Purpose
To compare the diagnostic accuracy of the pattern electroretinogram (pattern ERG) to that of standard automated perimetry (SAP), short-wavelength automated perimetry (SWAP), and frequency-doubling technology (FDT) perimetry for discriminating between healthy and glaucomatous eyes.
Design
Cross-sectional study.
Methods
Eighty-three eyes of 42 healthy recruits and 92 eyes of 54 glaucoma patients (based on optic disc appearance) from the University of California, San Diego, Diagnostic Innovations in Glaucoma Study were tested with pattern ERG for glaucoma detection (PERGLA; Lace Elettronica, Pisa, Italy), SAP, SWAP, and FDT within 9 months. Receiver operating characteristic (ROC) curves were generated and compared for pattern ERG amplitude and SAP, SWAP, and FDT mean deviation and pattern standard deviation (PSD). Sensitivities and specificities were compared and agreement among tests was described.
Results
The area under the ROC curve for pattern ERG amplitude was 0.744 (95% confidence interval = 0.670, 0.818). The ROC curve area was 0.786 (0.720, 0.853) for SAP PSD, 0.732 (0.659, 0.806) for SWAP PSD, and 0.818 (0.758, 0.879) for FDT PSD. At 95% specificity, sensitivities of SAP and FDT PSD were significantly higher than that of pattern ERG amplitude; at 80% specificity, similar sensitivities were observed among tests. Agreement among tests was slight to moderate.
Conclusion
The diagnostic accuracy of the pattern ERG amplitude was similar to that of SAP and SWAP, but somewhat worse than that of FDT. Nevertheless, the pattern ERG may hold some advantage over psychophysical testing because of its largely objective nature.
Glaucoma is a neurodegenerative disease that results in optic nerve damage and characteristic visual field loss. Although glaucoma is treatable, early detection is important because lowering intraocular pressure reduces the rate of glaucomatous progression. Psychophysical tests of visual function have been developed to detect early glaucomatous visual loss, and standard automated perimetry (SAP) is the current clinical standard. SAP is a relatively nonselective test in that all subtypes of retinal ganglion cells are sensitive to its stimulus. More recently, function-specific tests that target sub-populations of retinal ganglion cells preferentially, though not exclusively, have become available. These function-specific tests include short-wavelength automated perimetry (SWAP) and frequency-doubling technology (FDT) perimetry. SWAP presents a blue light on a yellow background to emphasize the response characteristics of the blue-yellow (koniocellular) pathway and FDT perimetry uses a rapidly reversing contrast grating to emphasize the response characteristics of the magnocellular pathway.
The psychophysical tests of visual function described above are subjective in nature. Their results can be affected by fatigue, inattention to stimulus presentation, and learning effects. Furthermore, the decision criterion can vary from participant to participant. As an example, when testing with psychophysical tests, “trigger-happy” participants are more likely to respond to near-threshold targets than participants that adopt a more conservative decision strategy. Electrophysiological tests of visual function have the advantage of being generally objective and unaffected by patient response. The pattern electroretinogram (pattern ERG) is an electrophysiological test that assesses the function of retinal ganglion cells by isolating the ganglion cell response using a reversing checkerboard or grating pattern that carries no change in average luminance over time. Recently, a pattern ERG measurement paradigm designed specifically for glaucoma detection (pattern ERG for glaucoma detection) has been introduced that attempts to optimize stimulus (eg, short test duration) and recording (eg, use of skin electrodes) characteristics for ease of clinical use.
In the current study we compared the diagnostic accuracy of objective pattern ERG for glaucoma detection to that of the subjective SAP, SWAP, and FDT tests for discriminating between healthy eyes and those with glaucomatous optic neuropathy.
Methods
Participants
Eighty-three healthy eyes of 42 healthy recruits and 92 eyes with glaucomatous-appearing optic discs (ie, glaucomatous optic neuropathy) of 54 glaucoma patients enrolled in the University of California, San Diego Diagnostic Innovations in Glaucoma Study, were included in this study. All eyes were tested with pattern ERG and had good-quality stereophotography of the optic disc and reliable SAP, SWAP, and FDT within 9 months (eyes included were all Diagnostic Innovations in Glaucoma Study eyes meeting these criteria).
In addition to the testing described above, each study participant underwent a comprehensive ophthalmologic evaluation including review of medical history, best-corrected visual acuity testing, slit-lamp biomicroscopy, intraocular pressure (IOP) measurement with Goldmann applanation tonometry, gonioscopy, and dilated slit-lamp fundus examination with a 78-diopter lens. To be included in the study, participants had to have a best-corrected acuity better than or equal to 20/40 at study entry, spherical refraction within ±5.0 diopters (D) and cylinder correction within ±3.0 D, and open angles on gonioscopy. Eyes with coexisting retinal disease, uveitis, or nonglaucomatous optic neuropathy were excluded.
Eyes were classified as healthy or having glaucomatous optic neuropathy based on subjective clinical assessment of stereoscopic optic disc photographs. Simultaneous stereophotographs were obtained after maximal pupil dilation using a Topcon camera (TRC-SS; Topcon Instrument Corp of America, Paramus, New Jersey, USA). Each photograph was assessed by 2 experienced graders using a stereoscopic viewer (Asahi Pentax StereoViewer II; Asahi Optical Co, Tokyo, Japan) and a standard fluorescent light box. Each grader was masked to the participants’ identity, clinical diagnoses, results from the other grader, and other test results. When the 2 graders disagreed, a third experienced grader adjudicated. The classification of glaucomatous optic neuropathy was based on neuroretinal rim-thinning, notching, excavation, or nerve fiber layer thinning (focal or diffuse) characteristic of glaucoma. Neither IOP nor results from perimetric testing were considered when classifying eyes as glaucomatous optic neuropathy. Healthy recruits had appearing optic discs and no history of increased IOP (≥ 22 mmHg) in both eyes, regardless of or perimetric testing results.
The average age (95% confidence interval [CI]) of healthy individuals was 63.6 (61.4, 65.8) years and the average age of patients was 70.4 (68.2, 72.5) years [ P (t) < .02]. Fifty-five of 83 (66%) healthy eyes and 53 of 93 (57%) patients eyes were from females [ P (χ 2 ) = .24]. IOP at the time of pattern ERG recording was similar in healthy and glaucomatous optic neuropathy eyes [14.99 (14.13, 15.84) mm Hg and 14.21 (13.39, 15.02) mm Hg, respectively; P (t) = .19].
Pattern Electroretinogram Testing
A commercially available modification of the Glaid (Lace Elettronica, Pisa, Italy, software version 2.1.14) electrophysiology instrument, called PERGLA, was used to measure the pattern ERG response. The pattern ERG for glaucoma detection stimulus is a black and white (contrast 98%, mean luminance 40 cd/m 2 ), horizontal square wave grating (1.6 c/deg), counter-phasing at 8.14 Hz, presented on a computer monitor (14.1 cm diameter circular field). At a viewing distance of 30 cm, the display subtends 25 degrees centered on the fovea (assuming fixation towards a prominent central fixation circle). Responses from both eyes are measured simultaneously. Electrical signals from silver chloride skin electrodes (9 mm, adhered with conductive cream and tape) (both lower eyelids active, both temples referenced, forehead ground) are fed into a 2-channel differential amplifier, amplified (100 000-fold), filtered (1-30 Hz), and then digitized with 12-bit resolution at 4169 Hz. Before testing, the electrode impedance is monitored automatically and an on-screen indicator signals acceptable impedance (≤5 kΩ). Additionally, an on-screen oscilloscope displays background noise.
The pattern ERG for glaucoma detection software obtains each waveform by averaging 600 artifact-free time-periods (ie, sweeps) of 122.8 milliseconds (msec) each, synchronized with the contrast alternation of the stimulus grating. Two independent response blocks of 330 sweeps each are recorded and separated by a user-defined inter-stimulus interval. For each block, the first 30 sweeps are rejected from the average to eliminate onset effects from the steady-state recording. Sweeps containing spurious signals attributable to blinks and eye movements are rejected over a threshold voltage of ±25 μV. Resulting steady-state pattern ERGs take the form of near-sine waves that are Fourier transformed to isolate the harmonic component at the contrast reversal rate (16.28 reversals per second). In addition, a noise response is obtained by multiplying alternate sweeps by 1 and -1 before averaging. The noise response also is Fourier transformed at the contrast reversal rate to allow calculation of signal to noise ratio. Previous studies indicate that repeatability of pattern ERG for glaucoma detection measurements is excellent.
The same operator (A.T.) tested all participants. Test time was approximately 4 minutes per test, although preparation and electrode placement added approximately 5 minutes per examination. All eyes were refracted, appropriate corrections for viewing distance were made, and near acuity was Jaeger acuity of 1 or better for all participants. Participants were asked to fixate on a small circle in the center of the display screen and the operator monitored fixation subjectively.
Psychophysical Testing
All tests assessed the central 48 degrees (52 test points) of the visual field and required fixation by the patient (monitored by instrument software). Adequate refraction was provided for each device and the pupils had a diameter of at least 3 mm. The pupils were dilated when this requirement was not met. All tests were reliable, defined as false positives ≤15%, fixation losses and false negatives ≤33%, with no observable testing artifacts. The order of testing was randomized across participants.
Standard Automated Perimetry
Each participant was tested using the 24-2 program on the Humphrey Field Analyzer II, using the Swedish Interactive Thresholding Algorithm (SITA) version 4.1 (Carl Zeiss Meditec Inc, Dublin, California, USA). The target used in this achromatic test is a 0.43-degree flash of white light presented on a 10-cd/m 2 background for 200 msec. This instrument and its operation have been described in great detail previously.
Short-Wavelength Automated Perimetry
Each participant was tested using the 24-2 program with SITA (version 4.1). SWAP targets the short-wavelength-sensitive cones and pathway with a bluish (440-nm wavelength) narrow-band target of 1.8 degrees presented for 200 msec on a bright (100 cd/m 2 ) yellow background. The details of this test have been described elsewhere.
Frequency-Doubling Technology
Each participant was tested using FDT with the Humphrey Matrix (ie, 24-2) FDT Visual Field Instrument (Carl Zeiss Meditec, Dublin, California, USA) with Welch-Allyn technology (Skaneateles Falls, New York, USA) and the Zippy Estimation by Sequential Testing (ZEST) thresholding algorithm. FDT measures the contrast necessary to detect vertical grating targets that undergo counter-phase flicker. Each target subtends 5 degrees of visual angle and has a spatial frequency of 0.5 cyc/deg and counter phases with a temporal frequency of 18 Hz. The test is based on the frequency-doubling illusion and is sensitive to glaucomatous visual field loss. The details of the test have been described elsewhere.
Analyses
The Student t tests were used to compare continuous measures of pattern ERG amplitude and SAP, SWAP, and FDT mean deviation (MD) and pattern standard deviation (PSD) between healthy and glaucomatous optic neuropathy eyes. Differences with P ≤ .05 were considered statistically significant. For each visual function test, we generated receiver operating characteristic (ROC) curves for discriminating between healthy and glaucomatous optic neuropathy eyes. The relative diagnostic accuracies of pattern ERG and the psychophysical tests were assessed by comparing areas under the ROC curves. Statistically significant differences between areas under the ROC curves were determined using the method of DeLong and associates.
Using the ROC curve of each test, we derived abnormality cutoffs at moderate (approximately 80%) and high (approximately 95%) specificities for all parameters. The ROC-derived abnormality cutoffs associated with the set specificities of 80% and 95% were applied to classify each test as normal or abnormal to determine sensitivity. Sensitivities at each specificity cutoff were compared among parameters using McNemar’s test.
The level of agreement at the 80% and 95% specificity cutoffs among the tests (pattern ERG amplitude, SAP PSD, SWAP PSD, FDT PSD) also were assessed using the kappa (κ) statistic. Kappa values range from 0 to 1, with values between 0.00 and 0.20 indicating slight agreement, 0.21 and 0.40 indicating fair agreement, 0.41 and 0.60 indicating moderate agreement, 0.61 and 0.80 indicating substantial agreement, and 0.81 and 1.00 indicating almost-perfect agreement.
Results
Table 1 compares pattern ERG for glaucoma detection amplitude and SAP, SWAP, and FDT MD and PSD between healthy and glaucomatous optic neuropathy eyes. Significant differences (all P (t) ≤ .04) were found for all parameters between groups. Pattern ERG amplitude was decreased by approximately 31% in glaucomatous optic neuropathy eyes compared to healthy eyes, SAP threshold (most appropriate perimetry analog for pattern ERG amplitude) was decreased by approximately 9%, SWAP threshold was decreased by approximately 11%, and FDT threshold was decreased by approximately 18%.
| GON Mean (95% CI) | Healthy Eyes Mean (95% CI) | ANOVA P | |
|---|---|---|---|
| SAP | |||
| MD (dB) | −2.53 (−3.06, −2.01) | −0.12 (−0.67, 0.43) | <.0001 |
| PSD (dB) | 3.73 (3.22, 4.23) | 1.67 (1.14, 2.21) | <.0001 |
| SWAP | |||
| MD (dB) | −5.55 (−6.34, −4.76) | −4.32 (−5.16, −3.49) | .04 |
| PSD (dB) | 4.55 (4.15, 4.95) | 2.92 (2.49, 3.34) | <.0001 |
| FDT | |||
| MD (dB) | −3.80 (−4.55, −3.05) | 0.15 (−0.64, 0.94) | <.0001 |
| PSD (dB) | 4.50 (4.18, 4.83) | 2.78 (2.44, 3.12) | <.0001 |
| Pattern ERG | |||
| Amplitude (μV) | 0.53 (0.48, 0.59) | 0.77 (0.72, 0.83) | <.0001 |
Table 2 shows the areas under the ROC curves obtained for each parameter from each test, the 95% CI associated with each area under the ROC curve, and the sensitivities at set specificities of approximately 80% and 95%. We compared pattern ERG amplitude to SAP, SWAP, and FDT tests based on the abnormality cutoffs obtained for PSD only. Two reasons prompted this decision: 1) the ROC-derived PSD was the best parameter (that with the highest area under the ROC curve) for all 3 tests; and 2) PSD performs better at distinguishing between normal and glaucoma subjects than MD, although MD may be better for determining progression. Figure 1 shows the ROC curves for pattern ERG amplitude and SAP, SWAP, and FDT using PSD. The area under the ROC curve for FDT PSD (0.818) was significantly greater than that obtained for pattern ERG amplitude (0.744; P = .04). No statistically significant differences were observed between pattern ERG ROC curve area and those of SAP PSD (0.786; P = .17) and SWAP PSD (0.732; P = .41).
| AUC | 95% CI | Specificity/Sensitivity (%) | Criteria for 80% Specificity | Specificity/Sensitivity (%) | Criteria for 95% Specificity | |
|---|---|---|---|---|---|---|
| SAP | ||||||
| MD (dB) | 0.786 | 0.719, 0.853 | 81/65 | −0.89 | 95/41 | −1.92 |
| PSD (dB) | 0.786 | 0.720, 0.853 | 80/62 | 1.82 | 95/36 | 2.81 |
| SWAP | ||||||
| MD (dB) | 0.570 | 0.485, 0.656 | 80/33 | −7.36 | 95/19 | −9.22 |
| PSD (dB) | 0.732 | 0.659, 0.806 | 81/53 | 3.50 | 95/29 | 4.73 |
| FDT | ||||||
| MD (dB) | 0.789 | 0.722, 0.856 | 80/64 | −1.70 | 95/29 | −5.29 |
| PSD (dB) | 0.818 | 0.758, 0.879 | 80/61 | 3.19 | 95/46 | 3.86 |
| Pattern ERG | ||||||
| Amplitude (μV) | 0.744 | 0.670, 0.818 | 80/52 | 0.52 | 95/20 | 0.32 |
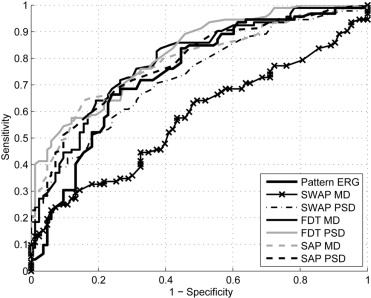
At 95% specificity, 18 of 92 (19.6%) of glaucomatous optic neuropathy eyes had an abnormal pattern ERG amplitude, 33 (35.9%) had an abnormal SAP PSD, 27 (29.3%) had an abnormal SWAP PSD, and 42 (45.7%) had an abnormal FDT PSD. McNemar’s tests showed that the sensitivities of SAP ( P = .02) and FDT ( P = .0002) were significantly greater than that of pattern ERG. No significant difference was found between the sensitivities of SWAP and pattern ERG ( P = .05). At 80% specificity, McNemar’s test showed no significant difference between the sensitivities of pattern ERG and those of SAP ( P = .20), SWAP ( P = .90), and FDT ( P = .23).
At 95% specificity, there was slight agreement between pattern ERG amplitude and SAP PSD (76% agreement, κ = 0.14, CI = 0.00, 0.31) and between pattern ERG amplitude and FDT PSD (74% agreement, κ = 0.19, CI = 0.03, 0.34), while the agreement between pattern ERG and SWAP PSD was fair (82% agreement, κ = 0.30, CI = 0.12, 0.49). Agreement among SAP, SWAP, and FDT PSD was substantial for all pairs ( Table 3 ). At 80% specificity, slight agreement was found between pattern ERG amplitude and SWAP PSD, while pattern ERG amplitude showed fair agreement with SAP and FDT PSD. There was fair to moderate agreement among the psychophysical tests.



