± 25.9 | 21.8 ± 24.7 | .000061 |
| Median (range) | 50 (0 to 100) | 75 (12.5 to 100) | 12.5 (–25 to 87.5) | |
| Behavioral subscales | ||||
| Mental health | ||||
| Mean ± SD | 28.9 ± 21.1 | 52.8 ± 22.8 | 23.9 ± 21.5 | .000003 |
| Median (range) | 25 (0 to 80) | 55 (10 to 95) | 20 (–15 to 75) | |
| Social function | ||||
| Mean ± SD | 51.8 ± 16.8 | 66.9 ± 17.5 | 15.1 ± 20.9 | .000661 |
| Median (range) | 50 (25 to 91.7) | 66.7 (33.3 to 100) | 16.7 (–25.0 to 58.3) | |
| Role limitation | ||||
| Mean ± SD | 36.3 ± 22.6 | 57.4 ± 20.9 | 21.2 ± 20.6 | .000009 |
| Median (range) | 37.5 (0 to 81.3) | 56.3 (0 to 100) | 18.8 (–18.8 to 68.8) | |
| Dependency | ||||
| Mean ± SD | 39.5 ± 25.6 | 60.4 ± 22.4 | 20.9 ± 21.7 | .000009 |
| Median (range) | 43.8 (0 to 87.5) | 62.5 (12.5 to 100) | 18.8 (–18.8 to 93.8) | |
| Ability to drive | ||||
| Driving | ||||
| Mean ± SD | 6.1 ± 18.7 | 14.6 ± 29.8 | 9.9 ± 20.5 | .278517 |
| Median (range) | 0 (0 to 75) | 0 (0 to 100) | 0 (0 to 75) |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


