Orbital Surgery
Introduction
In Chapter 1, we first met the patient with an assumed orbital tumor. We listened to the story of progression since the onset of the problem, measured, and recorded the physical and clinical signs and symptoms of orbital presentation, and determined the functional status of the ocular apparatus on the side of the affected orbit. This assessment, although not necessarily providing an accurate orbital diagnosis, is a baseline for future reference and defines the orbital disability that the patient expects to be resolved.
In most patients the problem is protrusion of an eye, alerting the physician to the probable need for surgical removal of an offending tumor, if the mass subsequently proves not to be amenable to nonsurgical methods of therapy. In a lesser number of patients, but one of great importance to the affected individual, the problem is loss of vision. Here, the physician is confronted with the question of whether expectant therapy of any type will make the vision better or worse.
In Chapter 2 of Section I, the “nitty-gritty” of preliminary diagnosis is further refined by a discussion of the principles of computed tomography, magnetic resonance imaging, echography, arteriography, venography, conventional radiography, and fine-needle aspiration biopsy, and their respective roles in the study of orbital tumors. Of these auxiliaries to diagnosis, computed tomography, and magnetic resonance imaging are the most important, and a comparison of their respective roles in diagnosis is again noted in Table 21.1 (see also Chapter 2).
After a long interlude of devoting chapters in Section II of the text to the classification, incidence, clinical features, imaging aspects, pathology, course, and management of the individual tumors, we now address the thrust and contents of Section III. Here, we will review the major surgical approaches to orbital tumors because surgery provides the most accurate access to orbital tissue for diagnosis and is the principal means for the eradication of most tumors.
We are omitting the remarks in the previous editions of this text concerning preoperative preparation of the patient, methods of anesthesia, postoperative care of the patient, the instrumentation necessary for orbital surgery, and the historical aspects of the subject. Nowadays most surgeons who work in this field have usually devoted a year or more to the study of the orbit beyond the usual residency training. Thus, they have an ample grasp of these ancillaries to the surgical management of orbital tumors.
In conclusion, the principal thrust of Section III focuses on the technical aspects of the various surgical approaches, augmented by descriptive modifications and “tricks-of-the-trade” that have proved useful to us.
Orbitotomy and Orbitectomy
In the several centuries that barbers, medical practitioners, and surgeons have been removing or incising various lumps or festerings in the orbital space, the orbitotomy—the operation of incising or opening into the orbit through the orbital margin (Dorland, 1985)—has been the mainstay approach. The first major departure from this oft-used route was an entry through the thin lateral wall of the orbit, the Krönlein (1888) approach. This operation was designed to provide better access to the posterotemporal orbit (the home of most orbital tumors) and bypassed the orbital margin. Subsequently, this operation was modified by many surgeons to include a bone flap encompassing the lateral orbital rim, and thereafter named the anterolateral or lateral orbital approach.
Other routes of entry into the orbit that evolved next did not include the orbital margin. One of these was the frontal craniotomy championed by Dandy (1941) that, by removing the orbital roof, improved access to the superoposterior orbit and the orbital face of the optic canal. An alternate route to the orbital apex as well as to the posteromedial orbit soon followed. This involved removal of the medial wall of the orbit through an intranasal approach. A recent modification in the last decade or so has been the elevation of roughly the upper one-fourth of the bony orbit, including the orbital rim, by the coronal approach. The most recent modifications of an orbitotomy are the lateral orbitotomy without the use of a bone flap and the transcaruncular medial orbitotomy.
We have broadened the definition of an orbitotomy to include all of these modifications for the purpose of our discussion. Our exposition of the subject starts with the oldest, straightforward, anterior approach.
Table 21.1 Comparison of Computed Tomography and Magnetic Resonance in Orbital Diagnosis | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||
Anterior Routes
Anterior routes include the superior, inferior, medial, and transconjunctival approaches. All are used less often when compared to their status in the 1973 edition of this text. Nevertheless, these routes remain a favored approach to lesions in the anterior orbit. In this area are the orbital tumors that are palpated through the eyelids or conjunctival fornices.
Tumors frequently managed by these approaches are the dermoid cyst; carcinomas secondarily invading the orbit from a sinonasal source; the frontal mucocele extending into the superonasal orbit; the non-Hodgkin lymphomas clustered around the trochlea or protruding into the conjunctival fornix; the inflammatory tumor extending along the back of the orbital septum; some lymphangiomas, capillary hemangiomas, and rhabdomyosarcomas; and the orbitofrontal organizing hematoma without intracranial extension. Excluded from this list are the palpable lacrimal gland tumors. These lesions are worthy of a wider exposure through a lateral or superolateral orbitotomy because their size is often larger than expected, there is a potential for malignancy, and a tendency for soft tissue seeding if incompletely removed or biopsied. Also, the tumors in this anterior zone may be explored under local anesthesia if the patient is amenable to such management.
In all the anterior approaches except the transconjunctival route, the location and length of the incision is first outlined on the skin with a marking pen. A subcutaneous injection of 1% xylocaine with 1:100,000 epinephrine is then administered along this line primarily for hemostasis but also to augment anesthesia. The same solution may be injected for appropriate nerve blocks if the operation is performed under local anesthesia.
In all the orbital operative procedures, regardless of the approach, we prefer bipolar rather than unipolar cautery for direct hemostasis. We are also strong advocates of meticulous hemostasis throughout the course of the operation rather than deferring this task to later stages of the surgical procedure. Meticulous hemostasis, in addition to facilitating exposure, generally obviates the need for leaving a drain postoperatively.
Superior Approach
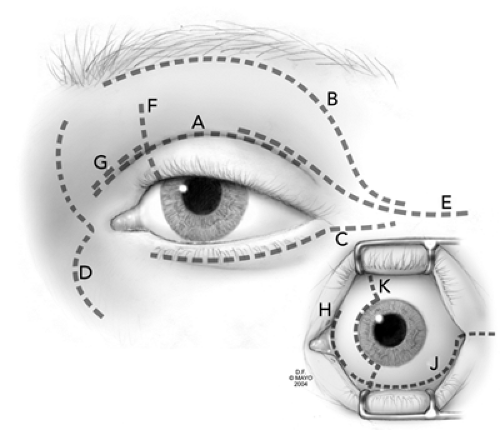
Superiorly there are three basic approaches, the lid crease, the sub-brow, and the transmarginal (lid-splitting)
incisions (see labels A, B, and F in Fig. 21.1). With each approach a preliminary marking of the superior orbital notch is made unless the location of the lesion is strictly confined to the superotemporal anterior orbit. This mark is an important reference throughout the subsequent dissection to the orbital exit of the supraorbital artery and nerve. Although the nicking of the artery is a source of annoying bleeding, the nerve is the more important of the two structures. We make every effort to preserve the nerve intact because surgical trauma to this structure usually results in a distressing postoperative anesthesia of the forehead. In the superior approach, we also try to work around the trochlea rather than disengaging this structure from its periosteal base.
incisions (see labels A, B, and F in Fig. 21.1). With each approach a preliminary marking of the superior orbital notch is made unless the location of the lesion is strictly confined to the superotemporal anterior orbit. This mark is an important reference throughout the subsequent dissection to the orbital exit of the supraorbital artery and nerve. Although the nicking of the artery is a source of annoying bleeding, the nerve is the more important of the two structures. We make every effort to preserve the nerve intact because surgical trauma to this structure usually results in a distressing postoperative anesthesia of the forehead. In the superior approach, we also try to work around the trochlea rather than disengaging this structure from its periosteal base.
The incision is curvilinear and parallel to the arch of the superior orbital rim and slightly below it. The midpoint of the incision lies over the palpable mass and the incision’s length can be extended nasally or medially to accommodate the necessary surgical exposure. The skin is incised either with a scalpel or a diathermy knife. We prefer to start with the blade that can be angled slightly to align parallel to the brow hair follicles that helps minimize the scar and preserve follicles. The diathermy or cutting cautery is best for incising the soft tissues underlying the skin.
If the goal of the operation is an evacuation of the cystic contents of a lesion in the peripheral orbital space or the curettement of a lesion extending into this space from the overlying bone (aneurysmal bone cyst, Langerhans cell histiocytosis, plasmacytoma of bone), the dissection is beveled toward the junction of the periorbita, periosteum, and orbital septum along the superior orbital rim. When this junction is incised, the levator complex and orbital contents with the covering periorbita are pushed downward and the peripheral space entered directly. As a rule bleeding is not a problem in this area except for the intrinsic vasculature of the lesion to be removed. At the completion of the operation, a simple approximation of orbital septum to periosteum is performed with interrupted sutures. A tight closure is not necessary. This will permit seepage of serum postoperatively from the peripheral space into the subcutaneous tissues of the upper eyelid.
If the goal of the operation is removal of a lesion in the anterior orbital space the orbit is entered through an incision beneath the arcus marginalis. The preaponeurotic fat is immediately encountered and can be manipulated or retracted with cotton-tipped applicators. In any manipulation of orbital fat, hemostasis must be particularly exquisite. Closure of the orbital septum need not be overly tight. In fact, a loose anatomic approximation is preferred. In this way, a retrobulbar hematoma or fluid collection under pressure may decompress itself before causing irreparable visual loss.
The principal premise of the lid-crease approach is cosmetic because the healed incision is hidden in a skin fold of the upper eyelid (Leone, 1979; Wolfley, 1985; Kronish and Dortzbach, 1988). Other advantages according to these publications are the excellent exposure contingent on the distensibility of the eyelid skin, the minimal dissection through familiar anatomic landmarks that is necessary to reach the offending lesion, and simple wound closure.
The superior palpebral fold usually is the site of the skin incision. If this fold is absent or poorly delineated, another eyelid crease can be selected so long as it is symmetrical with a skin fold in the other upper eyelid. An incision about 15 mm in length is made with a scalpel. Traction sutures can be inserted in the lips of the skin incision to augment exposure. The orbicularis muscle is tented with a forceps and incised down to the suborbicularis fascial plane. The orbitopalpebral mass is reached by blunt dissection along this plane.
Closure of the wound requires no deep sutures. In children the skin incision is closed with a continuous fine-diameter absorbable suture. In adults a nylon suture can be used. If it is necessary to deepen the lid crease at the time of closure, the lower margin of the skin incision can be anchored to the fascial covering of the underlying levator aponeurosis.
We find the lid-crease approach particularly suitable for the removal of small, well-circumscribed dermoid cysts that straddle the attachment of the orbital septum to the superior orbital margin. However, for those cysts and other lesions that are positioned posterior to the orbital septum, we believe the sub-brow approach provides better exposure and roomier working space.
Since the last edition of this book, the transmarginal eyelid incision (lid splitting) (Smith, 1966) has experienced a resurgence of interest (Kersten and Kulwin, 1999). This approach works quite well for lesions situated in the superior nasal quadrant, located primarily in the intraconal location but also for lesions straddling the intraconal/extraconal plane. We found excellent exposure from this incision for the lesion noted in Figure 21.2 (A and B). A straight iris scissor is used to create a perpendicular (to the lid margin) full thickness incision into the superior fornix. The incision is placed at the junction of the medial and central third of the eyelid. For reference, a previously placed superior rectus traction suture is helpful. Traction sutures on each lid margin will facilitate exposure into the orbit as shown (see Fig. 21.3A). Malleable retractors can be used to spread the tissues that expose the superior oblique tendon (Fig. 21.3B). The surgeon should now decide if working above or below the tendon will facilitate the procedure. In this example, the tumor was exposed by working above the tendon (Fig. 21.3C). After hemostasis was secured, the lid was repaired by a standard three-layered lid repair.
Inferior Approach
The inferior orbitotomy is the oldest of the several surgical approaches to orbital tumors. Compared to the superior approach, the inferior route traverses less important anatomic structures, is less vascular, and provides a wider view of the orbital space relative to the length of the incision. Nevertheless, the inferior orbitotomy is less frequently used than other surgical approaches because of a lesser number of primary orbital tumors in the inferior orbit. For the management of secondary tumors that invade the inferior peripheral space from a source in either the nasal cavity or maxillary antrum, the inferior orbitotomy is combined with some sinonasal procedure.
The anatomic objective of the inferior orbitotomy is to gain access to the junction of the periosteum of the maxillary bone with the periorbita covering the floor of the orbit and the fascia separating the tissues of the eyelid from the orbital space. This fascial divider corresponds in function and position to the orbital septum of the upper eyelid, but is much thinner. The skin overlying the inferior orbital rim also is very thin. This feature, combined with the tendency of healing skin to adhere directly to the underlying periosteum, creates a visible scar. This discourages skin incisions directly overlying the orbital rim.
To prevent this complication the incision is offset so that the skin can unite with the underlying layer of subcutaneous fascia or orbicularis muscle rather than the periosteum covering the bony orbital rim. This is done by placing the skin incision somewhere in the lower eyelid superior to the orbital rim. The incision is generally a millimeter or two below the lashes in a subciliary position (label C in Fig. 21.1). One could also use an incision slightly lower in a prominent wrinkle line, but after healing this may produce an obvious asymmetry compared to the other side.
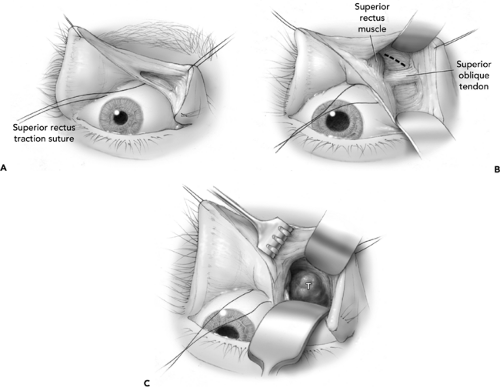 Figure 21.3 A: Transmarginal lid-splitting orbitotomy after lid is split and superior fornix is open. Traction sutures through the lid margin facilitate exposure. Preplaced superior rectus traction suture assists identification of the superior rectus muscle. B: With retractors in place superior oblique tendon is visualized. Tumor mass depicted in Figure 21.2 is shown by dotted line. C: Superior oblique tendon is reflected inferiorly to expose tumor mass. |
With either incision some tunneling along fascial planes is necessary to reach the orbital rim. The more inferior the incision the less tunneling required. With the inferior curvilinear incision the inferior rim is easily reached by tunneling between the skin and the surface of the orbicularis oculi muscle. This cleavage plane may also be used with the higher (subcilial) incision, or a plane established between the orbicularis oculi muscle and the thin fascial orbital septum.
Once the inferior orbital space is entered, visibility usually is adequate except for the tendency of the orbital fat to bulge into the entrance space. This fat may be retracted with cotton-tipped applicators or cottonoids. If the prolapsing fat is too exuberant the lobules can be shrunk with a light application of a desiccating current or a bipolar cautery. One must be cognizant of the location of the inferior oblique muscle when dealing with orbital fat during this approach to avoid postoperative diplopia. There are no major sources of hemorrhage in the inferoanterior orbital space but the blood-tinged fluid oozing from orbital fat should not be disregarded. If this source is not staunched it can result in a slowly progressive, postoperative hematoma infiltrating the overlying soft tissues of the eyelid. Closure of the orbital septum and the skin incision does not differ from that described for the superior orbital approach.
Transconjunctival Fornix Approach
If an orbital mass is easily palpated through the conjunctival cul-de-sac, or its bulge into the fornix is obvious, it is tempting to think the lesion can be easily removed by an approach through the conjunctival fornix. Orbital masses with such characteristics are usually seen in
the superotemporal cul-de-sac or the inferior fornix. To facilitate the conjunctival incision and exposure of such tumors, opposing malleable retractors are placed against the eyeball and the palpebral surface of the eyelid. Blunt and sharp dissection along the surface of the mass soon reveals that such tumors extend deeper into the orbit than first assumed. The dissection is prolonged and visibility is reduced by progressive bleeding. Therefore, the removal of the mass often proves incomplete or compromised in some way. Hemostasis in such situations is difficult and closure of the incision often is anatomically incomplete. The clinical result in such a case may be further complicated by a subsequent symblepharon. Obliteration of the orifices of the excretory ducts of the lacrimal gland may also occur if the incision is made in the superotemporal fornix.
the superotemporal cul-de-sac or the inferior fornix. To facilitate the conjunctival incision and exposure of such tumors, opposing malleable retractors are placed against the eyeball and the palpebral surface of the eyelid. Blunt and sharp dissection along the surface of the mass soon reveals that such tumors extend deeper into the orbit than first assumed. The dissection is prolonged and visibility is reduced by progressive bleeding. Therefore, the removal of the mass often proves incomplete or compromised in some way. Hemostasis in such situations is difficult and closure of the incision often is anatomically incomplete. The clinical result in such a case may be further complicated by a subsequent symblepharon. Obliteration of the orifices of the excretory ducts of the lacrimal gland may also occur if the incision is made in the superotemporal fornix.
For these several reasons, we do not encourage this approach for the routine removal of orbital tumors in the anterior space except for the circumscribed masses that may be near the anterior portion of an extraocular muscle as demonstrated by imaging.
Consequently, the transconjunctival approach is seldom used in our practice except to perform an incisional biopsy on an orbital mass that is pushing into the overlying cul-de-sac. The orbital tumors that frequently show this characteristic are non-Hodgkin lymphomas, sarcoid or sarcoidosis, and lymphangioma.
The exposure through a transconjunctival approach through the inferior fornix can be augmented and facilitated by two methods. If a lesion is located in the inferior-temporal, extraconal quadrant, one can transect the inferior crus of the lateral canthal tendon, the so-called swinging eyelid flap. (McCord and Moses, 1979) to enhance exposure (label J in Fig. 21.1). In fact, this will even facilitate exposure to lesions within the inferior nasal quadrant. Extension of the transconjunctival incision up across the caruncle (transcaruncular approach, see subsequent text) greatly enhances exposure in the inferior nasal quadrant (labels J and H in Fig. 21.1). One must anticipate the inferior oblique muscle. Shorr et al. (2000) either cut across the muscle at its origin and later resutured it or just reflected if off with the periorbita with no postoperative diplopia. After hemostasis is secured, the wound is secured by a simple conjunctival closure in which we use 6-0 plain gut sutures. If the inferior crus of the lateral canthal tendon has been taken down, it can be repaired as would any tarsal strip procedure.
Medial Approach
Lesions in the shaded area of Figure 21.4 are amenable to the several medial approaches that we will discuss. The curved contour of the superonasal orbital rim is the landmark for one of these approaches. The incision is gull-wing shaped over the medial canthal tendon to eliminate webbing. This is the standard external ethmoidectomy incision. Other options include a transmarginal, lid-splitting procedure (label F in Fig. 21.1) as discussed in the preceding text. One could also utilize the medial lid crease as shown in Figure 21.1 (label G). Ophthalmologists usually favor the latter approaches for lesions within the superior anteronasal space (the area between the periorbita and the eyeball), and rhinologists use the former for external ethmoidectomy, frontal sinusotomy, and lesions
bulging into the medial peripheral space from a source in the nasal cavity or paranasal sinus.
bulging into the medial peripheral space from a source in the nasal cavity or paranasal sinus.
In any of these approaches, a preliminary mark is made over the supraorbital notch as a reminder of the underlying neurovascular bundle. In addition several cross hatch marks may be made along the length of the external ethmoidectomy incision. These hatch marks serve as a guide for anatomic closure of the soft tissues at the completion of the operation, thus minimizing the puckering of tissues that so easily occurs in this quadrant of the orbit. The external ethmoidectomy incision is 25 to 30 mm in length and is roughly equal to one-fourth the circumference of the orbital rim. Any situation requiring wide exposure or extension below the medial canthal tendon is best served by incision D in Figure 21.1.
In the superonasal quadrant the sources of bleeding are the terminal branches of the frontal and dorsal nasal arteries in the area of the trochlea and the angular vein. The former vessels can be handled with the bipolar cautery but bleeding from the angular vein is better managed by clamping and ligation.
If the goal of the operation is to approach the anteronasal orbital space, the periosteum along the orbital rim need not be elevated. This space is entered as previously described under superior orbitotomy. If the intended approach is the peripheral orbital space, the periosteum is elevated to expose the ascending process of the maxilla and the lacrimal bone proximal to the posterior lacrimal crest. The periorbita likewise is elevated to expose the lamina papyracea and the orbital plate of the frontal bone. The main source of bleeding during this blunt dissection is the anterior ethmoidal artery. Closure of this artery by cautery is not very satisfactory. Instead, hemostasis is better secured by applying a small silver clip or ligation of the vessel.
The foramen of the anterior ethmoidal artery is an important topographic landmark in this dissection. It is located at the level of the cribriform plate and the junction between the ethmoid labyrinth inferiorly and the frontal bone superiorly. If the surgical procedure requires a more posterior extension of the dissection, the foramen of the posterior ethmoidal vessels marks the plane of the most posterior ethmoidal cells. If it is necessary to extend the incision inferiorly along the orbital rim, the periosteal stump of the medial canthal tendon should be preserved for reapproximation with its tendon at the time of closure of the incision.
If the periorbita and periosteum have not been too disrupted during the dissection, they can be loosely approximated at the time of closure. The subcutaneous tissue and skin can be closed in two layers.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree