Purpose
To evaluate the efficacy of the same-size host and donor trephine on reducing myopic refractive errors for patients with keratoconus when deep anterior lamellar keratoplasty (DALK) was used.
Design
Randomized controlled clinical trial.
Methods
One hundred eighteen eyes of 118 patients with keratoconus were enrolled. Using stratified blocked randomization, eligible eyes were allocated into the same-size trephine group or oversize trephine group. Postoperative uncorrected visual acuity (UCVA), best spectacle-corrected visual acuity (BSCVA), spherical equivalent (SE), topography data, and eye axial length were compared at each year for 5 years.
Results
The same-size trephine group had comparable UCVA to the oversize trephine group through 5 years after surgery. But the former had better BSCVA than the latter after 3 years of follow-up. At 5 years, mean BSCVA was 0.17 ± 0.10 logMAR in the same-size trephine group vs 0.25 ± 0.13 logMAR in the oversize trephine group ( P = .03). The same-size trephine group had lower topographic power than the oversize trephine group after 3 years of follow-up. At 5 years, mean topographic power was 45.30 ± 2.28 diopters (D) in the same-size trephine group vs 46.75 ± 2.60 D in the oversize trephine group ( P = .006). Eye axial lengths at 5 years were longer than those preoperatively as well as at 1 year follow-up after surgery in both groups.
Conclusions
The same-size host and donor trephine could reduce late-stage myopic refractive errors for the patients with keratoconus after DALK. The mechanism may be late-stage axial length increase with time.
Keratoconus is a, ectatic corneal disease characterized by the progressive forward extension of the corneas, along with thinning of the corneal stroma, which can lead to progressive irregular astigmatism. For patients with advanced keratoconus, high myopia and astigmatism due to progressive conical protrusion of the cornea, rather than an opacified cornea, is the most important factor responsible for poor visual acuity. Hence, the major goal of corneal transplantation for keratoconus is to restore the normal corneal curvature to facilitate vision correction. Though penetrating keratoplasty (PK) is a traditional technique for the treatment of keratoconus, deep anterior lamellar keratoplasty (DALK) is a replaceable surgical technique for PK, since it can preserve the patient’s own endothelium, thereby reducing the prevalence of immunologic reactions. To date, many studies have reported that the clinical outcomes from DALK are comparable to those from PK for the treatment of advanced keratoconus.
However, there are other studies that found that DALK is not superior to PK in some aspects. For instance, a series of studies have reported that DALK produced significantly higher myopia than PK. For the patients with keratoconus, postoperative myopic tendencies after DALK, along with preoperative preexisting refractive issues, may highlight the refractive irregularities; thereby, it may be difficult to attain optimal visual acuity.
Meanwhile, many previous studies had indicated that an increase in postoperative myopia is associated with the use of oversize donor buttons for patients with keratoconus when PK was conducted. The use of the same-size host and donor trephine could reduce the refractive errors after PK, although some complications, such as wound leak or iris prolapse, could occur in PK. However, unlike PK, an open-sky procedure, DALK is a closure surgical procedure, which eliminates the possibility of those complications.
Hence, we proposed that the same-size donor and recipient trephine may reduce refractive errors for patients with keratoconus after DALK. In this comparative case series, we describe the same-size donor DALK for the treatment of keratoconus and evaluate its clinical outcomes in comparison with conventional oversize donor DALK.
Methods
Ethics Statement
This randomized controlled clinical trial adhered to the tenets of the Declaration of Helsinki. The Ethics Committee of Zhongshan Ophthalmic Center (Guangzhou, China) approved the study protocol on July 14, 2006. Informed consent forms were signed by all participants.
Patients
Between September 5, 2007 and August 11, 2010, 118 patients (118 eyes) with keratoconus were enrolled in this randomized controlled clinical trial. The participants were stratified into 2 strata based on preoperative keratometry (≥60 diopters [D] or <60 D). Using stratified block randomization, the patients were assigned to the same-size donor group or oversize donor group in each stratum. Each group included 59 patients. The Table summarizes the characteristics and preoperative baseline data of patients. We limited the indication for surgery to intolerance to rigid gas-permeable contact lenses or unsuccessful fitting of the contact lens and poor spectacle-corrected visual acuity. Preoperatively, A- or B-scan ultrasonography was arranged as a routine examination. The patients with a feature of posterior scleral staphyloma were excluded from this study. Also, the patients with a history of hydrops or scarring down to the Descemet membrane were excluded from this study. The eyes without Descemet membrane exposure or with intraoperative and postoperative complications (eg, perforation of deep stroma or tearing of Descemet membrane, double anterior chamber, graft failure, or subsequent grafts) were excluded from visual, refractive, and topographic outcomes analysis ( Table ).
| Same-Size Group | Oversize Group | P Value | |
|---|---|---|---|
| Number of eyes | 59 | 59 | |
| Male: Female | 35:24 | 37:22 | |
| Age (mean ± SD) (y) | 24.69 ± 4.26 | 24.95 ± 4.50 | .773 |
| Preoperative BSCVA (logMAR) (Snellen equivalent) | 0.71 ± 0.10 (20/100) | 0.72 ± 0.09 (20/100) | .617 |
| Preoperative keratometry (D) | 57.37 ± 5.11 | 57.12 ± 5.97 | .462 |
| Preoperative refractive sphere (D) | −14.43 ± 4.16 | −13.88 ± 4.08 | .378 |
| Preoperative manifest astigmatism (D) | −6.49 ± 1.98 | −6.78 ± 0.84 | .522 |
| Preoperative axial length (mm) | 25.15 ± 1.18 | 25.22 ± 1.17 | .726 |
Main Outcome Measures
Main eye examinations, including uncorrected visual acuity (UCVA), best spectacle-corrected visual acuity (BSCVA), spherical equivalent (SE), topography data, and eye axial length were obtained at scheduled intervals by 1 of the 2 investigators (H.T., O.Y.). Corneal topography was performed using Orbscan П (Orbtek Inc, Salt Lake City, Utah, USA). Main outcomes were examined once a year for 5 years after surgery. The outcomes at each year of follow-up were compared between 2 groups.
Surgical Procedure
All patients underwent surgery by a single surgeon (H.T.) at Zhongshan Ophthalmic Center. Retrobulbar anesthesia was administrated in all surgeries.
DALK was carried out using the big-bubble technique with some modifications. In brief, a partial-depth incision of the recipient cornea was carried out with a Barron trephine (Katena, Denville, New Jersey, USA). The size of the recipient trephination was chosen to excise the entire cone beyond the iron ring. The trephine sizes were 7.75–8.25 mm in diameter. The trephination was set to cut 375 μm into the corneal stroma. A superficial keratectomy was carried out to reduce the corneal stroma with a crescent knife. A 30 gauge needle was attached to a 5 mL air-filled syringe. The needle was bent 60 degrees at 5 mm from the tip and inserted into the deep stroma with the bevel facing downward. Air was injected into the stroma in order to form a large air bubble between the Descemet membrane and corneal stroma. The space between the corneal stroma and Descemet membrane was maintained by injection of viscoelastic material. The posterior stroma was removed using blunt-tipped scissors, thereby exposing the Descemet membrane. If a big bubble could not be formed using the above technique, a layer-by-layer manual stromal dissection was carried out. Using this method, we undertook very deep lamellar grafting in which a very thin stromal layer was retained.
The donor buttons stored in EUSOL-C corneal storage media (Alchimia, Padova, Italy) for 15–30 days were used for all surgeries. The donor corneas were punched out with a Barron donor punch from the endothelial side. In the same-size group, the diameter of the donor punch was the same as the recipient trephination. In the oversize group, the diameter of the donor punch was 0.25 mm larger than the recipient bed. Before the donor buttons were punched, the Descemet membrane with endothelial layer was removed with a wet sponge. The 10-0 nylon suture was used to suture the button with 1 running, 20-bite suture. Complete suture removal was completed by 18 months; earlier removal was administered in the patients who presented with loosened sutures or vascularization of host cornea.
Postoperative Management
When complete corneal re-epithelialization was achieved, topical dexamethasone 0.1% was administrated 4 times a day for 1 month. Thereafter, topical dexamethasone 0.1% was reduced to 2 times a day for the second month and once a day for the third month. Topical steroid was discontinued 3 months after surgery. Topical artificial tears were applied 4 times a day for 12 months.
Statistical Analysis
Data were described by means ± standard deviation (SD) for continuous variables. SAS for Windows Version 8.1 (SAS Institute Inc, Cary, North Carolina, USA) was used for statistical analysis. Means using descriptive statistics were used to compare the treatment groups with respect to age, preoperative BSCVA, keratometry and manifest sphere, follow-up time, and time to suture removal. Analysis of covariance was used to compare the treatment groups for visual outcomes, eye axial length, spherical equivalent, and topography data analysis. Data on complications (including perforation of the deeper stroma, graft–host interface opacity, stromal rejection, suture loosening, vascularization of the host cornea, and elevation of intraocular pressure) and eyes that had hyperopic spherical equivalent errors were analyzed with Pearson χ 2 or Fisher exact test; P < .05 was considered significant.
Results
Visual Acuity
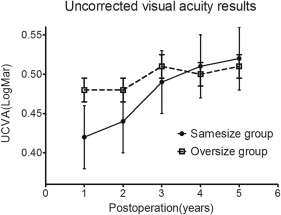
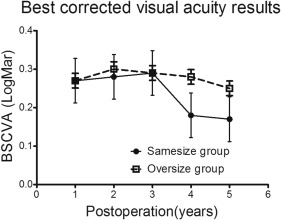
Figures 1 and 2 show UCVA and BSCVA after DALK treatment, respectively. The same-size and oversize groups were comparable in UCVA through 5 years of follow-up. The same-size and oversize groups were comparable in BSCVA through 3 years after surgery, but the former had better BSCVA levels than the latter after 3 years of follow-up. At 5 years, the same-size group had a mean BSCVA of 0.17 ± 0.10 logMAR (logarithm of the minimal angle of resolution); mean BSCVA was 0.25 ± 0.13 logMAR for the oversize group ( P = .03). A total of 40.00% (18/45) of eyes in the same-size group and 19.15% (9/47) of eyes in the oversize group reached 20/25 vision or better ( P = .028). At the 5-year postsurgical visit, the methods of achieving BSCVA seem to be identical in both groups. Twenty-three (23/45, 51.11%) and 26 (26/47, 55.32%) eyes were corrected with spectacles ( P = .686), whereas 18 (18/45, 40.00%) and 16 (16/47, 34.04%) eyes required rigid gas-permeable contact lens correction for BSCVA ( P = .554) in the same-size and oversize groups, respectively. Only 4 (4/45, 8.89%) and 5 (5/47, 10.64%) eyes did not require correction ( P > .999) in the same-size and oversize groups, respectively ( Figures 1 and 2 ).
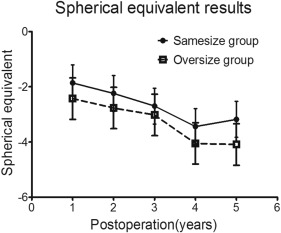
Spherical Equivalent
The same-size trephine group had a lesser degree of SE than the oversize trephine group after surgery, but the difference had no statistical significance between the 2 groups. Mean SE was −1.86 ± 2.88 D vs −2.43 ± 3.08 D ( P = 0.256) at 1 year, −2.25 ± 3.23 D vs −2.77 ± 3.32 D ( P = .456) at 2 years, −2.71 ± 3.13 D vs −3.02 ± 2.97 D ( P = .248) at 3 years, −3.45 ± 2.92 D vs −4.05 ± 3.34 D ( P = .206) at 4 years, and −3.18 ± 3.24 D vs −4.09 ± 2.40 D ( P = .240) at 5 years for the same-size and oversize groups, respectively. At 5 years of follow-up, 17.78% (8/45) of eyes in the same-size group and 8.51% (4/47) of eyes in the oversize group had a hyperopic spherical equivalent ( P = .226). Figure 3 demonstrates the changes of spherical equivalent in treatment groups during follow-up.