Purpose
To evaluate long-term outcomes of proton beam radiotherapy in the treatment of choroidal melanoma of the intermediate zone of the fundus.
Design
Retrospective interventional single-center study.
Methods
The study was a retrospective analysis with long-term follow-up of 62 patients with a minimum tumor-to-disc and tumor-to-fovea distance of 2 mm of choroidal melanoma in the intermediate zone of the fundus. Mean values of tumor prominence, largest basal diameter, and tumor distances to the optic disc and fovea were 7.6, 12.8, 5.2, and 4.6 mm, respectively. All patients were irradiated with a total proton dose of 60 cobalt gray equivalents.
Results
After proton beam radiotherapy, 71.0% of the patients received subsequent endoresection of the tumor. Only 18 patients (29.0%) did not require additional tumor resection and were analyzed as a separate group. For the total of patients, the median follow-up time was 70.3 months. The 5-year Kaplan-Meier rates of local tumor relapse, enucleation, and distant metastasis were 3.9%, 3.7%, and 13.4%, respectively. Cataract surgery was the most frequent secondary treatment in our cohort.
Conclusions
In this study we demonstrate the effectiveness of proton beam irradiation in tumor control and preservation of the globe in the analyzed patients. The rate of metastasis was not higher than previously described. Nevertheless, consecutive tumor surgery is frequently required to maintain the eye in patients with large-sized choroidal melanomas. In conclusion, proton beam radiotherapy allows preservation of the eye in mid-zone choroidal melanomas.
Enucleation for choroidal melanoma has been replaced by radiotherapy as an accepted treatment standard. The most popular techniques used for radiotherapy of choroidal melanoma are brachytherapy and proton beam therapy. There are currently more than 30 years of experience with proton beam irradiation. The advantage of using protons for irradiation is the fact that the proton energy can be modulated to cover target tissue of any shape and at any depth with a nearly uniform dose owing to its physical characteristics, including minimal scatter and delivery of maximum dose at the end of the beam path, known as Bragg peak. Several investigators have demonstrated a noninferiority of proton beam irradiation with respect to metastasis and tumor-specific survival. While proton beam therapy has been advocated for central choroidal melanoma with close proximity to the disc and fovea, studies demonstrated that tumors exceeding 6 mm in height can be successfully treated with proton beam therapy if the inflammatory reaction after irradiation is met with surgical excision of the tumor tissue via a transscleral approach or endoresection.
The present retrospective analysis presents the long-term outcomes in patients with proton beam irradiation for choroidal melanoma in the intermediate zone of the fundus at the Center for Ocular Oncology in Berlin.
Methods and Materials
Patients and Data Collection
This is a retrospective single-center study of patients with choroidal malignant melanoma treated with proton beam radiotherapy in Berlin. The study was approved by the institutional review board of the Charité–Universitätsmedizin Berlin, Germany, and was in accordance with the Declaration of Helsinki specifications. This study focuses on tumors in the intermediate zone of the fundus not attaching the ciliary body. The minimal distance of the unifocal lesion to both the optic disc and fovea was 2 mm. Consequently, exclusion criteria for this study were tumor invasion of the anterior eye segment, parapapillary location or location close to the fovea, and pretreatment of the tumor.
From 1998 until 2005, a total number of 62 patients met the inclusion criteria and received proton therapy. Fourty-four out of 62 tumors (71.0%) were regarded as large-sized, with a prominence of at least 6 mm, and were treated with a combination of primary irradiation and consecutive tumor endoresection performed during the interval between postirradiation day 10 and postirradiation day 30. No additional surgery was performed in 18 patients (29.0%) presenting only small and medium-size tumors and who were pooled in a separate group. The regular treatment for small to medium-size tumors without close proximity to the central ocular structures is ruthenium brachytherapy. Thus, the proton beam–treated group of small and medium-size tumors was rather small. There were no patients with extraocular growth or metastatic disease at the time of diagnosis.
The following clinical data were extracted from the patients’ medical records: treatment type, age, sex, affected eye, tumor location, tumor height, largest basal diameter, fluorescein angiography, and presence of subretinal fluid. Furthermore, side effects of radiation, such as cataract formation or secondary glaucoma, were assessed. Cataract was defined as the first occurrence of lens opacity before or after irradiation. Glaucoma was defined as persistent intraocular pressure of more than 20 mm Hg. Patients with optic nerve damage or with a local therapy at the present time owing to a known history of glaucoma were regarded as glaucoma patients, as well. Visual field was not done to detect abnormalities for diagnosis. Similarly, preexisting sight-threatening ocular diseases and previous surgical interventions were evaluated, as well as visual acuity prior to radiotherapy and during the follow-up. Follow-up visits were usually planned 6, 12, 18, and 24 months after radiotherapy and then annually. Follow-up data that were acquired by the end of December 2010 were considered for this study.
Treatment Protocol
A 68-megaelectron-volt proton beam at the Helmholtz-Zentrum Berlin was used to deliver the irradiation dose. Eye position and tumor margins were demarcated by tantalum marker clips prior to radiotherapy. During treatment the patient’s head was immobilized using an individually manufactured mask, and positioned by means of 3D digital radiography with a precision of about 0.3 mm. Irradiation was applied in 4 fractions of 15 cobalt gray equivalents on 4 consecutive days. A total dose of 60 cobalt gray equivalents was homogeneously distributed over the planned target volume, comprising the clinical target volume plus 1 mm of safety margin (ie, the 90% isodose level) in the surrounding area.
Endoresection of the Tumors
In cases with large tumors, endoresection was performed as previously described. In short, after posterior vitreous detachment, decalin was used to attach the retina. Subsequently, the tumor tissue was endoresected with a cutter-probe with the intraocular pressure elevated to prevent excessive hemorrhage from the tumor ground. After endolaser of the margins of the coloboma left behind after complete resection of all tumor tissue, the intraocular pressure had slowly been reduced to normal. An exchange of the decalin with silicone oil was performed at the end of the procedure.
Outcome Measures
Case-dependent prescription of the therapeutic regime called for a division of patients into 2 groups, 1 of which presented with smaller tumors that did not require any additional surgical therapy after proton beam irradiation (n = 18). In contrast, patients with more prominent tumors who received local endoresection after proton beam irradiation were evaluated within the other group (n = 44).
Local tumor control was defined as no evidence of tumor recurrence within the follow-up. Tumor recurrence was assumed if an increase in tumor volume of more than 25% was observed over 2 examination intervals at least 6 months after radiotherapy. Either a tumor growth at the margins of the lesions that was observed from fundus photography or an increase of the tumor prominence of about 1 mm or more upon ultrasound examination was considered recurrent or tumor progression. A pseudo-increase in size that was caused by tumor exudation or hemorrhages on the tumor surface was not considered as treatment failure but as a sign of radiation retinopathy.
Endpoints for survival analysis were enucleation, tumor recurrence, occurrence of metastatic disease, and tumor-related death. Secondary enucleation was performed only as a last resort in blind painful eyes.
Statistical Analysis
For time-to-event analysis the Kaplan-Meier method was applied. A P value below .05 was considered statistically significant. Computations were performed with IBM SPSS Statistics release 20.0 (SPSS Inc, Chicago, Illinois, USA).
Results
Sixty-two patients met the inclusion criteria of our retrospective study. The mean age was 57.7 years (standard deviation [SD] 13.6 years). Twenty-nine patients were female, 33 were male. The overall range for tumor prominence was 1.7–12.6 mm (mean 7.6 mm, SD 2.9 mm), and it was 6.6–21.5 mm (mean 12.8 mm, SD 3.4 mm) for the largest tumor diameter. The respective fovea and disc distances ranged from 2.0 both to 10.1 mm and 11.3 mm with a mean distance of 4.6 mm to the fovea (SD 2.2 mm) and 5.2 mm to the optic disc (SD 2.1 mm). For the proton beam radiotherapy alone group (n = 18) the mean values of tumor height and largest tumor diameter were 3.9 mm (SD 2.0 mm) and 10.6 mm (SD 3.0 mm), respectively. The mean tumor-to-fovea distance was 3.7 mm, and the tumor-to-disc distance was centered at 3.9 mm (both SD 1.9 mm). In the endoresection group (n = 44) we found a mean tumor height of 9.1 mm (SD 1.5 mm), a mean largest tumor diameter of 13.7 mm (SD 3.2 mm), and mean distances to the fovea and the disc of 5.1 and 5.3 mm (SD 2.2 and 2.3 mm), respectively.
None of the patients demonstrated extraocular growth. Diabetes mellitus was present in 2 patients who were part of the endoresection group (3.2% of the cohort). One patient in each treatment group demonstrated glaucoma at pretreatment diagnosis. The median follow-up time was 77.2 months in the proton beam radiotherapy alone group and 64.4 months in the endoresection group. Nine eyes in the first group (50.0%) and 39 eyes in the latter group (88.6%) showed an exudative retinal detachment at presentation.
Tumor Regression
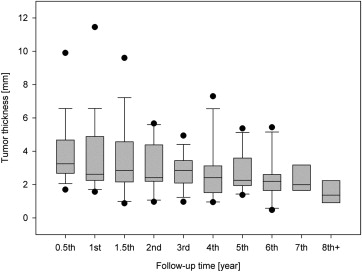
In the proton beam radiotherapy alone group, a slight tendency toward an initial swelling of the tumor could be observed in 3 patients during the first year after proton beam radiotherapy ( Figure 1 ), followed by a continuous decrease in median tumor prominence up to the end of follow-up, which was valid for all of the patients in this group. Five years after proton beam radiotherapy, the tumor volume, as determined by the largest tumor diameter and prominence, was reduced to about 65% of the volume at the initial presentation. In the surgical group there were no alterations of the tumor height owing to the almost complete surgical excision of the tumor tissue.
Survival and Local Tumor Control
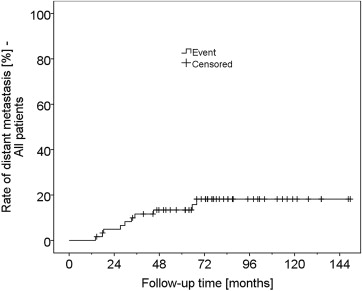
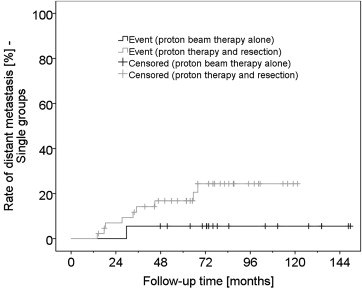
For the total of 62 patients, metastasis-free survival was estimated at a rate of 86.6% and 81.8% 5 and 10 years after proton beam radiotherapy, respectively ( Figure 2 ). Of 10 patients who developed metastasis, 8 died before completion of the 5-year follow-up (1 in the proton beam radiotherapy alone group, 7 in the endoresection group, Figure 3 ). Another 2 patients died of other causes, and 12 patients terminated the follow-up without known reason. Metastasis-related mortality was thus estimated at 10.6% and 16.9% within 5 and 10 years, respectively. Patients with smaller tumors who were treated by proton beam radiotherapy alone had a trend to a lower rate of metastasis-induced death compared to patients with large tumors and concomitant surgical therapy ( Figures 4 ).
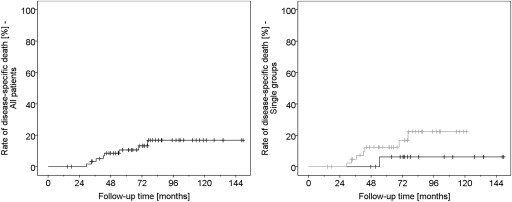
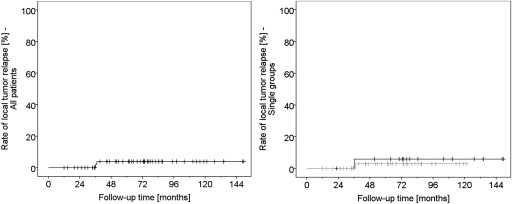
Local tumor control was estimated at 96.1% both after 5 and after 10 years. In total, 2 local recurrences developed during follow-up, 1 in each treatment group ( Figures 5 ).
Functional Outcome
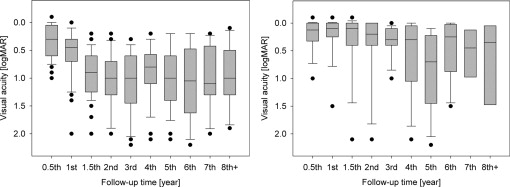
Both initial visual acuity and final visual outcome ( Figures 6 ) were better in the proton beam radiotherapy alone group, where the visual acuity on presentation was high (20/16–20/50 Snellen or −0.1–0.4 logMAR) in 15 patients (83.3%), medium (20/63–20/160 Snellen or 0.5–0.9 logMAR) in 2 patients (11.1%), and low (20/200 Snellen or 1.0 logMAR and worse) in 1 patient (5.6%). The final visual acuity in this group was high in 9 (50.0%), medium in 3 (16.7%), and low in 6 patients (33.3%).