Purpose
To describe nonglaucomatous retinal nerve fiber layer (RNFL) defects in patients with Behçet uveitis.
Design
Cross-sectional study and observational case series.
Methods
We reviewed the clinical photographs of patients with Behçet uveitis (n = 259), ocular toxoplasmosis (n = 120), and multiple sclerosis (MS)-associated uveitis (n = 40) for the presence of localized RNFL defects. Behçet patients with localized RNFL defects were invited to participate in a prospective evaluation including standard automated perimetry, spectral-domain optical coherence tomography (SD OCT), and RNFL thickness analysis. Main outcome measures were the prevalence of localized RNFL defects, perimetric and SD OCT findings, and RNFL thickness analysis.
Results
Sixty-two patients with Behçet uveitis (24%) had localized RNFL defect(s) without any visible scar. Twenty patients (17%) with ocular toxoplasmosis had a localized RNFL defect associated with a retinochoroidal scar in all. None of the MS patients had a localized RNFL defect. Of the 19 patients (24 eyes) with RNFL defect associated with Behçet uveitis who returned for follow-up, there was a corollary thinning on SD OCT B-scan in all and a corresponding visual field defect in 83%. RNFL thickness analysis was within normal limits in 54%, but revealed thinning in the thickness profile scale in 46% and in the pie chart in 21%.
Conclusion
In Behçet uveitis, localized RNFL defects may be caused by microvascular ischemia at the optic nerve head and/or at the posterior pole and may serve as a helpful ocular diagnostic clue and an indicator of posterior pole involvement, a risk factor for poor visual prognosis.
Behçet uveitis is classically described as a panuveitis characterized by occurrence of occlusive retinal vasculitis, vitritis, retinal infiltrates, and hypopyon. The disease has a relapsing-remitting course and ocular findings vary during the course of the disease. The appearance of retinal infiltrates denotes an activation of intraocular inflammation in the posterior segment. Retinal infiltrates may be superficial or deep, with or without accompanying retinal hemorrhages. They may be seen as solitary or multifocal, and of variable size and distribution. These infiltrates resolve typically without visible chorioretinal scarring. We have been observing localized retinal nerve fiber layer (RNFL) defects as sequelae of superficial retinal infiltrates affecting the posterior pole in patients with Behçet uveitis. Because of the transient nature of acute inflammatory signs, it is of utmost importance for the differential diagnosis of Behçet uveitis that the characteristic sequelae are recognized during the remission periods.
Localized RNFL defects have been described in glaucoma and toxoplasmic retinochoroiditis. Our aim was to investigate the occurrence of nonglaucomatous localized RNFL defects in patients with Behçet uveitis, ocular toxoplasmosis, and multiple sclerosis (MS)-associated uveitis. Herein we describe localized RNFL defects in Behçet uveitis that may serve as an ocular diagnostic clue.
Subjects and Methods
Fundus photographic archives of the Uveitis Service at the Department of Ophthalmology, Istanbul Faculty of Medicine, dating from November 2006 to February 2013, were used for the purpose of this study. The retrospective study protocol was approved by the Ethics Committee of Istanbul University, Istanbul Faculty of Medicine. The research follows the tenets of Declaration of Helsinki.
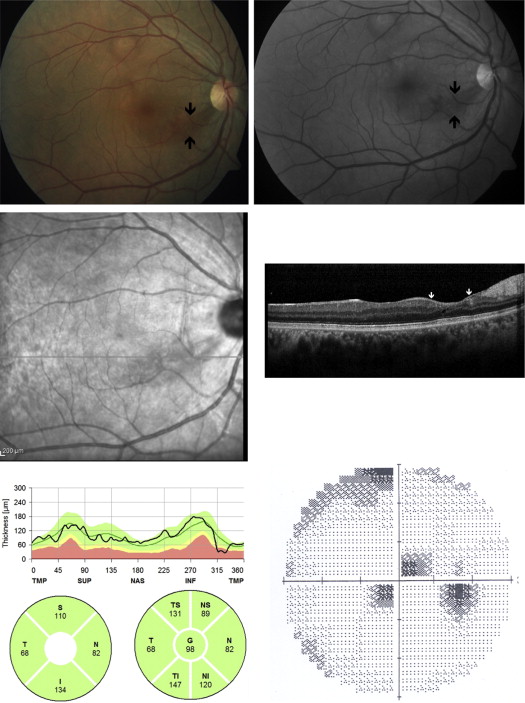
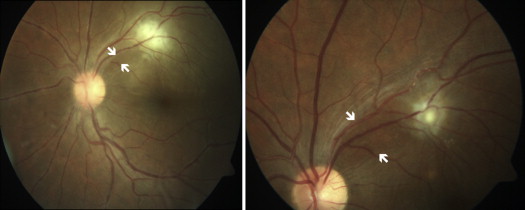
Two of the authors (M.O., I.T.-T.) independently reviewed color and red-free fundus photographs of 259 patients filed with the diagnosis of Behçet uveitis, 120 patients with toxoplasmic retinochoroiditis, and 40 patients with uveitis associated with MS. A localized RNFL defect was defined only when both reviewers agreed on the presence of this finding. A localized RNFL defect was previously defined in glaucoma patients as a wedge-shaped and/or slit-like area. In the present study, localized slit-like/wedge-shaped RNFL defects in patients with Behçet uveitis were defined as papillomacular bundle defects ( Figure 1 ) and inferior/superior arcuate defects ( Figure 2 ) based on the location.
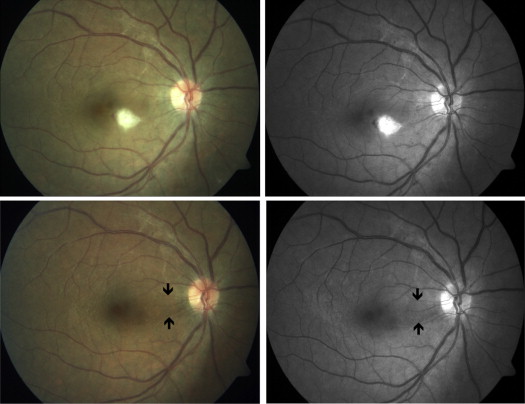
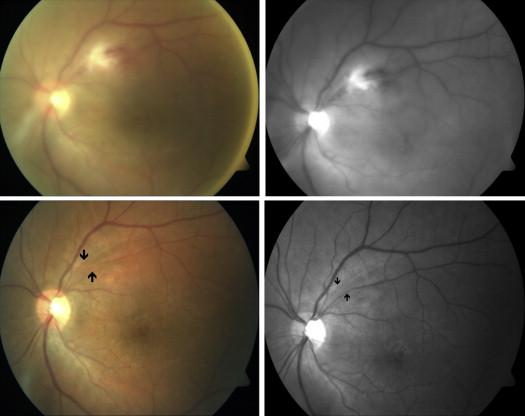
Color fundus photographs of patients with Behçet uveitis mostly comprised those taken at the time of fluorescein angiography (FA), and also those taken at the time of a posterior uveitis attack and after the resolution of a previous uveitis attack. Since the vitreous haze may not allow FA at the onset of severe attacks, it is mostly performed to evaluate the magnitude of retinal capillary leakage and extent of retinal capillary nonperfusion later in the disease course. In our clinic FA is also routinely performed in patients with Behçet uveitis who are in a clinically quiescent period to evaluate the therapeutic response, especially when tapering or discontinuation of treatment is considered.
The diagnosis of Behçet disease is based on the criteria set of the International Study Group for Behçet’s Disease. Color fundus photographs of patients with ocular toxoplasmosis have been taken when there was an active retinochoroiditis and over the follow-up of such lesions. Color fundus photographs of patients with MS uveitis have been routinely taken before performing FA.
Exclusion criteria in the Behçet uveitis group included end-stage findings and clinical evidence of glaucomatous damage (glaucomatous changes at the optic disc, such as undermining of the cup, diffuse or focal thinning of the rim, dot sign, and/or nasal shifting of retinal vessels). End-stage ocular involvement of Behçet disease was previously defined as eyes with total optic atrophy, occluded vessels that look like white cords, and diffuse retinal atrophy with variable scarring and pigmentation. Seventy-six eyes of 62 Behçet patients were identified with localized RNFL defect(s) on color and red-free fundus photographs. Among these, 48 patients could be contacted by telephone and invited for further evaluation, and 28 of them agreed to participate in this study. Clinical records of these patients were reviewed for age, sex, and disease duration. Patients also completed visual field, spectral-domain optical coherence tomography (SD OCT), and RNFL analyses.
Color and red-free fundus photographs were acquired with a digital camera (Carl Zeiss Meditec, Hennigsdorf, Germany) after maximum pupil dilation. Photographs showing the RNFL defect at the posterior pole were taken at 50-degree field of view and reviewed on an LCD monitor. Standard automated perimetry was performed using the Humphrey automated field analyzer HFA III 750 (Humphrey Systems, Inc, Dublin, California, USA) with the Swedish interactive threshold algorithm (SITA) standard 30-2 algorithm. The reliability of the visual field was evaluated and the test was considered reliable if fixation losses and false-negative and false-positive rates were less than 20%.
Optical coherence tomography scanning and RNFL thickness measurements were performed using the Spectralis Heidelberg Retina Angiography (HRA) and OCT device (Heidelberg Engineering, Heidelberg, Germany). For evaluation of the RNFL both the pie chart and RNFL thickness profile scale were reviewed. If there was thinning in any quadrant or sector, it was considered as an abnormal RNFL thickness on the pie chart. On RNFL thickness profile scale, values of the scan around the optic disc are displayed with a black line. If the average thickness indicated by the black line is below the normative database it is defined as abnormal.
The statistical analysis included descriptive statistics for demographic data and clinical findings.
Results
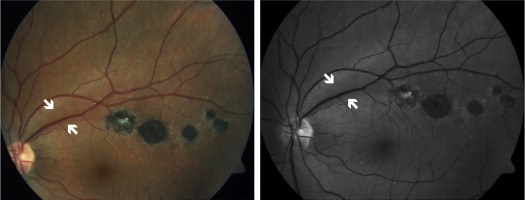
Sixty-two Behçet patients out of 259 (24%) revealed localized RNFL defect(s), and 15 patients had bilateral defects. Median disease duration was 24 months (range, 2–204 months). Sixteen of 62 patients (25.8%) had disease duration less than 1 year. Forty-six eyes out of 77 (60%) revealed papillomacular bundle defect, 23 eyes (30%) revealed superior/inferior arcuate defects, and 8 eyes (10%) showed co-occurrence of both defect types. One hundred thirty-five eyes of 120 patients with ocular toxoplasmosis revealed a visible chorioretinal scar. In 75 of the eyes, the scar was at the posterior pole. Among these, 20 eyes (27%) had a localized RNFL defect ( Figure 3 ). Serial color fundus photographs of a patient revealed the evolution of the RNFL defect over 3 weeks of follow-up ( Figure 4 ).
A localized RNFL defect was not seen in any of the eyes in the MS group, including 7 eyes of 4 patients with diffuse optic disc pallor.
Among 35 eyes of 28 patients with Behçet uveitis who returned for further evaluation, 3 eyes of 3 patients with a diagnosis of glaucoma and 8 eyes of 6 patients with low reliability in visual field testing were excluded from analysis. Therefore, data of 24 eyes of 19 patients were included. None of the patients had end-stage ocular and/or neurologic involvement of Behçet disease and/or previous optic neuritis. No patient had active uveitis at the time of his or her visual field, SD OCT, and RNFL analyses.
Mean age of the patients was 33.3 ± 7.1 (range, 21–46) years. Fourteen patients were male and 5 patients were female. At the time of the study, the mean disease duration was 40.9 ± 35.7 (range, 12–144) months. There were papillomacular bundle defects in 14 eyes, superior/inferior arcuate defects in 8 eyes, and co-occurrence of these in 2 eyes. Although all patients had bilateral involvement of Behçet uveitis, 14 patients revealed unilateral localized RNFL defect.
Twenty-one eyes out of 24 (83%) revealed visual field defect corresponding to the localized RNFL loss. Corollary thinning was detected on OCT B-scan sections passing through the clinically visible localized RNFL defects in all eyes; however, RNFL thickness analysis was within normal limits in 13 eyes (54%). Retinal nerve fiber layer analysis revealed nerve fiber layer thinning in 5 eyes (21%) in the pie chart and in 11 eyes (46%) in the RNFL thickness profile scale ( Figure 5 ). Mean central macular thickness on OCT was 268.4 ± 28.7 μm (range, 197–327 μm).