Purpose
To characterize punctate hyperfluorescence spot as common choroidopathy in central serous chorioretinopathy (CSC) and polypoidal choroidal vasculopathy (PCV).
Design
Cross-sectional retrospective study.
Methods
A total of 150 patients with 50 each allocated to CSC, PCV, and typical neovascular age-related macular degeneration (AMD) groups were included. Punctate hyperfluorescence spot was determined using mid-to late-phase indocyanine green angiography and subfoveal choroidal thickness by enhanced depth imaging optical coherence tomography. Each group was subcategorized based on concurrent punctate hyperfluorescence spot.
Results
The punctate hyperfluorescence spot incidence was higher in CSC (80.0%) and PCV (86.0%) than in AMD (40.0%, P < .001), with similar contralateral findings (86.1%, 86.7%, and 60%, respectively, P = .014). Punctate hyperfluorescence spot lesions comprised clustered polyps connected to vascular networks mimicking PCV. Choroidal thickness was 370.7 ± 81.9 μm, 332.6 ± 101.6 μm, and 172.5 ± 80.1 μm in affected eyes ( P < .001) and 323.0 ± 70.5 μm, 306.4 ± 94.4 μm, and 180.2 ± 83.6 μm in contralateral eyes ( P < .001) in CSC, PCV, and AMD groups, respectively. In the AMD group, choroidal thickness was greater in eyes with punctate hyperfluorescence spot (204.8 ± 92.3 μm) than in those without punctate hyperfluorescence spot (150.2 ± 62.9 μm, P = .028) in affected eyes; however, the difference was not observed in contralateral eyes in the AMD group and in both eyes in the CSC and PCV groups.
Conclusions
Based on angiography and OCT, punctate hyperfluorescence spot may be a form of PCV, and CSC and PCV may share common choroidopathy distinct from typical neovascular AMD. However, infrequent PHS lesions along with thickened choroids in AMD eyes suggest that AMD may encompass a wide choroidal pathologic spectrum shared in part with PCV.
Indocyanine green angiography (ICGA) provides detailed images of choroidal vasculature and has generated a growing awareness of choroidal vascular diseases including central serous chorioretinopathy (CSC), polypoidal choroidal vasculopathy (PCV), and age-related macular degeneration (AMD). Typical CSC is easily diagnosed and does not require ICGA. However, atypical CSC or chronic CSC often mimics other conditions that threaten vision, such as PCV, and ICGA is enormously helpful in proper diagnosis by assessing choroidal vascular abnormalities, including filling delays in choroidal vasculature, abnormal choroidal venous dilation during the early phase, and choroidal vascular hyperpermeability. These ICGA findings suggest that CSC is a disease of choroidal circulation and vasculature.Interestingly, several studies reported that some CSC cases later developed into PCV, and a history of CSC is a risk factor for PCV development. PCV is also a choroidal vascular disorder and also has unique ICGA features, including a branching inner choroidal vascular network, hypofluorescent halo, and choroidal vascular hyperpermeability. The choroidal abnormalities observed in CSC and PCV, particularly choroidal vascular hyperpermeability, suggest a similar choroidopathy underlying both CSC and PCV. Studies have recently reported that the choroid is thicker in eyes with PCV or CSC than that observed not only in normal eyes but also in eyes with typical neovascular AMD, which further suggests a common pathogenesis between CSC and PCV.
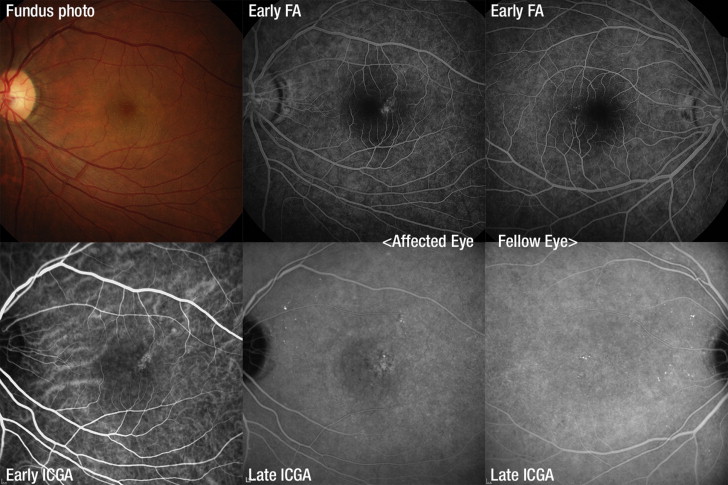
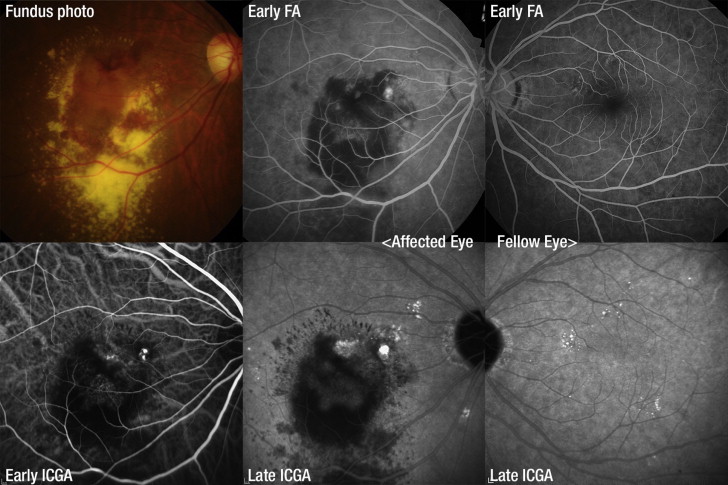
A punctate hyperfluorescence spot is a focal hyperfluorescent area observed during the mid to late phase of ICGA in both the CSC-affected eye and the unaffected contralateral eye. Tsujikawa and associates were the first to use the term “punctate hyperfluorescence spot,” although prior studies illustrated the punctate hyperfluorescence spot lesion in figures. They reported that punctate hyperfluorescence spot lesions frequently occurred within the macular area, outside the vascular arcade, or near the optic disc, and punctate hyperfluorescence spots seemed to arise in the inner choroid. Punctate hyperfluorescence spots also occur in eyes with PCV and the unaffected contralateral eye; several studies investigating ICGA findings in PCV unintentionally described punctate hyperfluorescence spots in eyes with PCV. However, there has been no study investigating punctate hyperfluorescence spot as a common choroidopathy underlying both CSC and PCV. These prior results underscore the need to investigate punctate hyperfluorescence spot as a common choroidal vasculopathy underlying CSC and PCV.
Methods
This retrospective observational study was conducted at a single center (Seoul National University Bundang Hospital) by the use of the database of the Seoul National University Bundang Hospital genetic study. Patients with newly diagnosed CSC, PCV, and typical neovascular AMD have been enrolled in the Seoul National University Bundang Hospital genetic study, after providing written informed consent, since July 2007. The present study was approved by the Seoul National University Bundang Hospital institutional review board and followed the tenets of the Declaration of Helsinki.
The database was used after September 2009, when spectral-domain (SD) optical coherence tomography (OCT) became available at the study site, and patients with CSC, PCV, and typical neovascular AMD were consecutively reviewed until December 2012, totaling 150 patients (50 patients each in the CSC, PCV, and typical neovascular AMD groups). Included patients underwent a thorough ophthalmic examination of at least the affected eye, including slit-lamp examination, fundus examination using a slit lamp with a 90 diopter lens and indirect ophthalmoscope, fundus fluorescein angiography (FA), SD OCT (Spectralis; Heidelberg Engineering, Heidelberg, Germany), and ICGA with a confocal laser-scanning system (HRA-2; Heidelberg Engineering).
Eyes were defined as CSC when the FA findings met 1 of the 2 following criteria: (1) a circumscribed subretinal fluid lesion with isolated leakage points; and (2) widespread leakage from retinal pigment epithelium (RPE) damaged regions. Eyes were defined as having PCV when the ICGA revealed subretinal nodular hyperfluorescence (polyps) at the border or within the branching vascular network (BVN). Eyes were defined as having typical neovascular AMD when choroidal neovascularization (CNV) was confirmed by FA and OCT. Eyes with PCV were not included in either CSC or typical neovascular AMD groups. In addition, eyes with true CNV accompanied by PCV were not included in any group. Patients meeting at least 1 of the following criteria were excluded: (1) significant cataract or media opacity; (2) high myopia (>−6 diopter); (3) macular hole; (4) thick subfoveal hemorrhage; (5) macular geographic atrophy; (6) previous photodynamic therapy; (7) previous surgical repair of retinal detachment, glaucoma, or ocular trauma; and (8) anti–vascular endothelial growth factor administration within the previous 6 months. Data from the contralateral eyes were also analyzed provided they showed no active leakage, exudative lesion, or polypoidal lesion on FA or ICGA.
We investigated punctate hyperfluorescence spots in all included eyes, both affected and contralateral eyes, using mid-to late-phase ICGA. The punctate hyperfluorescence spot location was categorized as follows: (1) between 1-mm-diameter and 6-mm-diameter ring surrounding the fovea (perifovea); (2) within 1.5 mm of the disc margin (peripapilla); and (3) the rest of the retina. The punctate hyperfluorescence spot distribution was categorized as follows: solitary pattern (1 punctate hyperfluorescence spot or several scattered punctate hyperfluorescence spot lesions) and clustered pattern (several punctate hyperfluorescence spot lesions clustered in 1 or several areas). In affected eyes, punctate hyperfluorescence spot located inside or within 500 μm of the causative lesions was not included in the analyses; the causative lesion was categorized as follows: (1) all active vascular leakage in all subjects, (2) BVN with polyps in PCV subjects, and (3) CNV membrane (CNVM) in AMD subjects.
Using enhanced depth imaging OCT, the choroidal thickness beneath the fovea was measured manually by 1 operator (S.J.P.) after thorough discussion and agreement on measuring method among authors; choroidal thickness was defined as the vertical distance between the Bruch membrane or the RPE–Bruch membrane complex outer border and the innermost chorioscleral interface. To evaluate the interobserver reproducibility of our measuring method, OCT images from 30 randomly selected participants (10 each allocated to CSC, PCV, and AMD groups) were independently evaluated by 2 authors (S.J.P. and B.H.K.) masked to clinical information. The intraclass correlation coefficient (ICC [2, 1]) was calculated for the choroidal thickness. Eyes with typical neovascular AMD were subcategorized using OCT as follows: type 2, which had a CNVM localized predominantly in the subretinal space; and type 1, which had the CNVM localized predominantly in the sub-RPE space.
The incidence of punctate hyperfluorescence spot in affected and unaffected eyes was compared between the patient groups using the χ 2 test. The t test or 1-way analysis of variance (ANOVA) was used to compare corresponding mean values. P values <.05 were considered statistically significant. All statistical analyses were conducted using SPSS version 16 (SPSS Inc, Chicago, Illinois, USA).
Results
In total, 150 affected eyes and 128 fellow eyes (43, 45, and 50 in the CSC, PCV, and typical neovascular AMD group, respectively) were reviewed. Within the 3 50-patient groups, 41 (82%), 32 (64%), and 22 men (44%) comprised the CSC, PCV, and typical neovascular AMD groups, respectively. Of 50 typical neovascular AMD patients, 33 patients had type 1 CNV and 17 patients had type 2 CNV. In affected eyes, the ICGA showed punctate hyperfluorescence spot in 40 CSC eyes (80.0%), 43 PCV eyes (86.0%), and 20 typical neovascular AMD eyes (40.0%) ( P < .001). In fellow eyes, the ICGA showed punctate hyperfluorescence spot in 37 CSC eyes (86.1%), 39 PCV eyes (86.7%), and 24 typical neovascular AMD eyes (60.0%) ( P = .014). The punctate hyperfluorescence spot locations in affected and unaffected eyes in the 3 groups are summarized in the Table . In typical neovascular AMD patients, the punctate hyperfluorescence spot was not associated with the CNV type ( P = .903). Representative punctate hyperfluorescence spot lesions in affected and fellow eyes of patients with CSC ( Figure 1 ), PCV ( Figure 2 ), and typical neovascular AMD ( Figure 3 ) are shown. Various punctate hyperfluorescence spot features and schematized depictions of punctate hyperfluorescence spots, polyps, and vascular networks are shown in Figure 4 . Punctate hyperfluorescence spot lesions comprised clustered polyps connected to vascular networks and mimicked small PCV.
| Affected Eyes | CSC | PCV | AMD | P Value |
|---|---|---|---|---|
| N | 50 | 50 | 50 | |
| Age (y) | 48.0 ± 7.4 | 66.5 ± 7.9 | 70.3 ± 6.4 | <.001 a |
| Men | 41 (82.0%) | 32 (64.0%) | 22 (44.0%) | |
| PHS | 40 (80.0%) | 43 (86.0%) | 20 (40.0%) | <.001 b |
| Location | ||||
| Perifovea | 13 (26.0%) | 10 (20.0%) | 5 (10.0%) | |
| Peripapilla | 7 (14.0%) | 22 (44.0%) | 9 (18.0%) | |
| Others | 33 (66.0%) | 36 (72.0%) | 17 (34.0%) | |
| Subfoveal CT (μm) | 370.7 ± 81.9 | 332.6 ± 101.6 | 172.5 ± 80.1 | <.001 a |
| With PHS | 373.9 ± 81.7 | 339.8 ± 99.4 | 204.8 ± 92.3 | |
| Without PHS | 357.7 ± 85.9 | 288.86 ± 11.9 | 150.2 ± 62.9 | |
| P Value c (with vs without PHS) | .688 | .222 | .028 |
| Fellow Eyes | CSC | PCV | AMD | P Value |
|---|---|---|---|---|
| N | 43 | 45 | 40 | |
| PHS in fellow eye | 37 (86.1%) | 39 (86.7%) | 24 (60.0%) | .014 b |
| Location | ||||
| Perifovea | 17 (39.5%) | 18 (40.0%) | 7 (17.5%) | |
| Peripapilla | 8 (18.6%) | 18 (40.0%) | 10 (25%) | |
| Others | 30 (49.8%) | 36 (80.0%) | 23 (57.5%) | |
| Subfoveal CT (μm) | 323.0 ± 70.5 | 306.4 ± 94.4 | 180.2 ± 83.6 | <.001 a |
| With PHS | 319.9 ± 64.9 | 310.7 ± 97.0 | 193.5 ± 93.2 | |
| Without PHS | 320.5 ± 95.1 | 250.6 ± 67.3 | 186.3 ± 74.4 | |
| P Value c (with vs without PHS) | .984 | .188 | .799 |
a P value was calculated by 1-way analysis of variance.
b P value was calculated by χ 2 test.

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


