Purpose
To compare patient-centered outcomes in patients with proliferative diabetic retinopathy (PDR) treated with ranibizumab vs panretinal photocoagulation (PRP).
Design
Randomized clinical trial.
Methods
Setting : Multicenter (55 U.S. sites). Patient Population : Total of 216 adults with 1 study eye out of 305 adults (excluding participants with 2 study eyes, because each eye received a different treatment) with PDR, visual acuity 20/320 or better, no history of PRP. Intervention : Ranibizumab (0.5 mg/0.05 mL) vs PRP. Main Outcome Measures : Change from baseline to 2 years in composite and prespecified subscale scores from the National Eye Institute Visual Function Questionnaire-25 (NEI VFQ-25), University of Alabama Low Luminance Questionnaire (UAB-LLQ), and Work Productivity and Activity Impairment Questionnaire (WPAIQ).
Results
For the NEI VFQ-25 and UAB-LLQ composite scores, ranibizumab–PRP treatment group differences (95% CI) were +4.0 (-0.2, +8.3, P = .06) and +1.8 (-3.5, +7.1, P = 0.51) at 1 year, and +2.9 (-1.5, +7.2, P = .20) and +2.3 (-2.9, +7.5, P = .37) at 2 years, respectively. Work productivity loss measured with the WPAIQ was 15.6% less with ranibizumab (-26.3%, −4.8%, P = .005) at 1 year and 2.9% (-12.2%, +6.4%, P = .54) at 2 years. Eighty-three ranibizumab participants (97%) were 20/40 or better in at least 1 eye (visual acuity requirement to qualify for an unrestricted driver’s license in many states) at 2 years compared with 82 PRP participants (87%, adjusted risk ratio = 1.1, 95% CI: 1.0, 1.2, P = .005).
Conclusions
Though differences in some work productivity and driving-related outcomes favored ranibizumab over PRP, no differences between treatment regimens for PDR were identified for most of the other patient-centered outcomes considered.
The Diabetic Retinopathy Clinical Research Network ( DRCR.net ) Protocol S, Prompt Panretinal Photocoagulation (PRP) vs Intravitreal Ranibizumab with Deferred Panretinal Photocoagulation for Proliferative Diabetic Retinopathy (PDR), determined that ranibizumab resulted in visual acuity not more than 5 letters worse than PRP treatment at 2 years. Secondary outcomes favoring ranibizumab included better average visual acuity over the course of 2 years, less visual field loss, fewer vitrectomies, and lower likelihood of developing diabetic macular edema (DME) with vision loss, supporting the consideration of ranibizumab as a treatment alternative to PRP for patients with PDR. However, ranibizumab does involve a greater number of treatments and visits than PRP. Over 2 years, eyes in the ranibizumab group had a median of 10 injections (for PDR and DME) and 22 visits compared with a median of only 1 injection (for DME) and 16 visits (over 2 years) in the PRP group. Of note, supplemental PRP was performed in 45% of the eyes in the PRP group.
The analysis of 2-year results did not identify statistically significant treatment group differences in the composite or subscale scores of the National Eye Institute Visual Function Questionnaire-25 (NEI VFQ-25) or the University of Alabama at Birmingham Low Luminance Questionnaire (UAB-LLQ). In the current report, the results of additional analyses of these questionnaires as well as the Work Productivity and Activity Impairment Questionnaire (WPAIQ) are provided. These patient-centered outcomes could influence treatment decisions for using anti–vascular endothelial growth factor therapy in the management of PDR. Additional outcomes related to driving also are considered.
Methods
Overview
The DRCR.net conducted a multicenter (55 U.S. sites) randomized trial of 394 eyes among 305 adults enrolled between February and December 2012 ( clinicaltrials.gov Identifier: NCT01489189 ). Participants were at least 18 years old and had type 1 or type 2 diabetes mellitus. The study eye had PDR, no history of PRP, and a best-corrected visual acuity letter score of 24 or better (approximate Snellen equivalent 20/320 or better). Eyes with or without central-involved DME were eligible. Eyes were randomly assigned to prompt PRP or ranibizumab injections (Lucentis; Genentech Inc, South San Francisco, California, USA). The use of ranibizumab for diabetic retinopathy only in the presence of DME is listed as an indication within its prescribing information in the United States. Both groups received ranibizumab injections as needed for DME. Participants and investigators were unmasked to study treatment. The study adhered to the Declaration of Helsinki and was approved by multiple institutional review boards. The protocol and statistical methods have been reported previously, and more details can be found in the Supplemental Appendix (Supplemental Material available at AJO.com ).
At baseline, 1 year, and 2 years, participants completed the NEI VFQ-25 and UAB-LLQ. The WPAIQ was completed at baseline, at 4 weeks, and at each assessment visit (16, 32, 52, 68, 84, and 104 weeks). All subsequent analysis of DRCR.net data for this report included the 216 study participants (102 in the ranibizumab group and 114 in the PRP group) with only 1 study eye. The 89 participants with both eyes enrolled in the trial were not included because 1 eye was assigned to each treatment group. Because surveys were administered at the patient level and treatment was administered at the eye level, a treatment group comparison is not possible. Electronic-Early Treatment Diabetic Retinopathy Study (E-ETDRS) visual acuity testing following a protocol refraction was performed in each eye along with binocular E-ETDRS visual acuity testing with the participant’s habitual correction (“everyday” glasses or contacts).
Questionnaires
The NEI VFQ-25 is a 25-item questionnaire measuring self-reported vision-targeted health status. It contains 1 question on general health and 11 vision-related scales: general vision, ocular pain, near activities, distance activities, social functioning, mental health, role difficulties, dependency, driving, color vision, and peripheral vision. A composite score averages each of the vision-related scales.
The UAB-LLQ is a 32-item questionnaire originally developed to assess vision under low luminance and at night in studies of age-related macular degeneration. It is composed of the following 6 scales, which are averaged to form a composite score: driving, extreme lighting, mobility, emotional distress, general dim lighting, and peripheral vision.
The WPAIQ for specific health problems is a 6-item questionnaire yielding 4 scores related to employment and everyday activities: absenteeism (percent work time missed owing to vision), presenteeism (percent impairment while working owing to vision), work productivity loss (percent overall work impairment owing to vision; a composite of the absenteeism and presenteeism scales), and activity impairment (percent activity impairment owing to vision).
The NEI VFQ-25 and UAB-LLQ were interviewer-administered with all instructions read aloud, or self-administered in the clinic or at home up to 14 days before the study visit. The WPAIQ was always interviewer-administered. Spanish versions were available for the NEI VFQ-25 and WPAIQ but not the UAB-LLQ. Fifteen participants who completed the NEI VFQ-25 and WPAIQ at randomization in Spanish did not complete the UAB-LLQ.
Outcomes
For each of the questionnaire subscales the following outcomes were prespecified: (1) the mean change from baseline to 1 and 2 years, and (2) for the NEI VFQ-25 and UAB-LLQ, the proportion of participants gaining or losing at least 10 points (changes judged to be clinically relevant). These same outcomes were evaluated post hoc for the NEI VFQ-25 and UAB-LLQ composite scores. The following subscales were designated a priori as primary interest because investigators believed they were the ones most likely to show a difference: driving, peripheral vision, and color vision from the NEI VFQ-25; driving, peripheral vision, and general dim lighting from the UAB-LLQ; and work productivity loss from the WPAIQ.
Because patient-centered outcomes can differ substantially by whether the study eye is the better- or worse-seeing eye, a post hoc subgroup analysis of the outcomes described above was performed using continuous outcome data where the study eye was the better-seeing eye, as defined previously. A second post hoc subgroup analysis was conducted to determine if the treatment effect differed by whether the study eye had DME at baseline, for which ranibizumab was required by protocol in both groups (based on sex- and machine-specific thresholds of retinal thickness and visual acuity impairment of 20/32 or worse).
Because the driving subscales were of particular interest, 2 driving-related outcomes were prespecified: (1) number (%) of participants driving at baseline who stopped driving wholly or partly because of vision and (2) number (%) of participants driving at night at baseline who stopped driving at night wholly or partly because of vision. In addition, the following driving-related outcomes were evaluated post hoc: (1) the number (%) of participants driving among participants who, at baseline, were driving or had given up driving wholly or in part because of vision, (2) number (%) of participants with visual acuity letter score (approximate Snellen equivalent) of at least 69 (20/40 or better, the legal limit for driving in many U.S. states) (a) in at least 1 eye, regardless of which eye was the study eye, (b) in the better-seeing eye, for participants whose study eye is the better-seeing eye, and (c) using both eyes (binocular visual acuity).
Change in visual acuity, as well as visual acuity area under the curve (AUC), which were evaluated for the full cohort in the primary manuscript, were evaluated post hoc for the subgroup of participants with 1 study eye. AUC analyses were also conducted post hoc for the NEI VFQ-25 and UAB-LLQ composite scores and the WPAIQ work productivity loss score.
Statistical Analyses
Mean change in questionnaire scores from baseline was analyzed with analysis of covariance adjusting for baseline score and optical coherence tomography (OCT) central subfield thickness. Missing outcomes were imputed using last observation carried forward. Sensitivity analyses included only observed data ( Supplemental Tables 18–30 ; Supplemental Material available at AJO.com ).
For dichotomous outcomes, Poisson regression with a robust error variance was used to estimate the relative risk while adjusting for OCT central subfield thickness and baseline outcome values. Only participants who completed the 2-year visit were included in AUC analyses. AUC was calculated according to the trapezoidal rule and is analogous to taking a weighted average of the outcome over the course of the study.
To control the type I error rate, P < .01 was prespecified as statistically significant for analyses of the subscales of primary interest and suggestive of a difference for all other subscales, with 99% confidence intervals presented. For all other outcomes, P < .05 was considered suggestive of a difference, rather than definitive, with 95% confidence intervals (CI) presented. All statistical analyses were conducted using SAS software, Version 9.4 (SAS Institute Inc, Cary, North Carolina, USA).
Results
Relevant baseline characteristics and questionnaire scores appeared balanced by treatment group ( Supplemental Tables 1 and 2 ; Supplemental Material available at AJO.com ). The median age was 54, 95 (44%) were women, 119 (55%) were white, 40 (19%) had type 1 diabetes, and the median HbA 1c was 8.5%. Excluding deaths, overall completion rates for the NEI VFQ-25, UAB-LLQ, and WPAIQ were 86%, 88%, and 88%, respectively, at 1 year and 85% at 2 years for all 3 questionnaires ( Supplemental Table 3 ; Supplemental Material available at AJO.com ). The NEI VFQ-25 and UAB-LLQ were interviewer-administered 53% and 52% of the time.
The treatment group difference (ranibizumab group outcome minus PRP group outcome) in visual acuity, adjusted for baseline vision and central subfield thickness, was similar to the full cohort: +8.7 (95% CI: +5.3, +12.1, P < .001) at 1 year and +3.9 (95% CI: −0.0, +7.9, P = .05) at 2 years. The change in visual acuity over the course of 2 years (AUC) was greater with ranibizumab by +6.2 letters (95% CI: +3.9, +8.4, P < .001).
National Eye Institute Visual Function Questionnaire-25
At 1 year, composite score change from baseline was greater with ranibizumab, although not statistically significant (+4.0, 95% CI: −0.2, 8.3, P = .06), as was the case among all other subscales ( Figure 1 , Supplemental Table 4 ; Supplemental Material available at AJO.com ). At 2 years, the difference in composite score change from baseline was +2.9 (95% CI: −1.5, +7.2, P = .20) and changes in prespecified subscales of driving, color vision, and peripheral vision were not significantly different between treatment groups. Results were similar for the analysis of gain/loss of 10 points and the better-seeing eye subgroup ( Supplemental Tables 5 and 6 ; Supplemental Material available at AJO.com ), as well as for gain/loss of 5 points (data not shown).The difference in composite score change over the course of 2 years was greater with ranibizumab by +3.1 (95% CI: +0.0, +6.2, P = .050).
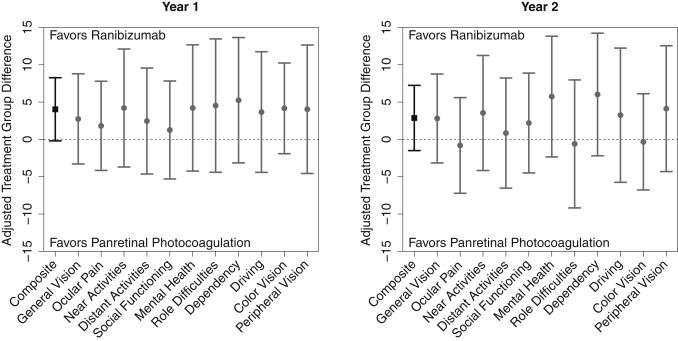
At 1 year, the treatment effect for the driving subscale (interaction P = .002) differed by whether the study eye had DME at baseline ( Supplemental Table 7 ; Supplemental Material available at AJO.com ). Specifically, eyes with baseline DME (n = 36) tended to have better outcomes with PRP, while eyes without baseline DME (n = 146) tended to have better outcomes with ranibizumab. For example, the difference in driving subscale change from baseline was −16.9 (99% CI: −35.4, +1.7) less with ranibizumab for eyes with DME vs +8.2 (95% CI: −0.4, +16.7) more with ranibizumab for eyes without DME. This interaction was not observed at 2 years (interaction P = .69 for driving subscale).
University of Alabama Low Luminance Questionnaire
Change from baseline to 1 year was numerically greater with ranibizumab among all subscales and the composite score (+1.8, 95% CI: −3.5, +7.1, P = .51), although not statistically significant ( Figure 2 , Supplemental Table 8 ; Supplemental Material available at AJO.com ). At 2 years, there were also no significant differences identified in the composite score (+2.3, 95% CI: −2.9, +7.5, P = .37) or in any of the subscales. Results were similar for the analysis of gain/loss of at least 10 points and the better-seeing eye subgroup ( Supplemental Tables 9 and 10 ; Supplemental Material available at AJO.com ), as well as for gain/loss of at least 5 points (data not shown). The change in composite score AUC was +1.2 greater with ranibizumab, although not statistically significant (95% CI: −2.5, +4.9, P = .53).
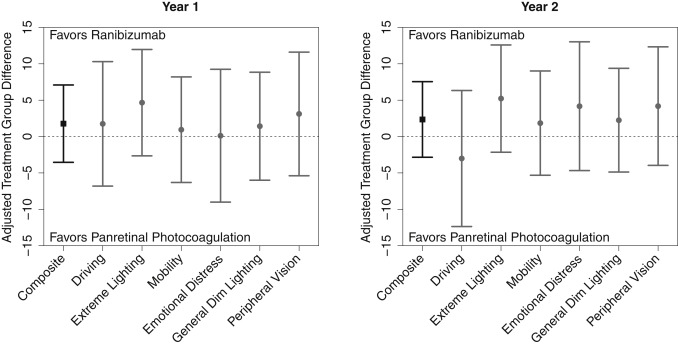
The treatment effect on the change in composite score (interaction P = .008) and mobility subscale (interaction P = .002) at 1 year, but not 2 years ( P = .36, P = .05, respectively), differed by whether the study eye had baseline DME ( Supplemental Table 11 ; Supplemental Material available at AJO.com ). Eyes with baseline DME (n = 34) tended to have better outcomes with PRP, while eyes without baseline DME (n = 139) tended to have better outcomes with ranibizumab.
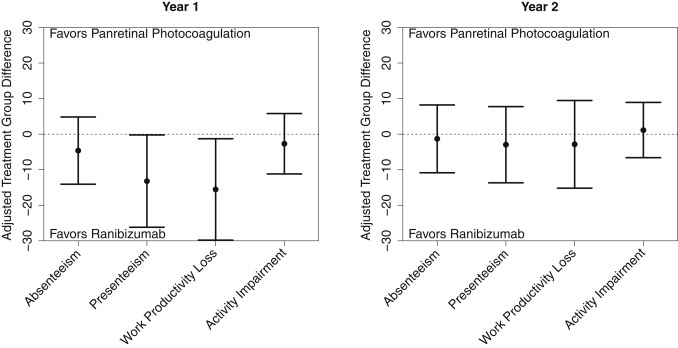
Work Productivity and Activity Impairment Questionnaire
The change from baseline ( Figure 3 , Supplemental Table 12 ; Supplemental Material available at AJO.com ) in all 4 WPAIQ scores was numerically greater with ranibizumab at 1 year. This included a significant difference in the prespecified scale of interest, the work productivity loss score (-15.6%, 99% CI: −29.8%, −1.3%, P = .005), indicating less productivity loss with ranibizumab vs PRP. No significant differences were identified at 2 years, including the work productivity loss score (-2.9%, 99% CI: −15.2%, +9.5%, P = .54). The AUC analysis of change in the work productivity loss scale demonstrated a difference of −10.3% (95% CI: −16.6%, −4.0%, P = .002), indicating less productivity loss with ranibizumab compared with PRP over the course of 2 years.