Purpose
To describe new options for diagnosis and severity grading of dry eye disease.
Design
Perspective on technological advancements to identify tear dysfunction and their value in diagnosing and grading dry eye disease.
Methods
Evidence is presented on new and evolving technologies to measure tear stability, composition, and meniscus height and their role in dry eye diagnosis and therapeutic efficacy grading is assessed.
Results
Evolving concepts regarding pathogenesis and new technologies to evaluate the tears and ocular surface have improved the ability to diagnose, classify, and grade the severity of dry eye disease. New technologies include noninvasive imaging of tear stability and tear meniscus height as a measure of tear volume and tear composition (osmolarity, lacrimal factors, inflammatory mediators, growth and differentiation factors). Approved tests, such as tear osmolarity and tear imaging, are being integrated into clinical practice and may eventually supplant certain traditional tests that have greater variability and less sensitivity. Other tests, such as molecular assays of tears and conjunctival cells, are currently being used in studies investigating pathogenesis and therapeutic mechanism of action. They may eventually translate to routine clinical practice.
Conclusions
New technologies have emerged that can noninvasively evaluate the tears and measure disease-associated compositional changes. These tests are being integrated into clinical practice and therapeutic trials for diagnosis, classification, and severity grading of dry eye disease.
The past few years have provided a number of advancements in the understanding of dry eye disease, particularly with regard to new testing options for diagnosis and grading of severity of this condition. The term keratoconjunctivitis sicca was created to describe the predominantly aqueous deficient dry eye found in Sjögren disease. Although historically the term dry eye has been applied, alternate terminology of dysfunctional tear syndrome was recommended by the Delphi panel in 2006 to emphasize that aqueous deficiency was not the only cause of tear dysfunction. The International Workshop on Dry Eye (DEWS) expanded the classification of dry eye disease in 2007. This Perspective describes the rationale and clinical application of new methods of diagnosis and evaluation of tear dysfunction present in dry eye disease and dysfunctional tear syndrome.
Current Definition and Classification of Dry Eye Disease/Dysfunctional Tear Syndrome
The Report of the International Workshop on Dry Eye Disease (DEWS) was published in 2007 and defines dry eye as a multifactorial disease of the tears and ocular surface that results in symptoms of discomfort, visual disturbance, and tear film instability with potential damage to the ocular surface. It is accompanied by increased osmolarity of the tear film and inflammation of the ocular surface. The classification system proposed by the same workshop ( Figure 1 ) identified separate categories of aqueous deficient dry eye and evaporative dry eye as distinct etiologic entities, both of which may occur in any given patient. Recent research has revealed that both mechanisms frequently coexist and that the evaporative form is more frequent than aqueous deficiency alone.
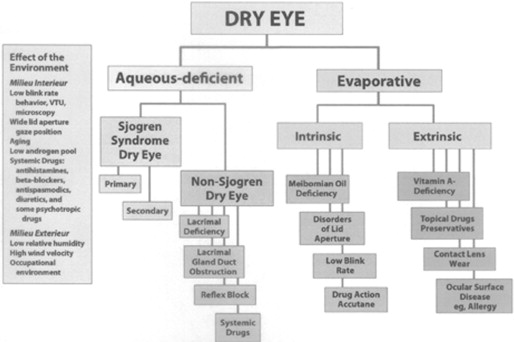
A further thesis of the DEWS Report was that elevated tear osmolarity and inflammation of the ocular surface/lacrimal system were critical elements in the pathogenesis of the disease. Recent research confirms the importance of both elevated tear film osmolarity and inflammation, as well as the interrelationship between those features. Methods for measurement of both osmolarity and inflammation have improved and are now available as point-of-service testing.
Identifying Disease of the Lacrimal Functional Unit
The lacrimal functional unit regulates production, distribution, and clearance of tears to meet ocular surface demands. Disruption of this integrated functional unit at any point can result in tear instability, clinically significant tear dysfunction, and ocular surface inflammation. Both lacrimal gland disease/dysfunction with tear hyposecretion or meibomian gland disease with increased tear evaporation can reduce tear volume. In contrast, diseases that interfere with tear distribution and clearance, such as conjunctivochalasis, commonly have normal or even elevated tear volume but an abnormal tear composition that is associated with tear instability and ocular surface epithelial disease. Ocular irritation and ocular surface dye staining, traditional measures to diagnose and gauge severity of tear dysfunction, are common to all conditions affecting the lacrimal functional unit. Tests to accurately evaluate tear stability and measure tear production/volume may improve the ability to identify disease in the lacrimal functional unit and diagnostic classification of tear dysfunction. Furthermore, there is increasing evidence suggesting that molecular assays to measure lacrimal secreted tear constituents or levels of inflammatory/differentiation factors in the conjunctival epithelium may also be useful parameters for diagnosis and severity grading.
Identifying Disease of the Lacrimal Functional Unit
The lacrimal functional unit regulates production, distribution, and clearance of tears to meet ocular surface demands. Disruption of this integrated functional unit at any point can result in tear instability, clinically significant tear dysfunction, and ocular surface inflammation. Both lacrimal gland disease/dysfunction with tear hyposecretion or meibomian gland disease with increased tear evaporation can reduce tear volume. In contrast, diseases that interfere with tear distribution and clearance, such as conjunctivochalasis, commonly have normal or even elevated tear volume but an abnormal tear composition that is associated with tear instability and ocular surface epithelial disease. Ocular irritation and ocular surface dye staining, traditional measures to diagnose and gauge severity of tear dysfunction, are common to all conditions affecting the lacrimal functional unit. Tests to accurately evaluate tear stability and measure tear production/volume may improve the ability to identify disease in the lacrimal functional unit and diagnostic classification of tear dysfunction. Furthermore, there is increasing evidence suggesting that molecular assays to measure lacrimal secreted tear constituents or levels of inflammatory/differentiation factors in the conjunctival epithelium may also be useful parameters for diagnosis and severity grading.
Noninvasive Measurement of Tear Stability and Tear Volume
Tear stability has been traditionally measured by fluorescein tear break-up time; however, several instruments have been introduced in the past decade that use image analysis software to evaluate tear smoothness and stability noninvasively in sequentially captured images of ring mires reflected off the precorneal tear layer. Studies using these devices have found more rapid and extensive ring distortion (break-up) in tear dysfunction that correlates with severity of corneal epithelial disease. These noninvasive devices appear to have advantages over the conventional invasive fluorescein break-up method because they evaluate more data points and the software can detect alterations that may not be visually apparent. It is likely that these noninvasive methods will gain popularity for clinical evaluation and as therapeutic endpoints.
Until recently, there have been no commercially available clinical tests to evaluate tear meniscus dimensions as a measure of tear volume. Clinicians have relied on the Schirmer test as an indirect measure of tear production and volume; however, it is widely recognized that this test is variable and capable of inducing reflex tearing that can mask low basal tear production and volume. Biomicroscopic measurement of tear meniscus height and use of reflective meniscography have not gained widespread acceptance. Noninvasive direct high-resolution measurement of tear meniscus dimension by anterior segment optical coherence tomography represents a major advance in the ability to gauge tear volume in unstimulated basal conditions. A number of published studies have used optical coherence tomography to compare tear volume to clinical ocular surface disease parameters and have found that optical coherence tomography has the potential to sub-classify tear dysfunction. Optical coherence tomography–measured inferior tear meniscus height was found to be a surrogate for tear volume. This parameter was reduced in patients with aqueous tear deficiency and correlated with clinical parameters, including noninvasive tear break-up time and severity of corneal fluorescein staining. An inferior tear meniscus height <0.30 mm was found to have sensitivity of 67% and specificity of 81% for dry eye using the Japanese Dry Eye criteria. The ability of this technology to stratify tear dysfunction based on tear volume may improve diagnostic classification for epidemiologic studies, therapeutic decision making, and clinical trials. Alex and associates found a moderate statistically significant inverse correlation between optical coherence tomography–measured tear meniscus height and severity of corneal fluorescein staining in response to a low-humidity environment.
Measurement of Compositional Changes of Tear Film
Osmolarity of the tear film has been measured in the research setting for many years. Determination of osmolarity can be measured by freezing point depression, vapor pressure, or electrical impedance/conductance of a solution. The Clifton osmometer was most often used to measure freezing point depression but was cumbersome in the handling of microvolumes of tear and prolonged duration of measurement. The Advanced Instruments Nanoliter Osmometer (Advanced Instruments, Inc, Norwood, Massachusetts, USA), although less cumbersome, was also hampered by the manipulation of small quantities of tear with micropipettes. The Wescor vapor pressure osmometer (Wescor, Inc, Logan, Utah, USA) was easy to use for larger volumes of fluid but not reproducible for small samples of clinically obtainable tears. The TearLab instrument (TearLab, Inc, San Diego, California, USA) accurately measures osmolarity of 50-nanoliter microvolumes of tears in a rapid assay based upon lab-on-a-chip measurement of electrical impedance/conductance. The TearLab instrument is most adaptable to use in the clinic setting and has received Food and Drug Administration clearance for such use. This technique is highly accurate, with a variance of less than 1% in control solutions.
Elevated osmolarity of the tear film occurs in all types of dry eye and thus cannot differentiate aqueous-deficient from evaporative dry eye. It is, nonetheless, a reliable indicator of dry eye and now can be measured in the clinic setting in a rapid assay with the TearLab system.
Reduced levels of various constituent proteins in the tear film have been documented in tear dysfunction. Historically these proteins have been measured by agar diffusion, radial immunodiffusion, electrophoresis, enzyme-linked immunosorbent assay, and mass spectroscopy. One of the first proteins found to be reduced in dry eye disease was lysozyme. It was measured by the degree of diffusion into a gel of agar infused with Micrococcus lysodiekticus bacteria over a 3-day incubation. Lactoferrin has also been shown to be reduced in tears with tear dysfunction by both radial immunodiffusion and enzyme-linked immunosorbent assay testing. High-performance liquid chromatography further has revealed reduced lactoferrin levels, as has mass spectroscopy. Elevated levels of matrix metalloproteinases have been measured in dry eye disease and probably reflect related inflammation.
More sophisticated testing by proteomic analysis has been applied to evaluation of tear proteins and promises to amplify our understanding of both normal and abnormal tear physiology. The complexity of the multiple proteins in tears in both health and disease requires comprehensive and precise measurement of individual proteins and their interactions. At the present time up to 1543 different proteins have been identified in normal tears. Patterns of altered protein composition in dry eye also have been identified. Although the sophisticated equipment needed to perform such proteomic testing is still limited to academic centers, reports of techniques applicable to the clinical setting are emerging.
Measurement of Inflammatory/Differentiation Biomarkers
As noted, biomarkers are an evolving option for diagnosis and severity grading of tear dysfunction. In 1998, the National Institutes of Health Biomarkers Definitions Working Group defined a biomarker as “a characteristic that is objectively measured and evaluated as an indicator of normal biological processes, pathogenic processes, or pharmacologic responses to a therapeutic intervention.” Biomarkers assessing lacrimal gland function and ocular surface health and differentiation have been identified. Immunoassays and biochemical methods have been used to detect proteins secreted by the lacrimal glands (eg, lactoferrin, epidermal growth factor) or produced by epithelial or inflammatory cells on the ocular surface. Studies have reported reduced concentrations of lacrimal proteins, increased concentrations of inflammatory mediators, and altered concentrations of mucins or differentiation-associated proteins (eg, MUC5AC) in patients with aqueous tear deficiency. Elevated S100AB and A9 peptides were detected in tears of patients with meibomian gland disease. Increased matrix metalloproteinase-9 activity in the tears has been found in aqueous tear deficiency and meibomian gland disease. Potential problems with tear assays that increase variability of test results include inability to obtain an adequate sample in patients with low tear volume or induction of reflex tearing during sample collection.
As an alternative to tear-based assays, differentiation factors and inflammatory mediators can also be assayed in conjunctival cells removed from the conjunctival surface with a brush or membrane. Proteins have been measured by immunostaining or flow cytometry and levels of mRNA transcripts by polymerase chain reaction. Cellular expression of biomarkers provides a snapshot of the cell status since they reflect events at the instant of collection, whereas tear sampling, annoyingly influenced by the potential for reflex tearing, may provide a more blurred picture.
The Table lists biomarkers that have been found to have moderate to strong correlation with clinical parameters, or have changed in response to treatment in clinical trials.
| Marker | Clinical Correlation | Reference |
|---|---|---|
| HLA-DR | Decreased with CsA and tofacitinab treatment | |
| MMP-9 | Symptom severity, corneal fluorescein staining, conjunctival lissamine green staining | |
| Tear EGF | Ocular surface rose Bengal staining, corneal fluorescein staining, conjunctival lissamine green staining | |
| Tear IL-6 | Ocular surface rose bengal staining, corneal fluorescein staining, conjunctival lissamine green staining | |
| Tear IL-8, MIP-1α, IL-1β | Corneal fluorescein staining, conjunctival lissamine green staining, | |
| Tear CXCL9 (I-TAC) | Basal tear secretion, keratoepitheliopathy, goblet cell density | |
| Tear proteins | In subjects with MGD | |
| S100A8 and A9 | Grittiness, transient blur | |
| Lactoferrin | Eye pain and tearing | |
| Lipocalin | Tearing, lid heaviness | |
| MUC16 mRNA | Tear meniscus | |
| MUC16 cellular | Lissamine green staining, Dry eye symptom questionnaire | |
| MUC5AC tears | Lissamine green staining |
Until recently, assays to measure inflammatory biomarker mediators in tears and conjunctival epithelium have been performed in research studies at academic centers to evaluate risk factors and pathogenesis. A semiquantitative commercial immunoassay to measure tear matrix metalloproteinase-9 has been approved in Europe, Canada, and, most recently, the United States.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


