Purpose
To investigate the relationship between the clinical characteristics of polypoidal choroidal vasculopathy (PCV) and choroidal vascular hyperpermeability seen on indocyanine green angiography.
Design
Retrospective, consecutive, interventional case series.
Methods
We reviewed the medical records and the angiograms of 89 patients with PCV. The relationship between choroidal vascular hyperpermeability and background factors, associated clinical manifestations, and treatment responses to intravitreal injections of ranibizumab were evaluated.
Results
Of the 89 patients with PCV, 31 patients (34.8%) demonstrated choroidal vascular hyperpermeability. The patients with choroidal vascular hyperpermeability more frequently showed bilateral neovascular membrane than those without choroidal vascular hyperpermeability ( P = .009) and had a significant relationship with a history of central serous chorioretinopathy (CSC) ( P = .01). Of the 98 eyes with treatment-naïve PCV, 34 eyes with choroidal vascular hyperpermeability demonstrated significantly greater subfoveal thickness than the 64 eyes without choroidal vascular hyperpermeability ( P < .001). However, no significant relationship was found between choroidal vascular hyperpermeability and the other biomicroscopic and angiographic phenotypes of PCV. Three monthly intravitreal injections of ranibizumab were performed on 57 patients with treatment-naïve PCV, and the presence of choroidal vascular hyperpermeability was significantly related to the persistent retinal fluid 1 month after the third ranibizumab injection ( P = .01).
Conclusions
The patients with PCV associated with choroidal vascular hyperpermeability more frequently demonstrated bilateral neovascular membrane, a history of CSC, a thickened choroid, and poor responses to intravitreal injections of ranibizumab than those without choroidal vascular hyperpermeability.
Polypoidal choroidal vasculopathy (PCV) is a clinical entity characterized by multiple recurrent serosanguineous detachments of the retinal pigment epithelium and neurosensory retina secondary to leakage and bleeding from choroidal vascular lesions. Although PCV is generally considered to be one of the subtypes of neovascular age-related macular degeneration (AMD), the precise pathogenesis and the origins of the neovascular components remain controversial.
In addition to the peculiar neovascular components, PCV is reportedly known to demonstrate a finding on indocyanine green angiography (ICGA); namely, choroidal vascular hyperpermeability. Choroidal vascular hyperpermeability, which is visualized as multifocal hyperfluorescence in the middle and late phases of an ICGA, was originally described as a characteristic finding in central serous chorioretinopathy (CSC). Sasahara and associates reported that in their study, 9.8% of the eyes with PCV demonstrated choroidal vascular hyperpermeability with significantly higher frequency than did 1.9% of the eyes with typical neovascular AMD. Thus, the authors concluded that choroidal vascular hyperpermeability might be one of the risk factors for the development of PCV.
Recent advancements in optical coherence tomography (OCT) have enabled the visualization of the cross-sectional image of the choroid. When examined by enhanced depth imaging OCT (EDI-OCT), eyes with PCV appear to demonstrate a significantly thicker choroid than do those with typical neovascular AMD. That difference was attributed to the higher prevalence of choroidal vascular hyperpermeability in PCV compared with typical neovascular AMD. In fact, Maruko and associates reported that the PCV eyes in their study that were associated with choroidal vascular hyperpermeability demonstrated greater subfoveal choroidal thickness than did those without choroidal vascular hyperpermeability. Thus, it can be assumed that choroidal vascular hyperpermeability may be one of the characteristic findings in PCV and might be related to the pathogenesis to a certain degree. The purpose of the present study was to investigate the clinical characteristics of PCV that are possibly related to the presence of choroidal vascular hyperpermeability.
Methods
This study involved the retrospective review of the medical records of 89 consecutive patients (23 female and 66 male) with treatment-naïve and symptomatic PCV in at least 1 eye who were treated at the Macula Service of Kyoto Prefectural University of Medicine. The mean age of the patients was 74.1 years (SD: 8.1 years; range: 52-89 years). At baseline, all 89 patients underwent a comprehensive ophthalmic examination including best-corrected visual acuity (BCVA) testing with Landolt C charts, color fundus photography, fluorescein angiography (FA) and ICGA using a confocal scanning laser ophthalmoscope (HRA-2; Heidelberg Engineering Inc, Dossenheim, Germany), and spectral-domain OCT (3D-OCT 2000; TOPCON Corp, Tokyo, Japan). ICGA was performed using an intravenous dose of 25 mg of the dye in 1 mL of aqueous solvent. The angiograms were obtained in the macula and periphery by moving the ocular fixation object inside the ophthalmoscope.
The diagnosis of a definite PCV lesion was based on the characteristic polypoidal structures at the border of the branching choroidal vascular networks visualized with ICGA. Sometimes, the polypoidal structures could be detected funduscopically by the presence of an orange-red retinal protrusion over the polypoidal lesions. To investigate the bilaterality of the neovascular membrane, the angiograms of the asymptomatic contralateral eyes were also carefully reviewed. When the contralateral eyes were shown to have branching choroidal vascular networks on ICGA or a fibrous scar formation, those cases were regarded as having bilateral involvement. In fact, the Japanese Study Group defined the presence of the branching choroidal vascular networks alone on ICGA as a “probable” case of PCV. Choroidal vascular hyperpermeability was defined as multifocal hyperfluorescence seen in the middle and late phases of the ICGA. The diagnosis of PCV and the presence or absence of choroidal vascular hyperpermeability were evaluated by 2 independent investigators (T.Y. and T.Y.). When the evaluation was inconsistent, the senior investigator (H.K.) made the final decision.
At the initial presentation, each patient was extensively interviewed using a structured questionnaire to obtain their background data in relation to sex, age, presence of diabetes, presence of hypertension, presence of hyperlipidemia, history of smoking (number of packs per day × number of years smoking), history of CSC, and history of steroid use. Hypertension, diabetes, and hyperlipidemia were considered to be present if the patient had a past medical history of the same or if the patient took hypertension medication, insulin or hypoglycemics, and lipid-lowering medications, respectively. The previous history of CSC was defined as the patient having had CSC in either side of his or her eyes, since CSC often reveals a bilateral pathologic feature of the choroid, even with a unilaterally evident manifestation. Steroid use is one of the prominent risk factors for CSC and choroidal vascular hyperpermeability is a common finding in CSC, so we included not only systemic steroid use but also chronic application of local steroids.
To investigate the relationship between choroidal vascular hyperpermeability and the other clinical manifestations of PCV, the following 10 characteristics were selected: (1) BCVA; (2) duration of visual symptoms; (3) presence of a subretinal hemorrhage larger than 1 disc area; (4) presence of serous or hemorrhagic pigment epithelial detachment larger than 1 disc area; (5) presence of concurrent type-2 choroidal neovascularization; (6) number of polyps; (7) size of the largest polyp; (8) greatest linear dimension (GLD) based on FA; (9) GLD based on ICGA; and (10) subfoveal choroidal thickness. GLD on FA was defined as the lesion dimension covering the area of possible neovascular membrane, including the areas of dye leakage, pigment epithelial detachment, and subretinal hemorrhage. GLD on ICGA was defined as the dimension of the definite PCV lesion consisting of the polypoidal lesions and the branching choroidal vascular networks. To measure the subfoveal choroidal thickness, the EDI-OCT method was applied by using the choroidal mode within the OCT. In all examined eyes, 6-mm horizontal and vertical scans, each consisting of a maximum of 50 averaged scans, were obtained through the center of the fovea. Next, the 1 scan of 2 scans in which the inner surface of the sclera was more clearly visualized was selected. Subfoveal choroidal thickness was defined as the distance between the hyperreflective line corresponding to the Bruch membrane beneath the retinal pigment epithelium and the inner surface of the sclera, using the caliper function of the OCT. The reliability of the choroidal thickness measurement method used in this study was validated by the findings in our previous report. All of the measurements were obtained by investigators (T.Y. and T.Y.) who were masked to the patients’ information. In addition, some of the 89 consecutive eyes included in the present study received 3 monthly intravitreal injections of 0.5 mg ranibizumab (Lucentis; Genentech, South San Francisco, California, USA). In those eyes, the frequency of complete resolution of retinal fluid (termed “dry macula”) detected by OCT was evaluated 1 month after the third ranibizumab injection, and whether or not choroidal vascular hyperpermeability and the dry macula had a significant relationship was investigated.
The obtained data were statistically analyzed in relation to frequency and description. BCVA was converted to logarithm of the minimal angle of resolution (logMAR) units before the analyses, and the χ 2 test was used for categorical analysis. The Fisher exact test was used if the expected cell count was fewer than 5, and means were compared by use of the Mann-Whitney U test. To investigate the background factors directly related to choroidal vascular hyperpermeability, and to evaluate whether the presence of choroidal vascular hyperpermeability had an association with the dry macula after 3 monthly intravitreal injections of ranibizumab, multivariate logistic regression analysis was performed. Forward and backward stepwise regression was performed using the likelihood-ratio method, in which change in the likelihood-ratio statistic, based on the maximum partial likelihood estimates for the covariate, was used for variable selection. A P value of <.05 was considered to be statistically significant, and all tests were 2-sided. All of the statistical analyses were performed with SPSS software version 18.0 (SPSS Inc, Chicago, Illinois, USA).
Results
Of the 89 patients, 11 patients (12.3%) had a definite PCV lesion in both eyes. Of the 11 patients with bilateral definite PCV lesions, 2 patients had a treatment history of PCV in 1 of the 2 eyes. With regard to the remaining 78 patients, 1 eye had a definite PCV lesion, 64 eyes showed no abnormal choroidal vascular membrane in the contralateral eye, 11 eyes exhibited suspicious branching choroidal vascular networks, and 3 eyes showed a fibrous scar formation. Therefore, 64 patients (71.9%) were regarded as having unilateral involvement and 25 patients (28.1%) were regarded as having bilateral involvement.
Of the 89 patients with a definite PCV lesion at least in 1 eye, 31 patients (34.8%) demonstrated choroidal vascular hyperpermeability. Of the 31 patients with choroidal vascular hyperpermeability, 25 patients (80.6%) showed choroidal vascular hyperpermeability bilaterally and the remaining 6 patients (19.4%) showed choroidal vascular hyperpermeability unilaterally. Fourteen of the 31 patients (45.2%) with choroidal vascular hyperpermeability showed bilateral involvement of neovascular lesion compared with 11 of the 58 patients (19.0%) without choroidal vascular hyperpermeability ( P = .009).
Table 1 demonstrates the comparisons of background factors between the 31 patients with choroidal vascular hyperpermeability and the 58 patients without choroidal vascular hyperpermeability. Of those factors, the history of CSC alone was significantly correlated with choroidal vascular hyperpermeability by means of univariate analysis ( P = .01). Forward and backward stepwise regression found that the history of CSC was the sole significant factor related to the presence of choroidal vascular hyperpermeability ( P = .01). None of the cases included in this study had undergone photodynamic therapy (PDT) with verteporfin for the treatment of CSC.
| Background Factor | Choroidal Vascular Hyperpermeability (+) (n = 31) | Choroidal Vascular Hyperpermeability (-) (n = 58) | P Value (Univariate) | P Value (Multivariate) |
|---|---|---|---|---|
| Male sex, n (%) | 23 (74.2%) | 43 (74.1%) | >.99 | – |
| Age (y) ± SD | 72.7 ± 9.4 | 74.8 ± 7.3 | .32 | – |
| Diabetes, n (%) | 6 (19.3%) | 10 (17.2%) | .80 | – |
| Hypertension, n (%) | 12 (38.7%) | 33 (56.9%) | .10 | – |
| Hyperlipidemia, n (%) | 2 (6.5%) | 13 (22.4%) | .08 | – |
| Smoking (packs per day × y) ± SD | 24.3 ± 32.0 | 20.8 ± 30.3 | .41 | – |
| History of CSC, n (%) | 8 (25.8%) | 3 (5.2%) | .01 a | 0.01 a |
| History of steroid use, n (%) | 2 (6.4%) | 3 (5.2%) | >.99 | – |
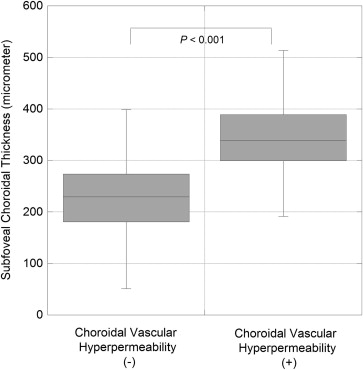
To investigate the relationship between choroidal vascular hyperpermeability and the other clinical manifestations of PCV, we evaluated 98 treatment-naïve eyes with a definite PCV lesion. Of those 98 eyes, the PCV lesion was located at the subfovea in 70 eyes, at the juxtafovea in 11 eyes, at the extrafovea in 10 eyes, and in the peripapillary area in 7 eyes. Choroidal vascular hyperpermeability was seen in 34 of the 98 eyes (34.7%) and was found to be significantly related to the greater subfoveal choroidal thickness ( P < .001) ( Figure 1 ). However, there was no evident relationship between the presence of choroidal vascular hyperpermeability and the biomicroscopic and angiographic phenotypes of PCV, except for the borderline significance of the GLD on ICGA ( P = .05) ( Table 2 ).
| Clinical Manifestation | Choroidal Vascular Hyperpermeability (+) (n = 34) | Choroidal Vascular Hyperpermeability (-) (n = 64) | P Value |
|---|---|---|---|
| BCVA (logMAR) ± SD | 0.64 ± 0.47 | 0.50 ± 0.41 | .16 |
| Duration of visual symptoms (mo) | 14.4 ± 31.0 | 11.9 ± 25.9 | .53 |
| Subretinal fluid, n (%) | 29 (85.3%) | 54 (84.4%) | .90 |
| Subretinal hemorrhage, n (%) | 3 (8.8%) | 8 (12.5%) | .74 |
| Pigment epithelial detachment, n (%) | 5 (14.7%) | 11 (17.2%) | .75 |
| Concurrent type 2 CNV, n (%) | 3 (8.8%) | 3 (4.7%) | .41 |
| Number of the polyps ± SD | 2.3 ± 1.2 | 2.6 ± 2.1 | .83 |
| Size of the largest polyp ± SD (μm) | 619 ± 349 | 547 ± 286 | .51 |
| GLD on FA ± SD (μm) | 4711 ± 2111 | 4400 ± 2622 | .25 |
| GLD on ICGA ± SD (μm) | 3826 ± 1701 | 3173 ± 1871 | .05 |
Of the 98 eyes with a definite PCV lesion, 57 consecutive eyes received 3 monthly intravitreal injections of ranibizumab. Of these 57 eyes, 40 eyes (70.2%) exhibited the dry macula 1 month after the third ranibizumab injection. Table 3 demonstrates the relationship between the dry macula and the clinical manifestations at baseline. Consequently, the absence of choroidal vascular hyperpermeability and the smaller size of the largest polyp were significantly related to the dry macula ( P = .005 and P = .03, respectively). Forward and backward stepwise logistic regression analysis found that the presence of choroidal vascular hyperpermeability and the smaller size of the largest polyp were the independent factors related to the dry macula ( P = .01 and P = .03, respectively). The representative cases are shown in Figures 2 and 3 .
| Clinical Manifestation | Dry Macula (+) (n = 40) | Dry Macula (-) (n = 17) | P Value (Univariate) | P Value (Multivariate) |
|---|---|---|---|---|
| BCVA (logMAR) ± SD | 0.51 ± 0.42 | 0.62 ± 0.49 | .37 | – |
| Duration of visual symptoms (mo) | 21.1 ± 41.2 | 13.2 ± 19.4 | .79 | – |
| Subretinal fluid, n (%) | 32 (80.0%) | 16 (94.1%) | .25 | – |
| Subretinal hemorrhage, n (%) | 6 (15.0%) | 3 (17.6%) | >.99 | – |
| Pigment epithelial detachment, n (%) | 7 (17.5%) | 4 (23.5%) | .72 | – |
| Choroidal vascular hyperpermeability, n (%) | 10 (25.0%) | 11 (64.7%) | .005 b | .01 c |
| Concurrent type 2 CNV, n (%) | 3 (7.5%) | 2 (4.7%) | .63 | – |
| Number of polyps, n (%) | 2.3 ± 1.4 | 2.2 ± 1.3 | .80 | – |
| Size of the largest polyp ± SD (μm) | 509 ± 259 | 752 ± 407 | .03 c | .03 c |
| GLD on FA ± SD (μm) | 4541 ± 2111 | 4288 ± 1363 | .68 | – |
| GLD on ICGA ± SD (μm) | 3108 ± 1951 | 3564 ± 1297 | .18 | – |
| Subfoveal choroidal thickness ± SD (μm) | 247 ± 90 | 294 ± 85 | .08 | – |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


