Purpose
To analyze the functional outcome with regard to the development of visual acuity and radiation-induced optic neuropathy of patients with parapapillary choroidal melanoma treated with proton beam therapy.
Design
Clinical case series, retrospective study.
Methods
We evaluated 147 consecutive patients with parapapillary choroidal melanoma who received proton beam therapy as primary tumor treatment at the Helmholtz Center in Berlin from 1998 to 2005. A cumulative dose of 60 Cobalt Gray Equivalents (CGE) was delivered to the tumor and the optic disc received a minimum of 50 CGE. Kaplan-Meier analysis was used to assess ocular outcome and survival rates. For trend analysis of functional development, Wilcoxon-Mann-Whitney U test was used to compare the medians of 2 groups and Kruskal-Wallis test was used in the case of more than 2 groups.
Results
The mean follow-up time was 6.5 years (range 0.3-11.7 years). The most common side effects were radiation-induced optic neuropathy, retinopathy, and cataract. The median visual acuity before and within the first year after therapy was 0.4 logMAR (20/50), lapsing to 1.3 logMAR (20/400) after 3 years and 1.4 logMAR (20/500) after 5 years. During follow-up, no light perception developed in 17 cases (11.6%), mostly attributed to radiation-induced retinopathy, optic neuropathy, and secondary glaucoma. Enucleation was carried out in 14 patients (9.5%) because of local recurrence or severe side effects.
Conclusion
Radiation-induced optic neuropathy is an expected issue after proton beam therapy of parapapillary choroidal melanoma, and visual impairment is common during long-term follow-up, but some useful vision can be preserved in a considerable number of patients.
The Collaborative Ocular Melanoma Study (COMS) indicated that iodine plaque irradiation of medium-sized choroidal melanoma did not influence the overall survival rate as compared to primary enucleation. Today, irradiation has become widely accepted as a globe-preserving treatment modality in the majority of choroidal melanomas. Irradiation techniques comprise brachytherapy with, for example, iodine-125 or ruthenium-106 plaques or teletherapy; foremost proton beam therapy; and stereotactic radiosurgery (gamma-knife). In tumors with para- or peripapillary locations, the involvement of the optic disc is unavoidable, since it has to be included in the treatment target irrespective of the chosen treatment technique. Irradiation induces microvasculopathy owing to endothelial cell damage of the small vessels, leading to vascular occlusion, edema, and atrophy of the optic disc in the late stage. Radiation-induced optic neuropathy is anticipated in these cases. Parapapillary location is a relative constraint for brachytherapy, as a sufficient irradiation warrants plaque placement beneath the tumor, which might be hindered by the optic nerve. Therefore, these tumors have been excluded from the COMS study.
Though notched plaques allow the treatment of juxtapapillary melanomas, the correct placement is complex and prone to malpositioning or tilting. Especially in irregularly shaped tumors or those with a large base, local tumor control can only be ensured at the expense of damaging the surrounding normal tissue, since it has to be included in the irradiation field. Moreover, brachytherapy affects dose distributions that typically account for very high radiation loads delivered to the macula and optic disc when the plaque has to be sutured close to these critical structures. Therefore, eccentric plaque placement has been practiced in some centers, taking into consideration the possibility of a higher recurrence rate. Whereas brachytherapy is a rather common method, there are few centers worldwide that offer proton beam therapy. The advantage of proton beam therapy is the individually matched beam, offering the possibility of sparing critical structures of the eye while applying a homogeneous dose to the tumor. This is because of the physical dose distribution of the proton beam in tissue, resulting in a high dose in the target and an abrupt dose drop behind, the so-called Bragg peak. Although proton beam treatment planning is not impeded by a parapapillary location, owing to a safety margin of typically 1.5-2.5 mm, a significant dose will be applied to the optic disc and anterior optic nerve.
In this series, we examined the radiation-related side effects and the functional outcome of 147 patients with primary parapapillary choroidal melanoma treated with proton beam therapy, to investigate the patient benefits and risk factors for visual impairment during long-term follow-up.
Methods
In this retrospective study we included all patients treated with proton beam therapy at the cyclotron facility in the Helmholtz Center in Berlin who received a minimum irradiation dose of 50 Cobalt Gray Equivalents (CGE) to the optic disc, owing to the parapapillary location of the tumor, between December 1998 and December 2005. The study was approved by the Institutional Review Board of the Charité – Universitätsmedizin Berlin, Germany, and was in accordance with the Declaration of Helsinki specifications.
Treatment was performed with a total dose of 60 CGE, usually delivered in daily fractions of 15 CGE on 4 consecutive days. Treatment technique and dosimetric details have been published earlier. All tumors were homogeneously covered by the full therapeutic dose, adding a minimum safety margin of 1 mm (90% isodose) around the clinical target volume. Providing for customary beam conditioning, a steep dose fall-off to the 10% isodose level within 0.95 mm in depth and within 1.2-1.5 mm to the 50% isodose level in the transverse direction could be achieved in all patients. A tumor was considered parapapillary if the tumor was abutting the optic disc or if its posterior margin was <0.5 mm distant from the optic disc. Patients were excluded owing to planned and performed adjuvant tumor endoresection, which was performed after May 2000 in a prospective protocol in patients with a tumor thickness of ≥7 mm. We further excluded those patients who received proton beam therapy as secondary salvage therapy owing to local recurrences after a primary transpupillary thermotherapy (9 patients) or ruthenium plaque brachytherapy (3 patients). In total, 147 patients were included in this retrospective study. The irradiation took place at the cyclotron facility of the Helmholtz-Center Berlin (formerly Hahn-Meitner-Institut) using a 68 megaelectron volts (MeV) fixed proton beam as described previously. Prior to irradiation, all patients received a surgical tantalum clip placement. Treatment planning was performed with EYEPLAN software. The irradiation dose was applied in 4 sessions, each with 15 CGE, in 143 patients (97.3%) and with 8× 7.5 CGE in 3 patients (2.0%). In a single case (0.7%) a dose of 50 CGE was applied with 8× 6.25 CGE. Fractionation was performed with 8 applications, attempting to minimize radiation-induced side effects owing to the shape of the specific tumors. All patients were treated with a complete lid-sparing technique. The patients were examined at regular time intervals for up to 10 years after proton beam therapy. The missing follow-up data were completed by correspondence with the referring local ophthalmologists and the general practitioners responsible for the patients.
Circumpapillary extension was defined as contiguous tumor extension of ≥200 degrees around the optic disc. Subfoveal location was defined as a tumor extent within 0.75 mm of the foveal center. Visual acuity (VA) was quoted as logMAR and as Snellen based on 20 ft, if applicable. According to the MARAN protocol, counting fingers (CF, 1.9 logMAR), hand movements (HM, 2.0 logMAR), and light perception (LP, 2.1 logMAR) were successively added to the logMAR scale. Tumors were categorized according to the actual TNM classification scheme for choroidal melanoma. Local recurrence was defined as an increase in tumor basal diameter in comparison to the initial and following photographs or consistent increase of tumor thickness in the ultrasound. Radiation retinopathy was defined as the appearance of neovascularization, retinal hemorrhages, hard exudates, or cotton-wool spots. Rubeosis iridis and vitreous hemorrhage were evaluated independently. We did not differentiate between proliferative and nonproliferative retinopathy and maculopathy. Optic neuropathy was defined as edema, pallor, or hemorrhage of the optic disc. Secondary glaucoma was defined as persisting intraocular pressure of more than 20 mm Hg in patients without known history of glaucoma of another origin. The Kaplan-Meier analysis was used to assess ocular outcome and overall survival rates. The Wilcoxon-Mann-Whitney U test was used to compare the median tumor thickness as well as the median visual acuity of 2 groups, while the Kruskal-Wallis test was used in cases of 3 or more groups. Cox regression was performed for multivariate analysis. P < .05 was considered statistically significant. Statistical analyses were carried out with SPSS version 20.0 (SPSS Inc, Chicago, Illinois, USA).
Results
In total, we identified 147 patients with parapapillary choroidal melanoma with a mean age of 57 years (median 59 years, range 20-85 years). The mean follow-up time was 78 months (median 77 months, range 4-140 months). A total of 144 patients completed at least 1 year of follow-up; 125 patients were observed within a minimum of 5 years and 28 patients more than 10 years after proton beam therapy.
Tumor Characteristics, Local Recurrences, and Metastases
The mean values of the tumor thickness and the largest basal diameter were 3.7 mm (range 1.4-7.5 mm) and 11.0 mm (range 3.3-19.1 mm), respectively. According to the AJCC/UICC TNM protocol, 35.4% (52/147) of the tumors were categorized as T1, 49.0% (72/147) as T2, and 15.6% (23/147) as T3/T4 owing to the large basal diameter. In 98.0% of the patients (144/147) the tumor was abutting the optic disc; in particular, 51.0% of tumors (75/147) were situated circumpapillary. A subfoveal extension was present in 37.4% (55/147). In 19.0% (28/147) the tumor was located both circumpapillary and under the fovea. Forty-four of the 147 tumors (29.9%) were centered nasal-superior, 32 (21.8%) nasal-inferior, 35 (23.8%) temporal-inferior, and 36 (24.5%) temporal-superior. Extraocular growth was present in 3 of 147 patients (2.0%). Local recurrence was observed in 9 patients (6.1%) after a mean of 23 months (range 3-81 months). Metastasis developed in 23 patients (15.6%) after a mean of 45 months (range 10-130 months). In 1 additional case, metastasis was already present before proton beam therapy was initiated. This patient died 5 months after the initial diagnosis. During the follow-up a total of 28 of the patients (19.0%) died, of whom 17 had a proven melanoma-associated cause of death.
Side Effects and Secondary Treatment
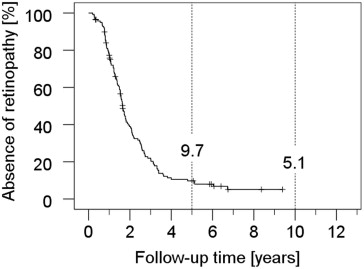
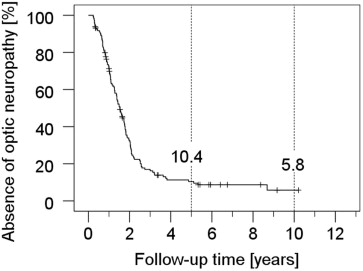
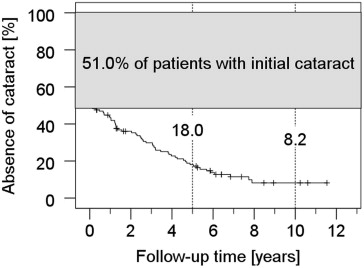
The eye preservation rate was 90.5% and 89.3% at 5 and 10 years after proton beam therapy, respectively. Altogether, enucleation was performed in 14 patients (9.5%) during follow-up. In 6 patients, enucleation was necessary owing to a local tumor recurrence. Another 8 eyes were removed at a mean time of 32 months (range 10-52 months) after proton beam therapy, mostly because of intractable pain and/or secondary neovascular glaucoma. Five-year rates for the retinopathy and optic neuropathy were 90.3% and 89.6%, respectively. The onset of both complications was early (mean ± SD 19 ± 14 months [range 3-104 months] for optic neuropathy and mean ± SD 22 ± 14 months [range 2-81 months] for retinopathy). Rubeosis (iris neovascularization) (mean ± SD 30 ± 22 months [range 6-98 months]), secondary glaucoma (mean ± SD 31 ± 30 months [range 3-131 months]), and vitreous hemorrhage (mean ± SD 31 ± 24 months [range 3-122 months]) were comparatively late complications. Secondary cataract occurred after a mean of 36 months (SD ± 24 months, range 2-95 months), considering only patients without cataract history at presentation (n = 72). The Kaplan-Meier estimators are shown in Figures 1-3 . According to Cox regression analyses, the occurrence of rubeosis ( P < .001), secondary glaucoma ( P < .001), vitreous hemorrhage ( P = .001), and retinopathy ( P = .01) was significantly dependent on the TNM size of the tumor, while it was not in the case of optic neuropathy ( P = .05) and other radiation-related side-effects, such as cataract and exudative retinal detachment ( P > .1 each). Laser treatment of the retina was the most common additional therapy (24.5%), followed by vitrectomy (18.4%) and cataract surgery (17.7%), predominantly taking place between the first and fourth year (mean 26 months, range 3-123 months). Vitrectomy was carried out at a mean of 28 months (range 4-123 months), mostly because of vitreous hemorrhage. In 2 cases, vitrectomy was combined with secondary endoresection of the tumor as a consequence of a presumed “toxic tumor syndrome” (no recurrence) and doubtful tumor control (recurrence suspected) 11 and 3 months after irradiation, respectively. Cyclodestructive treatment to control secondary glaucoma was performed in 6.1% of the patients during follow-up after a mean of 30 months. The median irradiated volume of the ciliary body, which received ≥50% of the dose, was 16 vol% or 2 clock hours (range 0-46 vol% or 0-6 clock hours). Patients who developed rubeosis, glaucoma, or opticoneuropathy had a significantly higher ciliary body involvement ( P < .001, P < .05, or P < .05, respectively, 2-sided t test for independent probes), while there was no difference regarding cataract.
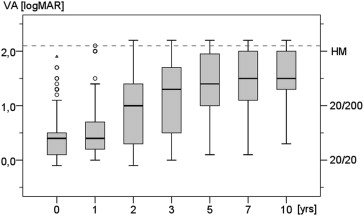
Functional Outcome
The median VA was 0.4 logMAR (20/50) before treatment in all patients, ranging from −0.1 logMAR (20/16) to CF. It remained unchanged during the first year after proton beam therapy. Visual deterioration took place between the second and the fifth year after proton beam therapy, the median acuity level dropping from 1.0 logMAR (20/200) to 1.4 logMAR (20/500) ( Figure 4 ). Patients whose tumors were situated outside the fovea (n = 75) presented with a significantly better VA during the follow-up period when compared to patients with foveal tumor extension ( P ranging from <.001 to .01 in yearly intervals). The initial median VA and the VA 1 year after proton beam therapy in the fovea-spearing group was 0.2 logMAR (20/32). From the second to the fifth year after proton beam therapy, VA slowly decreased to 1.3 logMAR (20/400). From the fifth year on, median VA was stable. In case of a subfoveal location of the melanomas (n = 72) the median initial VA was 3 lines worse (0.5 logMAR or 20/63 Snellen). VA remained stable for the first year, but rapidly dropped to 1.5 logMAR (20/630) within the next 4 years. From the fifth year on, median VA was stable at this level, ranging from 1.0 logMAR (20/200) to amaurosis. In contrast, there were 11 patients in the fovea-spearing group who maintained a VA of 0.3 logMAR (20/40) or better 5 years after proton beam therapy. Another 9 patients could be identified with a VA better than 1.0 logMAR until the end of follow-up. After setting the fovea dose as an independent variable, we compared the functional outcome of patients whose fovea could be completely spared from the irradiation field (n = 24) to those with the fovea partially or completely included in the field, reaching either the 1, 50, or 90 percent isodose level (ie, 1-29 CGE: n = 21, 30-53 CGE: n = 20, or 54-60 CGE: n = 82, respectively). In cases where the dose to the fovea reached the therapeutic range (90%-100% isodoses or 54-60 CGE), early deterioration of the VA was the most frequent consequence (in 58 out of 82 cases). The median vision for the whole high-dose group decreased within 3 years after proton beam therapy, at about 10 lines with respect to the vision prior to tumor therapy (from 0.5 to 1.5 logMAR or 20/63 to 20/630 Snellen). Instead, the majority of patients who got less than 54 CGE to the fovea could keep a VA of 1.0 logMAR (20/200) or better until the end of follow-up (in 42 out of 65 cases). This finding even proved to be unchanged whether the cumulated low-dose group had further been segregated according to the different isodose levels (null, 1-29, and 30-53 CGE). In the long run, visual deterioration predominated. In the end, 17 of 147 patients (11.6%) had no light perception during the follow-up. This was attributable to secondary glaucoma and/or radiation-induced retinopathy or optic neuropathy in these patients, of whom 7 had to be enucleated subsequently. According to the Kaplan-Meier analysis, the chance of avoiding no light perception was 89.9% at 5 years and 83.3% at 10 years ( Figure 5 ). VA was better by up to 4 lines in patients with small-sized tumors (T1), in comparison to medium or large ones (T2-4, P < .01), during the first 2 years. There was no significant difference in VA after the second year. Even in patients with challenging tumor and irradiation conditions, a stabilization of vision or even a gain of 2 lines and more could be found during the first 3 years. A comparison of 2 patients with circumpapillary melanoma is shown in Figure 6 . Past history of diabetes mellitus did not influence the visual outcome.