Purpose
To compare superior vs temporal approach in patients who underwent Descemet membrane endothelial keratoplasty (DMEK).
Design
Monocentric, prospective nonmasked study.
Methods
A prospective analysis of 53 DMEKs between January and September 2013 was performed at the Charité–Universitätsmedizin Berlin. Only DMEK cases with an incision size of 2.3 mm and with at least 1 month of follow-up were included. The surgically induced astigmatism (SIA), changes in corneal aberrations and in spherical equivalent, visual acuity, endothelial cell density, and complications were evaluated.
Results
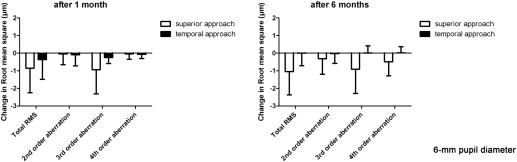
Visual acuity improved significantly (0.70 ± 0.39 logMAR vs 0.32 ± 0.31 logMAR after 1 month (n = 48), 0.19 ± 0.15 logMAR after 3 months (n = 46), and 0.16 ± 0.17 logMAR after 6 months (n = 47) ( P < .001)) regardless of the approach. SIA was significantly lower after temporal than after superior approach (1.42 ± 0.91 diopters [D] [n = 13] vs 0.81 ± 0.68 D [n = 13], P = .038). Change in total root mean square of all aberrations (RMS) ( P = .046) at 6 mm pupil diameter, and change in total RMS ( P = .019), third-order aberrations ( P = .007), and fourth-order aberrations ( P = .041) at 4 mm pupil diameter, demonstrated significantly lower results after temporal compared to superior approach. A higher rate of eyes after temporal approach underwent at least 1 rebubbling (39.1% vs 26.7%, P = .252). The endothelial cell density ( P = .053) and the change in spherical equivalent ( P = .145) did not differ significantly.
Conclusions
The temporal approach induces significantly less SIA and corneal aberration. There are no significant differences between superior and temporal approach according to the change in spherical equivalent, visual acuity, and endothelial cell density. The need for rebubbling is higher using the temporal approach.
Corneal endothelial disorders like Fuchs endothelial dystrophy and bullous keratopathy were mainly treated with Descemet stripping endothelial keratoplasty (DSEK) or Descemet membrane endothelial keratoplasty (DMEK) in past years. In comparison to penetrating keratoplasty (PK), main advantages of these lamellar surgical techniques include rapid healing, more predictable refractive outcomes, better corneal integrity, and faster functional rehabilitation.
DMEK is performed increasingly in cases of endothelial dysfunction, with promising results. This less invasive corneal transplant technique was first described by Melles and associates. It allows the transplantation of an isolated endothelium Descemet membrane layer without adherent corneal stroma.
In analogy to the development of phacoemulsification, it is of great interest to improve the surgical technique of DMEK to optimize the functional results. Improved surgical techniques for phacoemulsification during the past years led to a decreased postoperative corneal astigmatism, fewer postoperative complications, a shorter recovery, and an improved visual acuity. As described by many studies, the type and length of incisions as well as suture techniques may affect surgically induced astigmatism (SIA) and, therefore, the visual outcome. The effect of superior compared with temporal clear corneal incision (2.8–4 mm) was evaluated by different studies. Results showed a low surgically induced astigmatism after temporal corneal incision and a higher astigmatism after superior corneal incision.
In this study, we prospectively evaluated the visual and refractive outcome, the surgically induced astigmatism, the low- (LOAs) and high-order aberrations (HOAs), and the complication rates after superior vs temporal approach in patients who underwent DMEK surgery.
Patients and Methods
Patients
Between January 23, 2013 and September 24, 2013, 135 DMEK surgeries were performed at the Department of Ophthalmology, Charité–Universitätsmedizin Berlin, Campus Virchow Klinikum by an experienced surgeon (N.T.). For this study, only DMEK cases with an incision size of 2.3 mm and with at least 1 month of follow-up were included. In total, 53 DMEKs of 135 performed procedures were analyzed in this prospective study. Demographic data are presented in Table 1 .
| Superior Approach (12 o’clock) n = 30 | Temporal Approach (3 or 9 o’clock) n = 23 | Total n = 53 | |
|---|---|---|---|
| Female:male | 14:16 | 11:12 | 25:28 |
| Age (y, mean ± SD) | 70.5 ± 9.0 | 70.3 ± 8.5 | 70.4 ± 8.7 |
| Reason for Descemet membrane endothelial keratoplasty | |||
| % Fuchs endothelial dystrophy | 86.7% (n = 26) | 100.0% (n = 23) | 92.5% (n = 49) |
| % bullous keratopathy | 10.0% (n = 3) | 0.0% (n = 0) | 5.7% (n = 3) |
| % after Descemet stripping endothelial keratoplasty | 3.3% (n = 1) | 0.0% (n = 0) | 1.9% (n = 1) |
| Surgery | |||
| Descemet membrane endothelial keratoplasty | 46.7% (n = 14) | 26.1% (n = 6) | 37.7% (n = 20) |
| Triple procedure (Descemet membrane endothelial keratoplasty, phacoemulsification, and intraocular lens implantation) | 53.3% (n = 16) | 73.9% (n = 17) | 62.3% (n = 33) |
| Preoperative visual acuity (logMAR, mean ± SD) | 0.77 ± 0.44 (n = 30) | 0.61 ± 0.31 (n = 23) | 0.70 ± 0.39 (n = 53) |
This study followed the ethical standards of the Declaration of Helsinki. The patients gave informed consent for the treatment and for participation in the study. Institutional ethical approval was obtained by the Ethics Committee of the Charité–Universitätsmedizin Berlin (EA2/108/12) for this prospective study and clinical trial registration was performed ( http://www.clinicaltrials.gov , NCT02020044). HIPAA compliance was confirmed.
Preoperative and Postoperative Evaluation
Preoperative and postoperative examinations were performed 1 day before the operation as well as 1, 3, and 6 months after DMEK. Examinations included best-corrected visual acuity (BCVA), slit-lamp examination, applanation tonometry (Goldmann applanation tonometer; Haag Streit, Bern, Switzerland) or pneumatic tonometer (CT20D computerized tonometer; Topcon, Tokyo, Japan), autorefraction (KR-8900 Auto Kerato-Refractometer; Topcon Medical Systems Inc, Oakland, California, USA), and funduscopy. Distance visual acuity was tested with a Snellen chart and expressed as a Snellen decimal number. The Snellen decimal number was converted to logMAR by a visual acuity conversion table. Additionally, data on endothelial cell density (NONCON-ROBO CA specular microscope; Konan Medical Inc, Nishinomiya, Japan) were collected. The spherical equivalent was measured after subjective dioptometry. Only patients without additional cataract were included. The change in spherical equivalent was calculated as difference between postoperative and preoperative spherical equivalent. Corneal topography, astigmatism, LOAs, and HOAs (total root mean square of all aberrations [RMS], and second-, third-, and fourth-order aberrations) were measured using the Galilei dual Scheimpflug analyzer (V5.2.1; Ziemer Ophthalmic Systems AG, Port, Switzerland). SIA was calculated using the data determined by vector analysis. Through analysis of the change in LOAs and HOAs, the induced aberrations were calculated as the difference between postoperative and preoperative aberrations.
Graft and Surgical Technique
All patients received cultured grafts from the Cornea Bank Berlin. The minimum central endothelial cell density for transplantation grafts was 2000 cells/mm 2 .
Stripping of the Descemet membrane from the donor corneal stroma was performed immediately prior to transplantation in a standardized manner described in detail by Melles and associates. The diameter of the graft was between 8.0 mm and 9.0 mm. In all cases clear corneal incisions, 2.3 mm incision size, slit knife MSL23 (Mani, Inc, Toshihide Takai, Japan) without sutures were used. The clear corneal incision was made at the steeper corneal meridian. Descemetorrhexis was performed with a diameter between 8.5 and 9.0 mm. In cases of additional cataract a combined procedure (triple DMEK) with DMEK following standard cataract surgery was performed in 33 of 53 included eyes. A glass injector was used to insert the graft lamella. To attach the graft lamella the complete anterior chamber was filled with air and the air was left for 1 hour. After 1 hour, part of the air was released and exchanged with balanced saline solution, so that the anterior chamber was half filled with air.
In the following 2 weeks after surgery additional air injection (rebubbling) after surgery was performed in eyes with a combination of detachment of the graft lamella detected with the Spectralis OCT device (Heidelberg Engineering GmbH, Heidelberg, Germany) with an anterior chamber attachment, and focal corneal edema over more than 2 clock hours.
Standard postoperative topical treatment included prednisolone acetate 1% (5 times daily) and lubricant eye drops (5 times daily) and a combined antibiotic and steroid ointment at night. After 1 week, the ointment was stopped and prednisolone acetate 1% was used topically (5 times daily for the first month) with lubricant eye drops 5 times daily. The prednisolone acetate 1% was tapered down over a period of 9 months.
Statistical Methods
Statistical analysis was performed using IBM SPSS statistics 19 (SPSS Software, Munich, Germany). Normality was tested for all outcome measures. None of the measures followed a normal distribution. Therefore, nonparametric tests (Mann-Whitney U test, Wilcoxon signed rank test) were used for analysis. Descriptive statistics were expressed as median and range between minimum and maximum or mean ± standard deviation (SD). Log-rank test was used to evaluate statistical significance in the subdivided groups. A χ 2 distribution was used to analyze the distribution of proportions. If P values were less than .05, differences were considered as being statistically significant.
Results
One hundred thirty-five DMEK procedures were performed during the study period. Fifty-three of these 135 eyes fulfilled the study criteria. In 30 eyes a superior approach (12 o’clock) and in 23 eyes a temporal approach (3 or 9 o’clock) was chosen. In 33 of 53 included eyes a combined procedure (triple DMEK) with DMEK following standard cataract surgery was performed. The mean follow-up time was 170.8 ± 48.2 days.
Table 1 summarizes the demographic, surgical, and preoperative results for both groups. The data are not complete, as some patients missed follow-ups. Further, the measurement of endothelial cell density and the autorefractor were not always possible. If the preoperative astigmatism and the aberrations were not measurable using the Galilei dual Scheimpflug analyzer, surgically induced astigmatism and changes of aberrations could not be calculated for follow-ups.
Visual Acuity and Refractive Results
Data of visual acuity after superior and after temporal approach are presented in Table 2 . Wilcoxon signed rank test indicated a statistically significant improvement in mean visual acuity after 1, 3, and 6 months compared to preoperative visual acuity (0.70 ± 0.39 logMAR vs 0.32 ± 0.31 logMAR after 1 month [n = 48], 0.19 ± 0.15 logMAR after 3 months [n = 46], and 0.16 ± 0.17 logMAR after 6 months [n = 47], P < .001). There was no significant difference in the visual acuity between superior and temporal approach at any time ( P = .132, P = .173, P = .582, P = .766, Table 2 ).
| Superior Approach (12 o’clock) n = 30 | Temporal Approach (3 or 9 o’clock) n = 23 | Total n = 53 | P Values a | |
|---|---|---|---|---|
| Visual acuity (logMAR) | ||||
| Preoperative | 0.77 ± 0.44 (n = 30) | 0.61 ± 0.31 (n = 23) | 0.70 ± 0.39 (n = 53) | .132 |
| After 1 month | 0.30 ± 0.33 (n = 27) | 0.34 ± 0.29 (n = 21) | 0.32 ± 0.31 (n = 48) | .173 |
| After 3 months | 0.20 ± 0.18 (n = 26) | 0.16 ± 0.11 (n = 20) | 0.19 ± 0.15 (n = 46) | .582 |
| After 6 months | 0.16 ± 0.19 (n = 26) | 0.16 ± 0.14 (n = 21) | 0.16 ± 0.17 (n = 47) | .766 |
| Change in spherical equivalent b (D) | ||||
| After 1 month | −0.80 ± 2.12 (n = 7) | −0.25 ± 0.27 (n = 5) | −0.57 ± 1.60 (n = 12) | .807 |
| After 3 months | −0.09 ± 0.51 (n = 10) | −0.46 ± 0.79 (n = 6) | −0.23 ± 0.63 (n = 16) | .548 |
| After 6 months | −1.61 ± 1.90 (n = 8) | −0.19 ± 0.67 (n = 4) | −1.14 ± 1.70 (n = 12) | .145 |
| Surgically induced astigmatism (D) | ||||
| After 1 month | 1.47 ± 1.02 (n = 14) | 0.84 ± 0.40 (n = 15) | 1.15 ± 0.82 (n = 29) | .089 |
| After 6 months | 1.42 ± 0.91 (n = 13) | 0.81 ± 0.68 (n = 13) | 1.11 ± 0.85 (n = 26) | .038 |
| Endothelial cell density (cells/mm 2 ) | ||||
| Preoperative | 2316 ± 201 (n = 29) | 2268 ± 222 (n = 21) | 2296 ± 210 (n = 50) | .284 |
| After 1 month | 1795 ± 395 (n = 12) | 1708 ± 451 (n = 9) | 1753 ± 416 (n = 23) | .498 |
| After 3 months | 1757 ± 498 (n = 18) | 1836 ± 397 (n = 7) | 1794 ± 448 (n = 34) | .756 |
| After 6 months | 1753 ± 358 (n = 17) | 2024 ± 280 (n = 11) | 1859 ± 351 (n = 28) | .053 |
a P values indicate significant differences between the superior and temporal approach.
b Change in spherical equivalent was analyzed only for the patients after Descemet membrane endothelial keratoplasty without additional cataract surgery.
Table 2 presents the mean change in spherical equivalent of DMEK cases without additional cataract surgery at different times. The mean change in spherical equivalent after 6 months was −1.14 ± 1.70 diopters [D] (n = 12). Mean change in spherical equivalent did not differ significantly between superior and temporal approach at any time.
Surgically induced astigmatism was lower after temporal approach than after superior approach after 1 month and significantly lower after 6 months ( P = .038) ( Table 2 ).
When examining the subgroups DMEK and triple DMEK, there was no significant difference in surgically induced astigmatism and visual acuity.
Corneal Aberrations
The data of wavefront analysis of changes in LOA and HOA (corneal wavefront Zernike coefficients) at 6 mm pupil diameter are shown in Figure 1 , and at 4 mm pupil diameter in Figure 2 . Data of 6 eyes were excluded owing to low quality of aberration measurements.