Purpose
To evaluate the effects of a novel ab interno trabeculectomy device on human trabecular meshwork (TM).
Design
Laboratory evaluation.
Methods
The TM from human cadaveric corneal rim tissue was incised using 3 instruments: (1) novel dual-blade device; (2) microvitreoretinal (MVR) blade; and (3) Trabectome. Tissue samples underwent histologic processing and comparative analyses. Subsequently, human eye perfusion studies were performed to evaluate intraocular pressure (IOP)-lowering effects of each device. Main outcome measures were degree of TM removal by histology and IOP in a perfusion model.
Results
The MVR blade exhibited minimal removal of TM and obvious injury to the adjacent sclera. The Trabectome removed a large portion of the central TM, but leaflets of residual tissue remained and thermal injury was noted in all samples. The dual-blade device achieved a more complete removal of TM without injury to surrounding tissues. All devices resulted in statistically significant lowering of IOP during perfusion model studies. MVR blade treatment across 170.0 ± 14.1 degrees of TM resulted in a decrease of IOP from 18.5 ± 1.9 mm Hg to 12.8 ± 2.2 mm Hg ( P < .01). Trabectome treatment across 117.5 ± 12.6 degrees resulted in a decrease of IOP from 18.8 ± 1.7 mm Hg to 11.3 ± 1.0 mm Hg ( P < .01). Dual-blade device treatment across 157.5 ± 26.3 degrees resulted in a decrease of IOP from 18.3 ± 3.0 mm Hg to 11.0 ± 2.2 mm Hg ( P < .01).
Conclusions
The novel dual-blade device demonstrated a more complete removal of TM without residual TM leaflets or damage to surrounding tissues and significantly reduced IOP in a human eye perfusion model.
Glaucoma is a leading cause of blindness worldwide. The only known modifiable disease risk factor is intraocular pressure (IOP). Treatment centers on lowering IOP pharmaceutically with hypotensive medications or surgically through the use of lasers or incisional procedures. The main area of obstruction to aqueous outflow, with subsequent dysregulation of IOP, is thought to be located at the juxtacanalicular trabecular meshwork (TM) and distal outflow structures. Performing a goniotomy or trabeculotomy in adults with glaucoma has not been associated with great success in lowering IOP. In contrast, these procedures are more successful in congenital glaucoma, where a membrane covering the TM is thought to be a major factor in impedance of aqueous outflow. More recently, there have been attempts to use novel ab interno trabeculectomy procedures to remove TM in adult patients and results have been mixed.
One reason for poor long-term outcomes with this approach in adults might be related to incomplete removal of TM and membrane formation across the remaining TM leaflets with subsequent elevation in IOP. It is unclear how a more complete removal of TM tissue might compare to procedures that simply incise TM, such as goniotomy, or procedures that cauterize TM with tissue removal, such as Trabectome (Neomedix, Tustin, California, USA). This study investigates the preclinical use of a novel dual-blade device to more completely remove TM tissue. The dual-blade device is specifically designed to conform to the drainage angle anatomy of the human eye. The device is meant to perform an ab interno trabeculectomy by engaging TM and cutting the target tissue while minimizing leaflets left in place and damage to adjacent tissues. The device was designed and manufactured at the University of Colorado Eye Center (patent filing # 61637611). Tissue effects from the novel device are compared to those from a goniotomy using a microvitreoretinal (MVR) blade (BD, Franklin Lakes, New Jersey, USA) and cautery of TM with the Trabectome device. Human eye perfusion studies were also completed to assess the IOP-lowering efficacy of each approach.
Methods
Approval for this preclinical study was obtained from the Colorado Multiple Institutional Review Board for the use of human material prior to initiation of this study and the tenets of the Declaration of Helsinki were followed. Informed consent was obtained from donors or relatives for use in research by the eye bank from which human globes were obtained.
For histologic analyses, 6 corneal rim specimens were obtained from the Rocky Mountain Lions Eye Bank (Aurora, Colorado, USA) and the San Diego Eye Bank (San Diego, California, USA). Tissue samples were removed from the storage medium and mounted on a platform with the TM side facing up and secured in place using tissue pins. A total of 2 samples were used for each of the 3 treatment methods studied. An MVR blade was used to incise the central TM under microscopic visualization along the length of 2 corneal rims. For the Trabectome device, the foot plate of the device tip was inserted into the Schlemm’s canal under microscopic visualization. Once in place, the foot pedal was used to apply continuous ablation while advancing the tip slowly across the extent of the TM sample. A standard power setting of 0.8 W was used during treatment. The dual-blade device was used to incise the TM of 2 samples. The blade tip was used to incise TM in a manner similar to that used for goniotomy and the blade was then advanced in a clockwise fashion along the extent of the TM. At the distal end, the blade tip was tilted upwards to incise a complete ribbon of TM and the process was repeated in a counterclockwise fashion to incise the remaining TM tissue.
All tissue samples were then immediately preserved in 4% paraformaldehyde/phosphate-buffered saline overnight at 4 C and then radially cut into quadrants. Rim sections were processed for histology and embedded into paraffin so that the cut edge of the tissue was facing the front of the block. Tissue sections (6 μm thick) were cut and stained with Mayer’s hematoxylin-eosin Y (Richard-Allan Scientific, Kalamazoo, Michigan, USA). Bright-field imaging was performed using a Nikon Eclipse 80i microscope (Nikon, Melville, New York, USA) equipped with a Nikon D5-Fi1 color camera and a Nikon CFI 10×/Plan Fluor objective lens.
Human Eye Perfusion Studies
A total of 12 human globes from pseudophakic donors with no history of glaucoma were obtained from various eye banks around the country for perfusion studies on each device. The perfusion system used a standard programmable syringe pump (Pump 11 Plus; Harvard Apparatus, Holliston, Massachusetts, USA). Pressure was monitored via an in-line real-time pressure transducer (Research Grade Pressure Transducer; Harvard Apparatus) connected to a single-channel chart recorder (Pharmacia REC-481; Pharmacia/Pfizer New York, New York, USA). Polyethylene tubing with a 1.14 mm inner diameter (PE-160; Warner Instruments, Hamden, Connecticut, USA) was used for all connections.
In each case, the human globe was first prepared by injecting Dulbecco’s modified Eagle medium (DMEM; Invitrogen/Life Technologies, Carlsbad, California, USA) through the optic nerve with a 26-gauge needle until the globe had returned to a spherical shape. The perfusion line (terminating in another 26-gauge needle) was inserted diagonally through the anterior chamber of the eye, passing through the cornea and pupil and ending with the tip beneath the iris. The globe was surrounded by damp gauze and the perfusion pump (filled with DMEM) was set to an initial inflow rate of 7 μL/min. IOP was allowed to increase until it reached 30 mm Hg. The infusion rate was then reduced to 2-5 μL/min to maintain a steady-state IOP for at least 60 minutes prior to TM incision. A preoperative IOP was measured immediately prior to incision in each case. A 1.7 mm stainless steel keratome blade (BD) was used create a tri-beveled clear corneal incision near the limbus, and the anterior chamber was filled with enough viscoelastic (HealonGV; Abbott Medical Optics, Abbott Park, Illinois, USA) to maintain the anterior chamber and provide adequate visualization during the procedure in each case. Each technique was performed under gonioscopic view using a standard direct gonioscope with microscope assistance. The surgical procedure used for each device is described above. In each case, approximately 100-180 degrees of TM was treated. For each device, treatment was started 180 degrees away from the corneal wound and extended along the angle in a clockwise direction. The device was then extended in a counterclockwise direction from the same starting point. Every effort was made to treat the maximum amount of degrees possible with each device. In the case of the dual-blade device and Trabectome, the instrument was rotated 180 degrees after the initial pass to direct the device tip in the direction of treatment. IOP was allowed to reach a steady state before measuring the postprocedure IOP. Each of the 3 studied surgical techniques was performed on a total of 4 eyes.
Data Analysis
The mean and standard deviation of preprocedure and postprocedure IOP was calculated for each device as well as percent change in IOP. Student paired t tests were used to compare preprocedure and postprocedure IOP for each device. A calculated P value < .05 was considered to be statistically significant.
Results
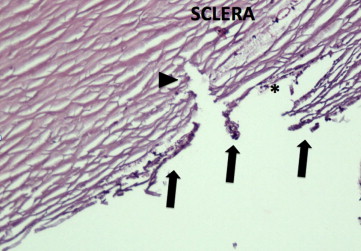
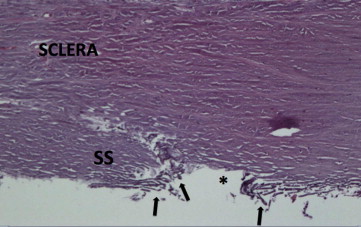
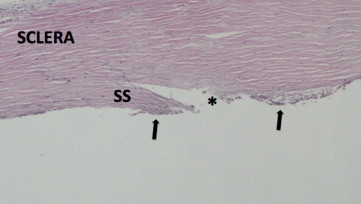
Two corneal rim sections were analyzed for each device. Six-micron-thick histologic sections were taken from various clock hours treated with each device and stained with Mayer’s hematoxylin-eosin Y (Richard-Allan Scientific). Findings were consistent across all sections from each device tested. Cuts with the MVR blade exhibited complete incision through the entire thickness of TM tissue. However, there was minimal removal of TM with large leaflets of tissue remaining over the Schlemm’s canal. The incision extended deeply through the Schlemm’s canal with obvious injury to the adjacent deep sclera in the majority of sections ( Figure 1 ). The Trabectome also achieved an opening through the entirety of TM tissue into the Schlemm’s canal. Although the device also removed a large portion of the central TM, significant leaflets of residual tissue still remained. The residual TM demonstrated extensive charring from thermal injury. Tissue debris was also noted to be occluding distal collector channels ( Figure 2 ). Tissue incised with the dual-blade device demonstrated a more complete removal of TM without collateral damage ( Figure 3 ).
Data from human eye perfusion studies are included in the Table . The extent of TM treatment varied between devices and between eyes from 100 to 180 degrees. All 3 treatment modalities achieved a significant reduction in measured IOP 30 minutes after treatment. Treatment with the dual-blade device and Trabectome resulted in a mean IOP reduction of 40% each, whereas the MVR blade achieved a 31% reduction. Although the percentage of IOP decrease was greater for Trabectome and the dual-blade device, there was no statistically significant difference in the IOP lowering between devices (dual-blade/MVR P = .13; dual-blade/Trabectome P = .96; Trabectome/MVR P = .12). There was no correlation between the number of degrees of TM treated and the percentage IOP change for any device (r 2 = 0.077-0.271).
| Eye | Degrees of Angle Treated | Preprocedure IOP | Postprocedure IOP | Absolute IOP Change | Percent IOP Change | P Value | |
|---|---|---|---|---|---|---|---|
| Dual-blade device | 1 | 140 | 17 | 10 | −7 | −41 | |
| 2 | 180 | 19 | 11 | −8 | −42 | ||
| 3 | 130 | 15 | 9 | −6 | −40 | ||
| 4 | 180 | 22 | 14 | −8 | −36 | ||
| Mean | 157.5 ± 26.3 | 18.3 ± 3.0 | 11.0 ± 2.2 | −7.3 | −40 | .00063 | |
| MVR blade | 1 | 180 | 20 | 14 | −6 | −30 | |
| 2 | 180 | 20 | 15 | −5 | −25 | ||
| 3 | 150 | 18 | 12 | −6 | −33 | ||
| 4 | 170 | 16 | 10 | −6 | −38 | ||
| Mean | 170.0 ± 14.1 | 18.5 ± 1.9 | 12.8 ± 2.2 | −5.8 | −31 | .00018 | |
| Trabectome | 1 | 120 | 18 | 12 | −6 | −33 | |
| 2 | 130 | 21 | 12 | −9 | −43 | ||
| 3 | 100 | 17 | 11 | −6 | −35 | ||
| 4 | 120 | 19 | 10 | −9 | −47 | ||
| Mean | 117.5 ± 12.6 | 18.8 ± 1.7 | 11.3 ± 1.0 | −7.5 | −40 | .00324 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


