Purpose
To review the indications and outcomes of children undergoing primary penetrating keratoplasty and identify prognostic factors for graft survival.
Design
Retrospective, interventional case series.
Methods
Medical records of every child 12 years of age or younger who underwent primary penetrating keratoplasty at Wills Eye Hospital Cornea Service between 2007 and 2015 were reviewed. Survival of the primary graft was analyzed using the Kaplan-Meier survival method.
Results
Forty-six eyes of 35 children underwent primary keratoplasty during the study period. The mean age at the time of primary keratoplasty was 24.6 ± 39.9 months. The mean follow-up duration was 36.4 ± 28.8 months. Congenital opacity was the most common diagnosis for primary keratoplasty (89.1%). The overall mean graft survival time was 45.2 ± 5.8 months, with a survival rate of 75.7% at 1 year. The 1-year graft survival rate was 51.9% and 90.7% in eyes with and without glaucoma, respectively. Cox proportional hazards regression analysis demonstrated that the presence of glaucoma ( P = .014) and concurrent operation during primary keratoplasty ( P = .049) were independent prognostic factors for poor graft survival. On the other hand, age of primary keratoplasty ( P = .626) and operation before or after primary keratoplasty ( P = .800 and P = .104, respectively) were not associated with poorer graft survival. Half of our patients were able to achieve ambulatory vision at the last follow-up.
Conclusion
Although pediatric penetrating keratoplasty is challenging, successful transplantation with good graft survival can be obtained. Better understanding of prognostic factors can possibly improve graft survival in the future.
Pediatric penetrating keratoplasty has been considered as high-risk keratoplasty and was not commonly performed until recently. With advancements in surgical technique, postoperative care, and knowledge of prognostic factors on graft survival, more corneal transplantations were performed and better outcomes were reported in pediatric patients. Nevertheless, there are many challenges unique to pediatric keratoplasty. Preoperative clinical assessment testing of visual function in preverbal children is often difficult. Small anatomic dimensions and decreased tissue rigidity require specialized surgical techniques. Congenital corneal opacity is the most common indication for keratoplasty in young children in developed countries. It is often associated with additional anterior segment abnormalities, which further increase the surgical complexities. Postoperative care; monitoring of complications such as graft rejection, glaucoma, and infection; amblyopia therapy; and management of associated ocular comorbidities are extremely demanding following pediatric keratoplasty.
The current study reviewed the indications and outcomes of children undergoing primary penetrating keratoplasty in a cornea referral center. Our goal was to identify prognostic factors for primary graft survival including age of transplantation, type of corneal opacity, presence of glaucoma, and ocular surgery performed prior, concurrent, or subsequent to primary keratoplasty.
Methods
This was a retrospective, interventional case series performed at the Wills Eye Hospital. The study protocol was approved by the Institutional Review Board and was conducted in accordance with the Declaration of Helsinki. Medical records of every child 12 years of age or younger who underwent primary penetrating keratoplasty at Wills Eye Hospital Cornea Service between 2007 and 2015 were reviewed. All patients had the informed consent signed by their parents or guardians. Eyes with primary keratoplasty performed in another hospital and lamellar keratoplasty were excluded.
Patient demographics; ocular diagnoses; associated ocular surgery that was performed prior, concurrent, or subsequent to primary keratoplasty; presence of glaucoma; postoperative complications; time to graft failure; number of repeat grafts; graft clarity at last visit; and visual acuity at presentation and last visit were extracted from medical records. For cases in which verbal or cognitive skills were inadequate to achieve accurate determination of visual acuity, the ability to fixate and follow targets was noted. Ambulatory vision was defined as vision of counting fingers at 3 feet or better. Graft failure was defined as the irreversible loss of central clarity, with date of onset of corneal clouding selected as time point of graft failure. Survival time of failed grafts was defined as the time between the date of surgery and recorded date of failure. For clear grafts or cases lost to follow-up, survival time was calculated as the interval between the date of surgery and the date of the last visit.
Surgical Technique
Surgical technique varied but included the use of Flieringa ring and interrupted 10-0 nylon sutures with 0.25–0.5 mm oversized donor diameter in most cases. Examinations under anesthesia were performed when adequate examination was not feasible in clinic. All patients were under collaborative care of corneal, glaucoma, and pediatric specialists.
Statistical Analysis
Statistical analyses were performed using the IBM/SPSS software version 21 (IBM/SPSS Inc, Chicago, Illinois, USA). Descriptive statistics was reported. Survival of the primary grafts was analyzed using the Kaplan-Meier survival method. Log-rank test was used to evaluate differences in survival between groups. Cox proportional hazards regression analysis was performed to investigate the simultaneous effects of a subset of variables on graft survival. P < .05 was considered statistically significant.
Results
A total of 59 penetrating keratoplasties (primary and repeat grafts) were performed at Wills Eye Hospital during the study period and were included in the current study. Two eyes were not included because 1 had primary keratoplasty in another hospital and 1 underwent lamellar keratoplasty. Forty-six eyes of 35 children (16 male, 19 female) underwent primary keratoplasty during the study period. Nine eyes (19.6%) had repeat keratoplasty performed with a mean of 2.4 transplantations (2–4 grafts). One 1 had a keratoprothesis after multiple graft failure. The mean age at the time of primary keratoplasty was 24.6 ± 39.9 months (range, 2 months to 12 years). Eleven patients (31.4%) had bilateral corneal transplantation. The mean follow-up duration was 36.4 ± 28.8 months (range, 4.5 months to 8.5 years). Congenital opacity was the most common diagnosis category for primary keratoplasty (89.1%), with Peters anomaly being the most common diagnosis (70.7%) in the group ( Table 1 ).
| Preoperative Diagnosis | Number of Eyes (n = 46) | % |
|---|---|---|
| Congenital opacity | ||
| Peters anomaly | 29 | 63.0 |
| Congenital glaucoma | 7 | 15.2 |
| Corneal dystrophy | 2 | 4.3 |
| “Sclerocornea” | 2 | 4.3 |
| Others | 1 | 2.2 |
| Acquired nontraumatic | ||
| Corneal ulcer | 3 | 6.5 |
| Acquired traumatic | ||
| Corneal laceration scar | 2 | 4.3 |
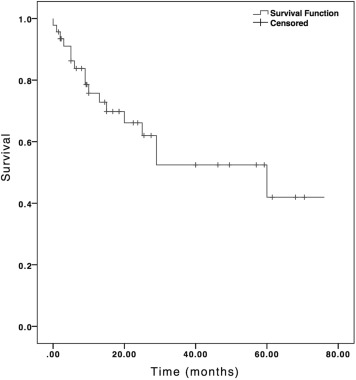
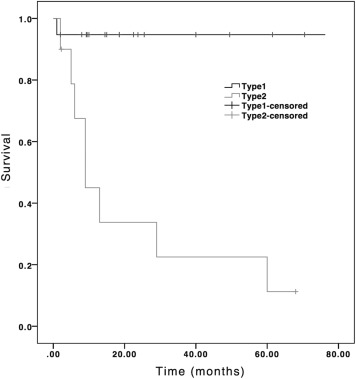
The overall mean graft survival time of the 46 primary penetrating keratoplasties was 45.2 ± 5.8 months (95% confidence interval [CI], 33.9–56.6 months), with a survival rate of 75.7% at 1 year ( Figure 1 ). For the 29 eyes (63.0%) with Peters anomaly, 19 were type I and 10 were type II disease. Type I Peters anomaly is a milder form characterized by a central corneal opacity with iris strands extending across the anterior chamber to cornea, whereas type II is a more severe form characterized by additional lens abnormalities, such as cataract or corneolenticular adhesion. The mean graft survival time was 73.0 ± 5.5 months (95% CI, 62.2–83.8 months) for type I and 22.6 ± 7.9 months (95% CI, 7.2–38.0 months) for type II ( P < .001). The 1-year graft survival rate was 94.7% for type I and 45.0% for type II Peters anomaly ( Figure 2 ).
Glaucoma was present in 21 eyes (45.7%). Eleven of those eyes (52.3%) had glaucoma diagnosed before first transplantation and 10 eyes (47.6%) developed glaucoma postoperatively. Graft survival of eyes without glaucoma was significantly higher than eyes with glaucoma ( P = .021). The 1-year graft survival rate was 51.9% in eyes with glaucoma and 90.7% in eyes without glaucoma ( Figure 3 ). Ocular surgery prior to primary keratoplasty was performed in 7 eyes (15.2%) and included goniotomy, trabeculotomy, trabeculectomy, glaucoma drainage device, and cyclophotocoagulation ( Table 2 ). There was no statistically significant difference in graft survival between eyes with or without prior operation ( P = .303), although the corresponding 1-year graft survival rate was 45.7% and 80.2%, respectively ( Figure 4 ). Concurrent procedures at the time of penetrating keratoplasty, including release of synechiae, pupilloplasty, lensectomy, anterior vitrectomy, and cyclophotocoagulation, were performed in 26 eyes (56.5%) ( Table 2 ). A trend toward better graft survival was seen in eyes without concurrent procedure at the time of transplantation ( P = .052). The 1-year graft survival rate was 64.1% and 88.8% for eyes with and without concurrent procedure, respectively ( Figure 4 ). Subsequent ocular surgery was performed after the first graft in 12 eyes (26.1%) ( Table 2 ). Similar graft survival was observed between eyes with or without subsequent surgery ( P = .928). The corresponding 1-year graft survival rate was 72.7% and 77.5%, respectively ( Figure 4 ).
| Number of Eyes (n = 26) a | % | |
|---|---|---|
| Prior to First Transplantation | ||
| Glaucoma drainage device | 3 | 6.5 |
| Goniotomy | 2 | 4.3 |
| Trabeculotomy | 2 | 4.3 |
| Trabeculectomy | 2 | 4.3 |
| Cyclophotocoagulation | 2 | 4.3 |
| Concurrent to first transplantation | ||
| Lensectomy | 14 | 30.4 |
| Anterior vitrectomy | 12 | 26.1 |
| Peripheral iridectomy | 10 | 19.5 |
| Release of synechiae | 7 | 15.2 |
| Pupilloplasty | 3 | 4.3 |
| Cyclophotocoagulation | 1 | 2.2 |
| Subsequent to first transplantation | ||
| Glaucoma drainage device | 5 | 10.9 |
| Cataract extraction | 4 | 8.7 |
| Strabismus surgery | 3 | 6.5 |
| Revision of glaucoma drainage device | 1 | 2.2 |
| Retinal detachment surgery | 1 | 2.2 |
Cox proportional hazards regression analysis demonstrated that the presence of glaucoma ( P = .014) and concurrent operation during primary keratoplasty ( P = .049) were independent prognostic factors for poor graft survival. On the other hand, age of primary keratoplasty ( P = .626) and operation before or after primary keratoplasty ( P = .800 and P = .104, respectively) were not associated with poorer graft survival.
Clear graft was present in 35 eyes (75.1%) at the last follow-up. Visual acuities at last follow-up visit are shown in Table 3 . Only 23.9% of eyes had ambulatory vision before keratoplasty. After corneal transplantation, 52.1% of eyes of were able to achieve ambulatory vision at the last follow-up. Major postoperative complications occurred in 18 eyes (39.1%). These included glaucoma, cataract, phthisis, ocular surface disease, and retinal detachment ( Table 4 ).



