Purpose
To evaluate and compare the regional relationships between visual field sensitivity and retinal nerve fiber layer (RNFL) thickness as measured by spectral-domain optical coherence tomography (OCT) and scanning laser polarimetry.
Design
Prospective cross-sectional study.
Methods
One hundred and twenty eyes of 120 patients (40 with healthy eyes, 40 with suspected glaucoma, and 40 with glaucoma) were tested on Cirrus-OCT, GDx VCC, and standard automated perimetry. Raw data on RNFL thickness were extracted for 256 peripapillary sectors of 1.40625 degrees each for the OCT measurement ellipse and 64 peripapillary sectors of 5.625 degrees each for the GDx VCC measurement ellipse. Correlations between peripapillary RNFL thickness in 6 sectors and visual field sensitivity in the 6 corresponding areas were evaluated using linear and logarithmic regression analysis. Receiver operating curve areas were calculated for each instrument.
Results
With spectral-domain OCT, the correlations (r 2 ) between RNFL thickness and visual field sensitivity ranged from 0.082 (nasal RNFL and corresponding visual field area, linear regression) to 0.726 (supratemporal RNFL and corresponding visual field area, logarithmic regression). By comparison, with GDx-VCC, the correlations ranged from 0.062 (temporal RNFL and corresponding visual field area, linear regression) to 0.362 (supratemporal RNFL and corresponding visual field area, logarithmic regression). In pairwise comparisons, these structure–function correlations were generally stronger with spectral-domain OCT than with GDx VCC and with logarithmic regression than with linear regression. The largest areas under the receiver operating curve were seen for OCT superior thickness (0.963 ± 0.022; P < .001) in eyes with glaucoma and for OCT average thickness (0.888 ± 0.072; P < .001) in eyes with suspected glaucoma.
Conclusions
The structure–function relationship was significantly stronger with spectral-domain OCT than with scanning laser polarimetry, and was better expressed logarithmically than linearly. Measurements with these 2 instruments should not be considered to be interchangeable.
Glaucoma is an optic neuropathy characterized by an accelerated loss of retinal ganglion cells. This results in a diffuse or local thinning of the retinal nerve fiber layer and eventually in loss of visual function. Automated perimetry is a well-established, highly reproducible, and accurate method for evaluating functional loss, but evaluation of structural changes has until recently been limited to qualitative analyses of the retinal nerve fiber layer and optic nerve head. Imaging devices introduced in the last decade now allow more quantitative measurement of the retinal nerve fiber layer, with optical coherence tomography (OCT) and scanning laser polarimetry both allowing direct measurement of the thickness of the retinal nerve fiber layer. Recent studies have reported significant correlations between visual field sensitivity and peripapillary retinal nerve fiber layer thickness evaluated with these 2 imaging methods. Some of these studies have evaluated and compared the strength of the associations between visual function and structure as assessed by time-domain OCT vs scanning laser polarimetry, and have generally reported that these associations were strongest with time-domain OCT measurements. Because of the fast Fourier transform algorithm and the high frame transfer rate, spectral-domain OCT can perform up to 27 000 axial scans per second, with an axial resolution of about 5 μm, whereas commercially available time-domain OCT has a velocity of 400 axial scans per second with a 10-μm axial resolution. Time- and spectral-domain OCT have different algorithms for retinal nerve fiber layer delineation, and thus their thickness measurements may have different associations with visual function.
The purpose of this study was to evaluate and compare the regional relationships between visual field sensitivity and retinal nerve fiber layer thickness as measured by spectral-domain OCT and scanning laser polarimetry. In addition, we evaluated and compared the abilities of spectral-domain OCT and scanning laser polarimetry to distinguish glaucomatous or suspected glaucomatous eyes from healthy eyes.
Methods
Patients
This was a prospective investigation conducted in a French university-affiliated glaucoma center. One eye was selected randomly from each of 120 subjects: 40 with glaucoma, 40 with suspected glaucoma, and 40 healthy subjects. All study participants underwent a full ophthalmic examination, including objective and subjective refraction, slit-lamp biomicroscopy, intraocular pressure measurement with Goldmann tonometry, gonioscopy, dilated fundus examination by indirect ophthalmoscopy, central corneal thickness measurement, and A-scan ultrasound biometry. Refraction was performed using an AR-360 autorefractometer (Nidek Co, Gamagori, Japan), gonioscopy using a Goldmann 3-mirror lens, and pachymetry and A-scan ultrasound biometry using an OcuScan RxP (Alcon Inc, Forth Worth, Texas, USA).
Inclusion criteria were age ≥18 years, best-corrected visual acuity better than or equal to 20/40, spherical refraction between −6.00 and +3.00 diopters, open angles on gonioscopy, no retinal disease or nonglaucomatous neuropathy, and no intraocular surgery except for uncomplicated cataract surgery. Glaucomatous eyes were defined as those with consecutive and reliable abnormal standard automated perimetry with abnormal Glaucoma Hemifield Test and pattern standard deviation outside 95% of normal limits, and optic nerve damage (asymmetric cup-to-disc ratio >0.2, rim thinning, notching, excavation, or retinal nerve fiber layer defect). Suspected glaucomatous eyes were defined as those with optic nerve damage (asymmetric cup-to-disc ratio >0.2, rim thinning, notching, excavation, or retinal nerve fiber layer defect) without repeatable abnormal standard automated perimetry results. Healthy eyes had an intraocular pressure <22 mm Hg by Goldmann applanation tonometry, normal-appearing optic discs, and no repeatable abnormal standard automated perimetry results.
Measurements
White-on-white standard automated perimetry was performed using a Humphrey Visual Field Analyzer (Zeiss-Humphrey Systems, Dublin, California, USA) using the C-24-2 SITA–standard strategy. A reliable visual field test was defined as having a <25% rate of fixation losses, and <20% false positive and false negative. The 2 points adjacent to the blind spot were excluded from the analysis. The 52 remaining points were grouped into 6 sectors, based on the topographic relationships between visual field locations and corresponding regions of the optic disc, as previously described by Garway-Heath and associates ( Figure 1 ). The mean threshold sensitivity of the 6 sectors was calculated by averaging the threshold sensitivities of each test point within the visual field region.
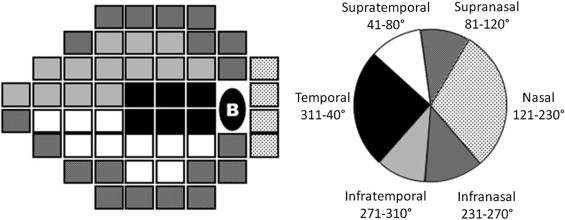
A commercially available spectral-domain OCT (Cirrus OCT, Carl Zeiss Meditec, Inc, Dublin, California, USA; software version 3.0) was used in the study. Imaging was performed using the 200 × 200 protocol optic disc cube (200 horizontal scan lines, each of 200 A-scans). Only well-focused, well-centered images, without eye movement and with a signal strength of 7/10 or more, were used. Parameters of the printout retained for the analysis were inferior, superior, nasal, temporal, average thickness, inter-eye symmetry, and retinal nerve fiber layer thickness for each of the 12 30-degree sectors. We also collected the 256 retinal nerve fiber layer thicknesses of the temporal-superior-nasal-inferior-temporal (TSNIT) curve; these corresponded to the thicknesses extracted from the 256 A-scans along the path of the 3.46-mm calculation circle located around the optic disc. The Cirrus OCT software has the facility to scroll a cursor along the TSNIT curve, making the 256 values appear successively. The screen recording software Camstudio ( http://camstudio.org/ ; accessed March 16, 2009) was used to record screen activity and create video files. Data were exported to a personal computer. The optical character recognition software ABBYY FineReader v 9.0 (Avanquest Software, La Garenne Colombes, France) was then used to extract the 256 retinal nerve fiber layer thicknesses of the TSNIT curve from the video sequences. These 256 values were grouped into 6 sectors, based on the relation between visual field and regions of the optic disc as described above: infranasal (231-270 degrees), infratemporal (271-310 degrees), temporal (311-40 degrees), supratemporal (41-80 degrees), supranasal (81-120 degrees), and nasal (121-230 degrees).
Scanning laser polarimetry measurements were performed using GDx variable corneal compensation (GDx VCC, Carl Zeiss Meditec, Inc; software version 5.5.0). Only well-focused, well-centered images with a quality scan score ≥8 were used. GDx VCC parameters of the printout retained for the analysis were inferior, superior, and TSNIT averages; TSNIT standard deviation; inter-eye symmetry; and nerve fiber indicator. We also extracted GDx VCC raw data to obtain the retinal nerve fiber layer thickness of 64 peripapillary 5.625-degree sectors under the instrument’s measurement ellipse. These 64 values were then grouped into 6 sectors as described above: infranasal (231-270 degrees), infratemporal (271-310 degrees), temporal (311-40 degrees), supratemporal (41-80 degrees), supranasal (81-120 degrees), and nasal (121-230 degrees). The 5.625-degree GDx sectors that covered 2 of the 6 optic disc sectors were distributed between the adjacent 2 sectors, with a weighting taking into account the overlap of the 2 adjacent areas.
Statistical Analyses
Independent-sample t tests and Mann-Whitney tests were used to compare means and percentages. Linear ( y = a + bx ) and logarithmic ( y = a + b .ln( x )) regression analysis were performed to evaluate the relationship between visual field sensitivity and retinal nerve fiber layer thickness in the 6 matching areas. Both linear and logarithmic regressions were used, because visual field sensitivity is expressed in logarithmic units (decibels). The statistical significance of r was tested using a t test. A 2-tailed paired t test was used to compare the strength of the association across instruments and linear or logarithmic regressions. The area under the receiver operator characteristic curve was used to assess the ability of each OCT or GDx parameter to differentiate between normal and suspected glaucomatous eyes, and between normal and glaucomatous eyes. An area under the receiver operator characteristic curve of 0.5 represents discrimination no better than chance; an area of 1 represents perfect discrimination. Differences between areas under the receiver operator characteristic curve among parameters were determined using the nonparametric method described by DeLong and associates. Statistical significance was set at P ≤ .05. SPSS statistical software version 17.0 (SPSS Inc, Chicago, Illinois, USA) was used for analyses.
Results
Patient Characteristics
One hundred and sixty-six patients were screened, 14 (8.4%) of whom were excluded for poor OCT quality criteria, 23 (13.9%) for poor GDx VCC quality criteria, and 28 (16.9%) for poor visual field quality or reliability criteria. Forty patients with glaucoma, 40 patients with suspected glaucoma, and 40 healthy volunteers were enrolled. Their demographic, biometric, and visual field data are shown in Table 1 .
| Glaucoma (n = 40) | Suspected Glaucoma (n = 40) | Healthy (n = 40) | P ( t Test) | |
|---|---|---|---|---|
| Age (years), mean ± SD | 63.4 ± 11.2 | 61.7 ± 12.7 | 60.9 ± 13.1 | >.1 |
| Gender (male/female) | 14/26 | 15/25 | 17/23 | >.1 |
| Spherical equivalent (D), mean ± SD | −0.90 ± 2.2 | −0.37 ± 1.7 | −0.64 ± 2.1 | >.1 |
| CCT (μm), mean ± SD | 528 ± 33 | 535 ± 34 | 538 ± 29 | >.1 |
| Axial length (mm), mean ± SD | 23.38 ± 1.12 | 23.11 ± 1.07 | 23.59 ± 1.27 | >.1 |
| Standard automated perimetry (dB) | ||||
| Mean deviation ± SD | −9.88 ± 6.93 | −1.73 ± 2.16 | −0.73 ± 1.56 | <.01 |
| Pattern standard deviation ± SD | 4.42 ± 4.85 | 2.06 ± 0.54 | 1.24 ± 1.28 | <.05 |
Structure–Function Relationships
Linear and logarithmic regression parameters are presented in Table 2 (retinal nerve fiber layer thickness measured by Cirrus OCT) and Table 3 (retinal nerve fiber layer thickness measured by GDx VCC). Analyses showed a significant relationship between measured retinal nerve fiber layer thickness and visual field sensitivity for most studied areas. Correlations (r 2 ) ranged from 0.082 (nasal retinal nerve fiber layer and corresponding visual field area, linear regression) to 0.726 (supratemporal retinal nerve fiber layer and corresponding visual field area, logarithmic regression) with Cirrus OCT, and from 0.062 (temporal retinal nerve fiber layer and corresponding visual field area, linear regression) to 0.362 (supratemporal retinal nerve fiber layer and corresponding visual field area, logarithmic regression) with GDx VCC. Linear and logarithmic associations were strongest between the supratemporal retinal nerve fiber layer and corresponding visual field areas, and between the infratemporal retinal nerve fiber layer and corresponding visual field areas, for both the Cirrus OCT and the GDx VCC ( Figure 2 ). Except for the temporal retinal nerve fiber layer sector, logarithmic associations were significantly stronger than linear associations for both the Cirrus OCT and the GDx VCC measurements ( P < .05). In pairwise comparisons, both linear and logarithmic structure–function associations were stronger with the Cirrus OCT measurement than with the GDx VCC measurements ( P < .01), except for the retinal nerve fiber layer nasal sector.
| Peripapillary Sector | Linear Regression | Logarithmic Regression | ||||
|---|---|---|---|---|---|---|
| r 2 | P | y = a*x + b | r 2 | P | y = a*ln(x) + b | |
| Temporal | 0.246 | <.001 | y = 0.17x + 16.4 | 0.224 | <.001 | y = 8.16ln(x) − 10.8 |
| Supratemporal | 0.644 | <.001 | y = 0.16x + 3.82 | 0.726 | <.001 | y = 13.7ln(x) − 38.8 |
| Supranasal | 0.321 | <.001 | y = 0.11x + 13.4 | 0.387 | <.001 | y = 11.4ln(x) − 32.1 |
| Nasal | 0.082 | >.5 | y = 0.11x + 17.5 | 0.096 | >.5 | y = 8.78ln(x) − 8.71 |
| Infranasal | 0.437 | <.001 | y = 0.19x + 6.80 | 0.548 | <.001 | y = 12.4ln(x) − 38.7 |
| Infratemporal | 0.602 | <.001> | y = 0.17x + 5.42 | 0.648 | <.001 | y = 14.8ln(x) − 40.3 |
| Peripapillary Sector | Linear Regression | Logarithmic Regression | ||||
|---|---|---|---|---|---|---|
| r 2 | P | y = a*x + b | r 2 | P | y = a*ln(x) + b | |
| Temporal | 0.062 | >.5 | y = 0.17x + 14.5 | 0.086 | =.001 | y = 9.12ln(x) − 15.6 |
| Supratemporal | 0.288 | <.001 | y = 0.17x + 10.5 | 0.362 | <.001 | y = 12.4ln(x) − 32.3 |
| Supranasal | 0.164 | <.001 | y = 0.12x + 12.6 | 0.186 | <.001 | y = 10.3ln(x) − 29.7 |
| Nasal | 0.088 | >.5 | y = 0.13x + 16.4 | 0.104 | >.5 | y = 6.68ln(x) − 10.8 |
| Infranasal | 0.168 | <.001 | y = 0.18x + 5.03 | 0.186 | <.001 | y = 13.6ln(x) − 33.4 |
| Infratemporal | 0.286 | <.001 | y = 0.15x + 11.6 | 0.324 | <.001 | y = 13.6ln(x) − 44.6 |
Diagnostic Performance of Spectral-Domain OCT and GDx VCC
Table 4 shows the ability of Cirrus OCT and GDx parameters to discriminate glaucomatous or suspected glaucomatous eyes from normal eyes, with sensitivities at ≥70% specificity and ≥90% specificity.



