MECHANISM OF CHOROIDAL NEOVASCULARIZATION
Angiogenesis is the formation of new blood vessels by the sprouting and splitting of existing vessels, a process that is implicated in both normal physiological processes such as the female reproductive cycle, hair growth, and wound healing, and in diseases such as cancer, psoriasis, rheumatoid arthritis, and various ocular conditions (
1). Retinal angiogenesis is central to such conditions as proliferative diabetic retinopathy, advanced retinopathy of prematurity, neovascular glaucoma, and proliferative sickle cell retinopathy (
2). Neovascular (exudative or “wet”) age-related macular degeneration (AMD) arises most often from choroidal neovascularization (CNV) or choroidal angiogenesis. While the most common cause for visual impairment from retinal neovascularization is vitreous hemorrhage, CNV most commonly leads to subretinal hemorrhage and only rarely leads to vitreous hemorrhage. Similarly, while retinal neovascularization can lead to preretinal fibrous proliferation and traction retinal detachment, CNV frequently leads to subretinal fibrous proliferation with disciform scar formation.
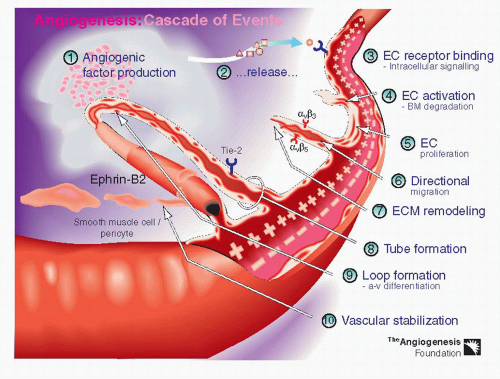
However, the molecular mechanism underlying retinal neovascularization and CNV share similarities, and much of our knowledge of the pathophysiology and therapeutics of CNV arises from experimental models of retinal angiogenesis. Normally, endothelial cells lining the blood vessels are “silent” as a result of an intricate balance of pro-angiogenic and antiangiogenic (angiostatic) factors. Pathological conditions such as hypoxia and ischemia perturb this balance and lead to an increased expression of pro-angiogenic factors and/or a downregulation of angiostatic factors (
3,
4 and
5). The resulting angiogenic drive “activates” endothelial cells and precipitates a complex cascade of cellular events: proliferation of endothelial cells, migration of endothelial cells, tube formation, and ultimately formation of blood vessel loops from endothelial cell tubes (
Fig. 34-1). Many stimulatory and inhibitory molecules are thought to be involved in this complex cascade: growth factors, adhesion molecules, extracellular matrix components, proteinase enzymes, upstream and downstream regulators, and others (
4). Therapies can be targeted at one or more steps of the angiogenic process.
Angiostatic factors include thrombospondin, angiostatin, endostatin, and pigment epithelium-derived factor (PEDF) (
3,
4). Identified pro-angiogenic factors include vascular endothelial growth factor (VEGF), fibroblast growth factor (FGF) families, transforming growth factors-alpha and -beta (TGF-alpha and TGF-beta), angiopoietin-1 and -2 (
3,
4). Members of the VEGF gene family include VEGF-A, VEGF-B, VEGF-C, VEGF-D, VEGF-E, and placental growth factor (PIGF) (
6). VEGF-A has been shown to play a key role in normal vasculogenesis and in angiogenesis in animals and humans, while the role of other members of the VEGF family appears more limited (
6,
7). VEGF-A was first described in 1983 as a “tumor vascular permeability factor” (VPF) that induced vascular leakage in skin (
8). VPF was subsequently found to be identical to an endothelial cell mitogen named VEGF (
9). Therefore, VEGF-A has two important functions: stimulation of angiogenesis and enhancement of vascular permeability. Both these properties are important in the pathogenesis of CNV in neovascular AMD.
As a result of alternative splicing and cleavage of signal sequences, multiple isoforms and cleavage products of VEGF-A exist. There are six major isoforms consisting of 121, 145, 165, 183, 189, and 206 amino acids (
10,
11). VEGF121 is the highly diffusible isoform, while VEGF165 is the most abundantly expressed in humans with only a portion of it being diffusible (
12). Both VEGF189 and VEGF206 are almost completely sequestered in the extracellular matrix (
12). While all isoforms are important in angiogenesis, VEGF165 has been purported to be the isoform most allied with neovascularization (
10,
13). All isoforms can be mobilized through extracellular cleavage by plasmin to generate the diffusible VEGF110 (
14).
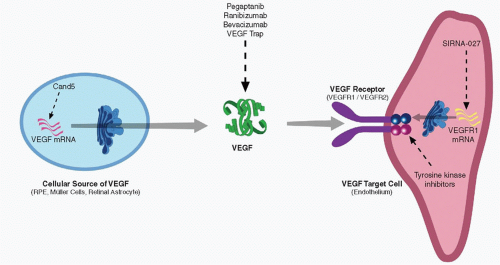
The biological effects of VEGF-A are mediated through the VEGF-specific tyrosine-kinase receptors VEGFR-1 (Flt-1), VEGFR-2 (Flk-1), and VEGFR-3 (Flt-4), and the neuropilins
(NP1, NP2) (
Fig. 34-2) (
6,
15). VEGFR-2 appears to be the main receptor responsible for the angiogenic effects of VEGF-A. NP1 may enhance the effectiveness of VEGFR-2-mediated signaling by presenting isoform VEGF165 to VEGFR-2 (
16). In humans, VEGFR-3 is thought to play a role in lymphangiogenesis (
17).
Laboratory and clinical findings implicate VEGF-A in the pathogenesis of neovascular AMD. VEGF-A expression can be induced in cultured retinal pigment epithelial (RPE) cells under hypoxic conditions (
18,
19). Increased VEGF-A concentrations are found in surgically excised CNV membranes (
20), in vitreous samples from patients with submacular hemorrhage and AMD (
21), and in RPE cells in the early stages of AMD (
22).
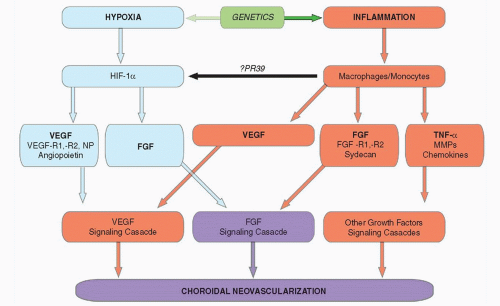
There is increasing evidence supporting the role of inflammation in both nonneovascular and neovascular stages of AMD (
23,
24). The complement system is part of the innate immune system. Polymorphisms in the gene encoding complement factor H, one component of the native immune system, are strongly associated with subtypes of neovascular AMD (
25). An illustration of current understanding of the pathogenesis of neovascular AMD is depicted in
Figure 34-3.