Purpose
To determine the influence of omega-3 supplementation on vitreous vascular endothelial growth factor A (VEGF-A) levels in patients with exudative age-related macular degeneration (wet AMD) receiving intravitreal anti-VEGF treatment.
Design
Prospective, randomized, open-label, single-center, clinical trial, consecutive interventional case series.
Methods
The study included 3 cohorts with wet AMD and a control group with epiretinal membrane or macular hole. Twenty wet AMD patients being treated with anti-VEGF were randomized to daily supplementation of antioxidants, zinc, and carotenoids with omega-3 fatty acids (docosahexaenoic acid and eicosapentaenoic acid; group 1, n = 10) or without omega-3 fatty acids (group 2, n = 10). They were compared with an anti-VEGF treatment-naïve wet AMD group (group 3, n = 10) and an epiretinal membrane or macular hole group (group 4, n = 10). Primary outcome was vitreal VEGF-A levels (at the time of anti-VEGF injection). Secondary outcomes were plasma VEGF-A and central foveal thickness. Patients with new submacular hemorrhage or any other treatment within 3 months were excluded. Final analyses included 9, 6, 7, and 8 patients in groups 1 through 4, respectively.
Results
Patients receiving omega-3s (group 1) had significantly lower levels of vitreal VEGF-A (141.11 ± 61.89 pg/mL) when compared with group 2 (626.09 ± 279.27 pg/mL; P = .036) and group 3 (735.48 ± 216.43 pg/mL; P = .013), but similar levels to group 4 (235.81 ± 33.99 pg/mL; P = .215). All groups showed similar values for plasma VEGF-A and central foveal thickness measurements.
Conclusions
This study demonstrated that omega-3 supplementation combined with anti-VEGF treatment is associated with decreased vitreal VEGF-A levels in wet AMD patients.
Age-related macular degeneration (AMD) is the leading cause of blindness in older individuals in the Western world. The aging of baby boomers is expected to lead to a 2-fold increase in the number of white person 65 years of age or older by 2031. Correspondingly, a doubling in the number of North Americans with AMD is expected. The exudative (wet or neovascular) form of AMD is associated most widely with central vision impairment and legal blindness. The 15-year cumulative incidence of wet AMD in Americans 75 years of age or older is 4.4%. By 2020, in the United States alone, it is estimated that nearly 3 million individuals will be affected by wet AMD. The progressive nature of wet AMD, its substantial societal and personal impact, and its high prevalence make it essential to develop clinical strategies to reduce its impact. It represents an important cause of morbidity and presents direct financial burdens of more than $10 billion in direct annual medical costs in the United States and accounts for significant loss of productivity. Designing efficient and cost-effective treatment methods therefore is highly desirable.
The management of wet AMD was revolutionized by the introduction of anti–vascular endothelial growth factor (VEGF) therapies. Regrettably, 5% to 10% of patients proceed to lose 3 lines or more of visual acuity (VA), and most exudative lesions show some sign of activity by the end of follow-up. In addition, increased numbers of thromboembolic events, possible neuronal toxicity, and higher incidence of geographic atrophy in patients with more frequent anti-VEGF injections also may be of concern. Thus, developing alternative or adjunct therapies to currently available anti-VEGF drugs may increase treatment success, slow AMD progression, and improve VA outcomes.
The abnormal and disproportionate growth of choroidal vessels associated with wet AMD likely stems from a compensatory angiogenic response to overcome an earlier phase of microvessel degeneration and reinstate metabolic equilibrium to the hypoxic macula. A potential strategy to influence and reduce the progression of wet AMD comes from directly modulating the cellular make-up of the retina. In this respect, the outer retina is highly concentrated in diet-derived long-chain polyunsaturated fatty acids (LCPUFAs) such as docosahexaenoic acid (DHA) of the omega-3 family and arachidonic acid of the omega-6 family. The capacity of lipids to play biological roles beyond energy storage and membrane structure long has been recognized. Importantly, dysregulation in lipid signaling is a salient feature of conditions associated with chronic inflammation such as metabolic syndrome, atherosclerosis, asthma, allergic response, autoimmunity, hypertension, cancer, and importantly in the context of the current study, ocular vasoproliferative diseases.
Because humans are limited in their capacity to biosynthesize omega-3 LCPUFAs de novo, their tissue status is modifiable via diet or supplement intake of DHA and eicosapentaenoic acid (EPA). The benefits of omega-3 supplementation on wet AMD consistently have been recognized in multiple observational studies, and although null results have been reported in a well-nourished nutrient-supplementing cohort with moderate to high risk of AMD progression, a clearer understanding of the impact of omega-3 supplementation on wet AMD could prove beneficial for streamlining therapeutic strategies. Furthermore, a number of fundamental studies have demonstrated the beneficial effects of omega-3 metabolites DHA and EPA on pathologic angiogenesis. Based on the current experimental and epidemiologic data linking omega-3 LCPUFAs and their potential beneficial role in angiogenesis, the purpose of the present pilot trial was to investigate the influence of omega-3 supplementation on VEGF-A levels in the vitreous of patients undergoing anti-VEGF treatment for wet AMD.
Methods
This pilot, prospective, randomized, open-label, single-center clinical trial, consecutive, interventional case series was conducted between February and August 2011. The study conformed to the tenets of the Declaration of Helsinki, was approved by the Institutional Review Board of the Maisonneuve-Rosemont Hospital affiliated with the University of Montreal, Quebec, Canada, and is a registered trial ( ClinicalTrials.gov identifier, NCT01819415 ).
Participants
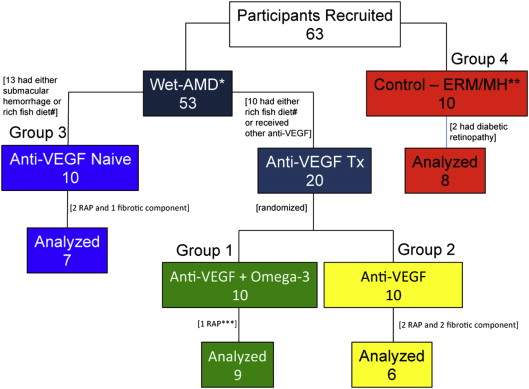
Sixty-three patients were screened for the study. Forty patients were deemed eligible participants and were enrolled at the Department of Ophthalmology Clinic, Maisonneuve-Rosemont Hospital, Montreal, after providing written informed consent ( Figure 1 ). Three cohorts consisted of active wet AMD patients (10 per group) who were eligible for anti-VEGF treatment (bevacizumab 1.25 mg/0.05 mL). They were compared with a non-AMD group with epiretinal membrane (ERM) or macular hole (MH; Figure 1 ). All participants were nonsmokers with regular consumption less than 1 serving of fish intake per week, according to a food-frequency questionnaire applied during recruitment.
Patients with wet AMD manifesting new thick submacular hemorrhage and those with treatment other than anti-VEGF or other anti-VEGF drugs within the last 3 months of study entry were ineligible.
Study Groups and Study Supplements
Twenty patients with active wet AMD who had undergone prior anti-VEGF treatment were divided in 2 groups and were randomized to receive oral supplementation as follows:
- 1.
Group 1 (n = 10): Vitalux plus Omega-3 (Alcon, Toronto, Ontario, Canada) 4 capsules/day; a formula containing the antioxidants β-carotene (5728 μg), vitamin C (500 mg), vitamin E (400 IU), zinc (25 mg), and copper (1 mg), as well as lutein (10 mg), zeaxanthin (2 mg), and omega 3 (1052 mg fish oil from sardine, mackerel, and anchovy [200 mg of DHA and 400 mg of EPA]).
- 2.
Group 2 (n = 10): Vitalux AREDS, 2 capsules/day, a formula containing the same concentration of antioxidants and minerals, lutein (10 mg), and zeaxanthin (500 μg; Figure 1 ).
- 3.
Group 3 (n = 10): patients with wet AMD starting anti-VEGF treatment (treatment naïve).
- 4.
Group 4 (n = 10): non-AMD patients with ERM or MH undergoing 25-gauge pars plana vitrectomy.
Patients from groups 3 and 4 were not taking any of the above-mentioned supplements.
Analytic Sample
We consecutively recruited 63 patients: 53 with wet AMD and 10 with ERM or MH. Of the wet AMD patients, 23 were excluded because of either higher omega-3 content in their diets, other anti-VEGF treatments, or new submacular hemorrhage. Of the 30 patients recruited with wet AMD, 8 were excluded from statistical analysis (1 from group 1, 4 from group 2, and 3 from group 3) because they either had retinal angiomatous proliferation or a large fibrotic component (more than 50%) of the choroidal neovascularization. Two of 10 patients with ERM or MH from group 4 also were excluded because they were found to have diabetes and mild nonproliferative diabetic retinopathy. A total of 22 patients with wet AMD (9 in group 1, 6 in group 2, and 7 in group 3) and 8 control patients were included for VEGF-A analysis ( Figure 1 ).
End Points
The primary outcome was vitreous VEGF-A levels, and secondary outcomes were plasma VEGF-A levels and central foveal thickness (CFT) measures. Vitreous and plasma VEGF-A levels were collected at the time of anti-VEGF treatment. At enrollment, we collected data on age, gender, number of previous anti-VEGF injections, time from last anti-VEGF injection, and Snellen visual acuity (converted to logMAR for statistical analysis; Table ).
| Parameter | Wet AMD | Control (Group 4) a | P Value | ||
|---|---|---|---|---|---|
| Anti-VEGF Plus Omega-3 (Group 1) | Anti-VEGF (Group 2) | Anti-VEGF Naïve (Group 3) | |||
| Age (y) | 79.6 ± 1.81 | 79.00 ± 1.98 | 83.38 ± 2.32 | 68.25 ± 3.56 | .0099 b |
| No. female (%) | 5 (56) | 3 (50) | 2 (29) | 7 (86) | .144 c |
| Visual acuity d | 0.61 ± 0.14 | 0.84 ± 0.17 | 0.78 ± 0.15 | n.a. | .6606 b |
| No. of previous injections | 8 ± 1.19 | 6 ± 1.51 | n.a. | n.a. | .5287 b |
| Time from last injection (wks) | 8 ± 0.40 | 8 ± 0.36 | n.a. | n.a. | .9999 b |
| No. of patients | 9 | 6 | 7 | 8 | |
a Epiretinal membrane and macular hole patients.
c Fisher exact probability test.
Anti–Vascular Endothelial Growth Factor Treatment Protocol
The anti-VEGF treatment regimen consisted of 3 loading doses followed by pro re nata injections based on disease activity measured monthly by spectral-domain optical coherence tomography (Cirrus, Carl Zeiss Meditec, Toronto, Canada). Fluorescein angiography also was performed on all patients with wet AMD on the day of the anti-VEGF injection (when vitreous biopsy and blood samples were collected).
Vitreous Biopsy
After the surgical field was sterilized using 5% povidone–iodine, patients were draped in a standard manner with placement of a lid speculum. A 27-gauge self-retaining infusion line (Insight Instruments, Stuart, Florida, USA) of balanced salt solution was placed first, followed by the placement of a 29-gauge trocar with a chandelier light connected to a mercury vapor light source (Synergetics, O’Fallon, Missouri, USA). The surgical view during the procedure was provided through a surgical operative microscope and a Volk contact lens (Volk direct image ×1.5 magnifying disposable vitrectomy lens; Volk Optical, Mentor, Ohio, USA).
The vitreous biopsy was performed using a 23-gauge sutureless Retrector system (Insight Instruments) in all patients. The model used in the study is a portable, battery-powered system with a maximum cut rate of 600 cpm (cuts per minute) and features a retractable sheathed guillotine 25-gauge cutter with an in-built needle (23 gauge). The needle was introduced bevel down through displaced conjunctiva in an oblique 1-plane tunnel into the vitreous cavity 3 to 4 mm from the limbus. At least 0.5 mL of undiluted vitreous fluid was cut and removed from the vitreous right above the macular region (at the premacular bursa) through controlled manual aspiration with the Retrector system. Bevacizumab 2.5 mg/0.1 mL was injected through the 29-gauge trocar after the vitreous biopsy.
The samples were split in 3 vials: 1 for VEGF-A levels, 1 for lipidomics analysis, and 1 for microbiologic analysis (to verify any contamination during vitreous biopsy). The entire procedure was performed in the minor procedure room within the Department of Ophthalmology Clinic at Maisonneuve Rosemont Hospital, Montreal, Canada.
Assessment of Vascular Endothelial Growth Factor A Levels
Vitreous and plasma samples were frozen on dry ice and immediately were stored at −80 C after biopsy, then centrifuged at 15 000 g for 5 minutes at 4 C before analysis. For plasma analysis, 5 mL venous blood was collected before vitreous biopsy and centrifuged at 3000 g for 15 minutes at 4 C to obtain plasma and was stored at −80 C until assayed. VEGF-A levels were quantified in supernatants using enzyme-linked immunosorbent assays according to manufacturer’s instructions (R&D Systems, Minneapolis, Minnesota, USA).
Statistical Analysis
Statistical analysis was performed using the 2-way analysis of variance nonparametric test, the nonparametric t test (Mann–Whitney U test), parametric Student t test, and the Student t test (GraphPad Prism). We applied the Fisher exact probability test to examine differences in the proportions of women and men in each group. All statistical analysis were performed using the same software (GraphPad Prism, La Jolla, California, USA). Comparisons across all groups yielded an exact P value of .144, suggesting no appreciable differences. Respective P values for comparisons of these proportions across people with wet AMD (groups 1, 2, and 3), between people with wet AMD in the clinical trial (group 1 vs group 2) and all people with AMD vs people with ERM or MH (combined groups 1 through 3 vs group 4) were 0.568, 0.376, and 0.092, respectively. All P values are 2-tailed. P values less than .05 were considered statistically significant. Data are expressed as mean ± standard error of the mean.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


