Purpose
To assess macular photoreceptor abnormalities in eyes with resolved branch retinal vein occlusion (BRVO) using adaptive optics scanning laser ophthalmoscopy (AO-SLO).
Design
Prospective observational cross-sectional case series.
Methods
After complete resolution of macular edema and retinal hemorrhage, 21 eyes (21 patients) with BRVO underwent full ophthalmologic examination and imaging with optical coherence tomography (OCT) and a prototype AO-SLO system. Cone density and spatial mosaic organization were assessed using AO-SLO images.
Results
Regular parafoveal cone mosaic patterns were clearly visualized with the prototype AO-SLO imaging system in the BRVO-unaffected side. However, in the side of the retina previously affected by the BRVO, cone mosaic patterns were disorganized and dark regions missing wave-guiding cones were apparent. Additionally, retinal capillaries were dilated, no longer had a uniform caliber, and had less direct paths through the retina. In the affected side, parafoveal cone density was significantly decreased, compared with the corresponding retinal area on the unaffected side ( P < .001). Furthermore, the hexagonal Voronoi domain ratio and the nearest-neighbor distances were significantly lower than in the unaffected side ( P < .05). These parameters were also correlated with photoreceptor layer integrity in the parafovea.
Conclusions
After BRVO-associated retinal hemorrhage and macular edema resolved, affected parafoveal cone density decreases and the cone mosaic spatial arrangement is disrupted, becoming more irregular. These cone microstructural abnormalities may extend to parafovea in the BRVO-unaffected side.
Branch retinal vein occlusion (BRVO) is the second most common retinal vascular disorder after diabetic retinopathy. In the eyes with BRVO, macular edema is a major complication and is often accompanied by a decrease in visual acuity (VA). Recent extensive investigations with optical coherence tomography (OCT) have demonstrated diverse macular pathomorphologies associated with BRVO. Murakami and associates examined the photoreceptor inner and outer segment junction (IS/OS) line after resolution of macular edema associated with BRVO and used it as a hallmark of foveal photoreceptor layer integrity. Subsequent reports combining microperimetry results and IS/OS line status on OCT images showed that retinal sensitivity within the macula is preserved in eyes with an intact IS/OS line following BRVO resolution. These OCT investigations revealed the importance of an intact photoreceptor layer in visual function. Unfortunately, some patients with an intact macular photoreceptor layer, as confirmed on OCT, still have residual subjective visual disturbances after VA recovery has been achieved.
One hypothesis for this subjective visual disturbance is that it is caused by pathologic changes in cones that cannot be detected with common OCT imaging. The primary reason these changes cannot be visualized is because of optical aberrations of the eye. An adaptive optics (AO) system consists of a wavefront sensor that measures ocular aberrations and a deformable mirror or spatial light modulator that compensates for these aberrations in real time. By incorporating AO technology into OCT or scanning laser ophthalmoscopy (SLO), these aberrations can be compensated for, allowing for clear imaging of individual cone photoreceptors in living eyes. To date, little information is available on cone patterns in eyes with BRVO, as examined using AO-imaging devices. In the current study, we used a prototype AO-SLO system to assess macular photoreceptor abnormalities in eyes with resolved BRVO and to study correlations with OCT findings.
Methods
The Ethics Committee at Kyoto University Graduate School of Medicine approved this prospective study, which was conducted in accordance with the tenets of the Declaration of Helsinki. Written informed consent was obtained from each subject before any study procedures or examinations were performed.
Participants
This prospective study included 21 eyes with resolved BRVO from 21 consecutive patients who were examined at the Department of Ophthalmology of Kyoto University Hospital between November 2012 and July 2013. The BRVO diagnosis was based on fundus examination and fluorescein angiographic findings, as determined by 2 retina specialists (A.T., T.M.). Cone mosaic images were taken from both previously affected and unaffected sides of the retina using an original prototype AO-SLO system. Inclusion criteria included: (1) history of symptomatic BRVO, in which retinal hemorrhage and retinal edema extended to the fovea; (2) complete resolution of retinal hemorrhage and macular edema; and (3) best-corrected VA better than 20/30 following BRVO resolution. Eyes with central retinal vein occlusion or hemicentral retinal vein occlusion were excluded from the current study. Eyes with coexisting ocular disease (eg, epiretinal membrane, glaucoma, diabetic retinopathy) were also excluded.
Ophthalmic Examinations
All subjects underwent comprehensive ophthalmologic examination, including measurement of Landolt best-corrected VA, intraocular pressure, and axial length (IOL Master; Carl Zeiss Meditec, Dublin, California, USA). Indirect ophthalmoscopy, slit-lamp biomicroscopy with a contact lens, color fundus photography, OCT imaging (Spectralis HRA+OCT; Heidelberg Engineering, Heidelberg, Germany), and AO-SLO imaging were also performed. All examinations were performed on the same day for each subject.
Adaptive Optics Scanning Laser Ophthalmoscope System
We developed an original prototype AO-SLO system in collaboration with Canon, Inc. The system is composed of 4 primary optical subsystems, which include the AO subsystem (includes the wavefront sensor and the spatial light modulator), the high-resolution confocal SLO imaging subsystem, the wide-field imaging subsystem, and the pupil observation subsystem. The observation system facilitates initial pupillary alignment with respect to the AO-SLO optical axis by adjusting the chin rest position. The wavefront sensor measures aberrations in the whole eye, and the spatial light modulator compensates for these aberrations. The AO-SLO system is confocal, enabling creation of “en face” images in any plane. These high-resolution images show individual cone photoreceptor cells.
Adaptive Optics Scanning Laser Ophthalmoscope Images
A series of AO-SLO images were taken in each eye at several locations within the macular area. The imaging series at each location was acquired by moving the focus from the retinal nerve fiber layer to the retinal pigment epithelium (RPE), with particular attention paid to the cone mosaic. To obtain cone mosaic images from both the previously affected and unaffected sides of the retina, we used 3 different imaging fields (large field of view [L]: 1700 × 1700 μm; middle field of view [M]: 820 × 820 μm; small field of view [S]: 340 × 340 μm). First, the AO-SLO was centered on the foveal center and the 3 field-of-view images (L, M, and S) were obtained. Next, middle field-of-view images (M) were collected that were centered 0.5 and 1.0 mm away from the foveal center in each direction (superior, nasal, inferior, temporal). Small field-of-view images were then collected with the AO-SLO images centered 0.5 and 1.0 mm superior and inferior to the foveal center. Finally, images centered 0.7 and 1.4 mm superotemporal, superonasal, inferotemporal, and inferonasal to the foveal center were obtained ( Figure 1 ). Images collected from the side previously affected with BRVO are shown in figure parts labeled B, while images from the unaffected side are shown in figure parts labeled A.
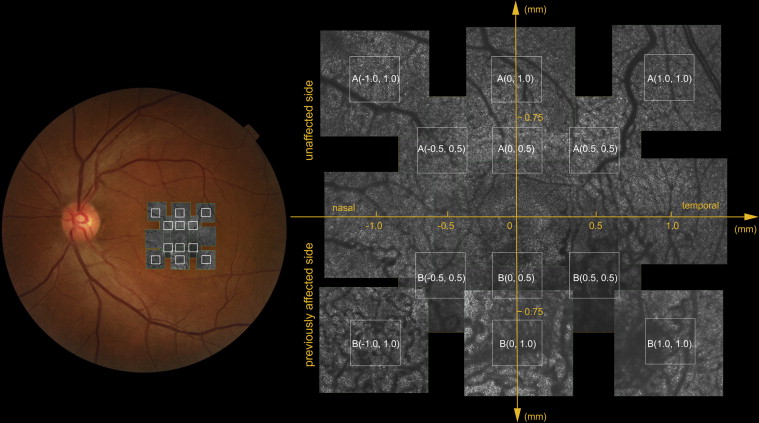
Final AO-SLO images were obtained by averaging 22 individual images acquired at each location. Scans were averaged in this way to reduce signal noise. Image location was verified by examining the correspondence between each montage and the area of interest. This was done by comparing high-magnification AO-SLO images with wide-field AO-SLO images. The AO-SLO image montage was also used to verify corresponding locations of other imaging modalities by matching retinal vessel patterns.
Analysis of the Cone Mosaic
Cone integrity was evaluated by applying an automated cone labeling process, previously established by Li and Roorda. When the algorithm failed to properly identify cones, cone labeling results were manually corrected. An experienced observer, who was masked to clinical findings, examined each image after automated cone labeling was completed. The observer then manually labeled areas where cones were visible but had not been identified by the software.
Cone density was estimated in selected areas where vascular component shadows were not present. The computer software was then instructed to estimate cone density by dividing the number of cones in each imaging area by the size of the imaging area. The area for quantification (80 × 80 μm) was identified manually by selecting the area without vascular shadows and by placing the computer cursor on the area to be quantified. This method has been used in previous studies, and interobserver reproducibility was high in normal eyes, as well as in eyes with retinitis pigmentosa. The location of the foveal center, defined here as the center of the foveal avascular zone, was determined using AO-SLO image montages. Cone density was measured in each location and overall cone density was taken as the mean of all measurements in all sections. Accurate scan lengths were determined in each eye using the adjusted axial length method to correct for the eye’s inherent magnification effect.
Spatial organization of the cone mosaic was determined by nearest-neighbor distances (NNDs) and Voronoi domains. Voronoi domains were constructed for each cell by defining points in the region that were closer to that cell than to any other cell in the mosaic. The NNDs were determined by calculating the minimum distance from the cell center to every other cell center in the mosaic. The expected NND was calculated as that of a perfect hexagonally packed mosaic, with a density equal to that in each location. The ratio of the observed average NND and the expected NND was calculated in each field of view.
Parafoveal Outer Photoreceptor Layer Integrity and Retinal Thickness
Vertical B-scan images (30 degrees) through the foveal center were obtained in each eye with the Spectralis HRA+OCT. To reduce speckle noise, 100 OCT images at the same location were acquired and averaged. Total retinal thickness and outer retinal thickness were measured 0.5 and 1.0 mm away from the foveal center ( Figure 2 ). Total retinal thickness was defined as the distance between the vitreoretinal interface and the inner border of the RPE. Outer retinal thickness was defined as the distance between the outer border of the inner nuclear layer and the inner border of the RPE. Thickness was measured with digital calipers built into the OCT software. Inner retinal thickness was calculated by subtracting outer retinal thickness from total retinal thickness.
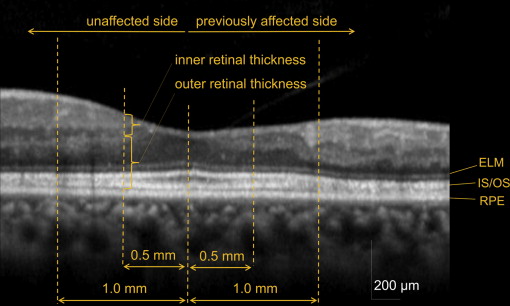
In averaged OCT cross-sections through the fovea, the integrity of the external limiting membrane (ELM) and the IS/OS line was determined 0.5 and 1.0 mm away from the foveal center by masked evaluators. Eyes in which the ELM and IS/OS lines were deemed intact were classified as ELM(+) and IS/OS(+), respectively. Eyes in which the ELM and IS/OS lines were deemed discontinuous were classified as ELM(±) and IS/OS(±), respectively. When the ELM and IS/OS lines could not be detected, eyes were classified as ELM(–) and IS/OS(–), respectively.
Statistical Analyses
Best-corrected VA was measured using the Landolt chart and converted to the logarithm of the minimal angle of resolution (logMAR) for analyses. We compared means of cone density, NNDs, and Voronoi parameters using the Mann-Whitney U test. Bivariate relationships were analyzed using the Pearson correlation coefficient. Differences between 2 retinal regions were tested for statistical significance using the paired t test. Statistical analyses were performed using SPSS software (version 19.0; SPSS, Inc, Chicago, Illinois, USA). Statistical significance was defined as P < .05.
Results
Twenty-one eyes from 21 patients (11 women, 10 men) with resolved BRVO were included in the study. Subjects ranged in age from 49-70 years and had an average age of 61.3 ± 6.0 years. Table 1 summarizes the characteristics of all included patients, both at the initial BRVO presentation and at the time of AO-SLO examination. At the initial visit, all patients had BRVO-associated fresh retinal hemorrhage and retinal edema involving the fovea. Foveal thickness measured greater than 250 μm in all 19 eyes examined with OCT at the initial visit. Initial mean logMAR VA was 0.26 ± 0.41 (Snellen equivalent = 20/36). At the time of AO-SLO examination, all eyes had a VA better than 20/30 and average logMAR VA was −0.06 ± 0.12 (20/17). Six eyes in this study had undergone treatment for their macular edema. Two eyes had undergone pars plana vitrectomy, 2 eyes received posterior sub-Tenon injections of triamcinolone acetonide, 1 eye was treated with intravitreal injections of bevacizumab, and 1 eye was treated with intravitreal injections of triamcinolone acetonide. Scatter laser photocoagulation to the nonperfused retinal area had been performed in 6 eyes. The mean duration between BRVO onset and the initial treatment was 13.1 ± 22.2 months. The mean interval between macular edema resolution and AO-SLO imaging was 22.1 ± 19.7 months.
| Number of eyes | 21 |
| Sex (female/male) | 11/10 |
| Conditions at initial BRVO presentation (obtained from records) | |
| Age (y) | 57.0 ± 5.6 |
| Visual acuity (logMAR) | 0.26 ± 0.41 |
| Foveal thickness (μm) | 435.9 ±163.6 |
| Cystoid macular edema (presence/absence) | 13/8 |
| Serous retinal detachment at the fovea (presence/absence) | 13/8 |
| Subretinal hemorrhage at the fovea (presence/absence) | 11/10 |
| IS/OS line at the foveal center (detectable/undetectable) | 10/11 |
| Perfusion status (ischemic/nonischemic) | 6/13 |
| Conditions at resolution (time of AO-SLO examination) | |
| Age (y) | 61.3 ± 6.0 |
| Visual acuity (logMAR) | −0.06 ± 0.12 |
| Foveal thickness (μm) | 258.4 ± 31.5 |
| Time from initial BRVO presentation (mo) | 51.1 ± 34.6 |
| ELM line at the foveal center (detectable/undetectable) | 19/2 |
| IS/OS line at the foveal center (detectable/undetectable) | 19/2 |
Examination with OCT confirmed complete resolution of retinal edema and retinal hemorrhage. Additionally, both the ELM and IS/OS lines were visible and intact at the foveal center in all 19 eyes imaged. However, the side of the retina previously affected by BRVO often had slightly distorted layer structures, as well as retinal thinning ( Supplementary Table , available at AJO.com ). Although inner retinal thickness was not significantly reduced, outer retinal thickness was significantly smaller than in the unaffected side.
Retinal Vascular Abnormalities Visible With Adaptive Optics Scanning Laser Ophthalmoscopy
Retinal capillaries were seen as black shadows on the cone mosaic on AO-SLO images. In the BRVO-unaffected side, retinal capillaries appeared identical to those seen in a normal retina. However, in the BRVO-affected side, most capillaries were dilated, varied in caliber, and seemed to have a longer, more tortuous path than capillaries in the normal retina ( Figure 3 ).
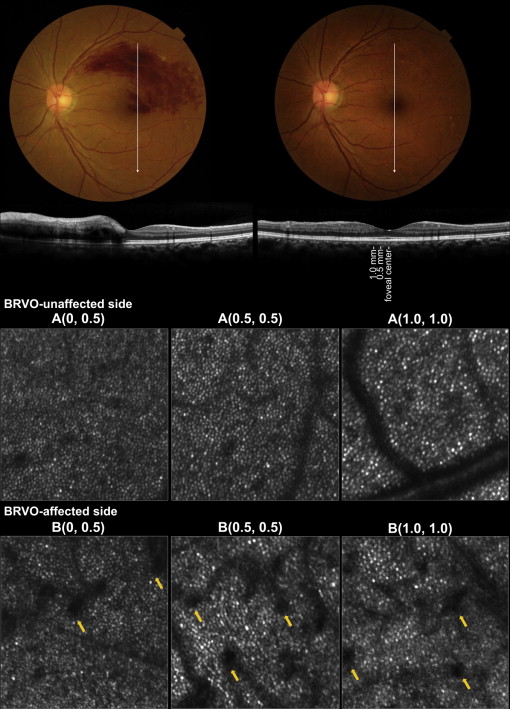
The Cone Mosaic on Adaptive Optics Scanning Laser Ophthalmoscope Images
Figure 4 shows AO-SLO images from an eye with a resolved BRVO. In the BRVO-unaffected side, cone mosaic patterns were regular, with only a minimal number of dark regions. However, in the affected side, cone mosaic patterns were disorganized and had numerous dark areas that were missing wave-guiding cones. Table 2 shows the cone density of each measured area. In both unaffected and affected sides, cone density decreased as the distance from the foveal center increased. At each eccentricity examined, cone density was significantly lower in the affected side than in the corresponding unaffected side ( P < .001). Additionally, hexagonal Voronoi domain ratio and average NND/expected NND ratio were significantly lower on the affected side than on the unaffected side (both P < .05).
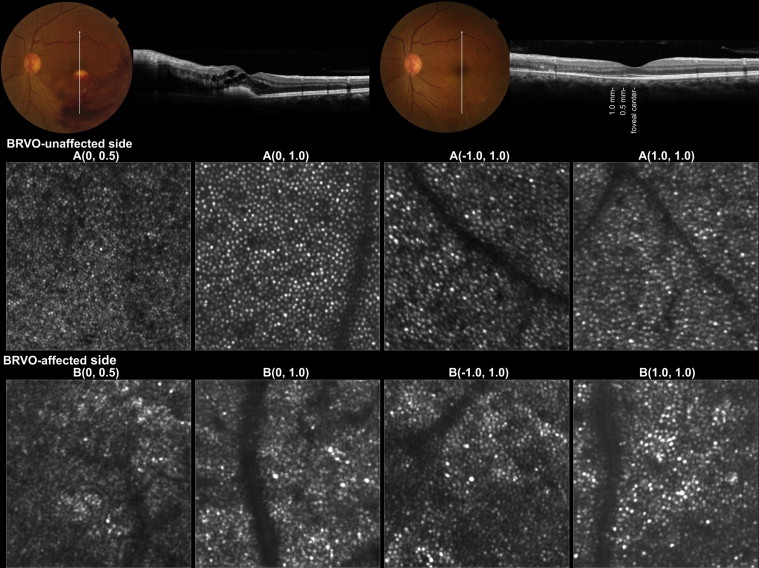
| Cone Density (Cones/mm 2 ) | Hexagonal Voronoi Domain Ratio (%) | Average NND/Expected NND | |
|---|---|---|---|
| A(0, 0.5) | 20 515 ± 3523 | 31.9 ± 7.1 | 0.50 ± 0.07 |
| B(0, 0.5) | 9210 ± 5188 | 20.4 ± 9.7 | 0.33 ± 0.15 |
| P value | <.001 | .003 | .042 |
| A(0, 1.0) | 15 192 ± 3890 | 29.2 ± 6.7 | 0.47 ± 0.08 |
| B(0, 1.0) | 9412 ± 4800 | 20.5 ± 8.1 | 0.30 ± 0.12 |
| P value | <.001 | <.001 | .002 |
| A(−0.5, 0.5) | 17 450 ± 4149 | 27.7 ± 5.2 | 0.49 ± 0.07 |
| B(−0.5, 0.5) | 8143 ± 4944 | 15.8 ±1 0.4 | 0.28 ± 0.15 |
| P value | <.001 | .005 | <.001 |
| A(−1.0, 1.0) | 15 289 ± 2802 | 25.5 ± 4.4 | 0.42 ± 0.07 |
| B(−1.0, 1.0) | 9789 ± 3887 | 18.5 ± 7.6 | 0.34 ± 0.12 |
| P value | <.001 | <.001 | .041 |
| A(0.5, 0.5) | 18 520 ± 2569 | 32.3 ± 5.6 | 0.52 ± 0.07 |
| B(0.5, 0.5) | 11 144 ± 5565 | 22.4 ± 10.6 | 0.36 ± 0.15 |
| P value | <.001 | .001 | <.001 |
| A(1.0, 1.0) | 15 267 ± 2982 | 27.4 ± 4.5 | 0.48 ± 0.06 |
| B(1.0, 1.0) | 8415 ± 4306 | 19.1 ± 9.4 | 0.33 ± 0.13 |
| P value | <.001 | <.001 | <.001 |
Relationship Between Outer Photoreceptor Layer Integrity and Cone Density
In vertical OCT cross-sections through the fovea, the integrity of the ELM and the IS/OS lines was examined 0.5 mm away from the fovea. The IS/OS line integrity 0.5 and 1.0 mm away from the foveal center was significantly correlated with regional cone density ( Table 3 , Supplementary Figure ). In addition, both the hexagonal Voronoi domain ratio and the average NND/expected NND ratio were positively correlated with IS/OS line integrity 0.5 and 1.0 mm away from the foveal center. The ELM line integrity at the same locations also showed similar correlations with regional cone density, hexagonal Voronoi domain ratio, and average NND/expected NND ratio.
| Cone Density (Cones/mm 2 ) | Hexagonal Voronoi Domain (%) | Average NND/Expected NND | ||||
|---|---|---|---|---|---|---|
| r | P Value | r | P Value | r | P Value | |
| 0.5 mm away from the foveal center | ||||||
| Integrity of ELM line | 0.39 | .011 | 0.21 | .071 | 0.36 | .018 |
| Integrity of IS/OS line | 0.52 | <.001 | 0.45 | .002 | 0.54 | <.001 |
| 1.0 mm away from the foveal center | ||||||
| Integrity of ELM line | 0.41 | .007 | 0.50 | <.001 | 0.46 | .002 |
| Integrity of IS/OS line | 0.46 | .002 | 0.63 | <.001 | 0.53 | <.001 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


